Part –III: Utilities of Active Methylene Compounds and Heterocycles Bearing Active Methyl or having an Active Methine in the Formation of Substituted and Fused Pyridines
This review discusses how we use the most common and novel synthesized active methylene compounds as well as heterocycles having active methyl or methine in the syntheses of a wide variety of substituted and fused pyridines interesting medical and pharmaceutical importance. Many synthetic approaches were used for the preparation of target heterocyclic systems such as cyclocondensation reactions, ring opening-ring closure, cycloaddition, acid-or base-catalyzed reaction, intermolecular cyclization and self-condensation have been reviewed in this paper. Also, the antimicrobial activity carried out on some newly selected synthesized pyridines and their fused derivatives were reported.
## I. INTRODUCTION
Pyridine derivatives are important heterocyclic systems whose preparation, reactivity, and properties are of continuing interest. The biological activity associated with naturally occurring and synthetic pyridines has led to the development of pyridine-containing medicinal scaffolds and investigations into their pharmacological properties [1]. Benzopyridines specially quinolinones are the effective moiety in a large number of natural and synthetic heterocyclic compounds that exhibit significant antibiotic activity with a wide variety of significant medicinal, pharmacological, and industrial applications[2]. In continuation to our recent target in setting on some heterocyclic reviews discuss utility of active methylene compounds and heterocycles bearing active methyl or having an active methine in their structures in heterocyclization of five-, six-, and seven-membered heterocyclic systems of important applications in many fields such as medicine, agriculture, pharmacology and pharmaceutical. consequently, we recently publish some reviews [3-6] and this review summarized our work done in the last three decades and it involves the methods developed for the syntheses of substituted and fused pyridine derivatives using the titled compounds and reported the antimicrobial activity of some selected synthesizes compounds.
## II. FORMATION OF SUBSTITUTED PYRIDINES
Some active methylene compounds such assmalononitrile, cyanoacetohydrazide, caynoacetamide, cyanothioacetamide, and an enaminone were used for the synthesis of some pyridines bearing heterocyclic substituents and tested their biological activities.
### a) Using Synthesized Enones
$\alpha$, $\beta$ -Unsaturated ketones are called chalcones or enones was synthesized via the interaction of aromatic aldehydes with compounds having active methylene or methyl groups in their structures. The activated influence of the carbonyl group on the exocyclic double bond render the enones susceptible to the cycloaddition reaction forming the target pyridines. The preparation of a novel pyridines bearing substituted heterocyclic systems requires the formation of biheterocyclicenones such as the enone (3), which synthesized via Knoevenagel condensation of 3-formylchromone (1) with thiazolidene-2, 4-dione (2) having an active cyclic methylene group [7]. Whereas, the biheterocyclic enone 6 performed by condensation of formyl derivative of pyrazolobenzothieno-pyrimidinidine (4) with 4-acetylpyridazinone 5 having an active methyl group \[8\](Scheme 1).
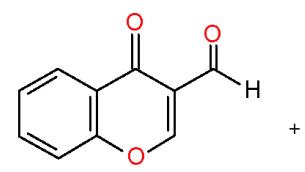 (1)
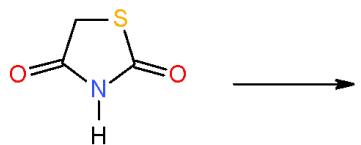 (2)
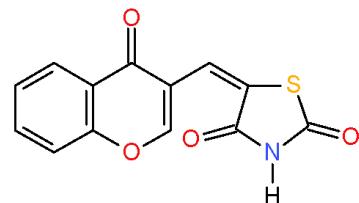 (3)
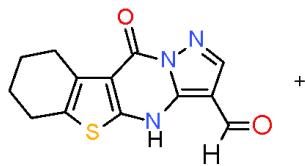 (4)
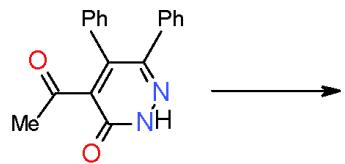 (5)
 (6) Scheme 1: Formation of bi-heterocyclic enones
The action of malonitrile (7) upon the two enones 3 and 6 showed two different behaviors. The presence of chromone with an active methine at C2 in the enone 3 facilitate the attack by the carbanion at C2 causing ring-opening of the $\gamma$ -pyron ring followed by ting closure producing the target pyridine. Thus, the reaction of chromenylthiazolidine derivative 3 with malonitrile (7) in basic medium afforded the intermediate 8, which underwent Dimorth rearrangement under the reaction condition to yield oxopyridinecarbonitrile 9. Compound 9 also obtained directly by the action of cyanoacetamide (10) upon biheterocyclicinenone 3 \[7\](Scheme 2).
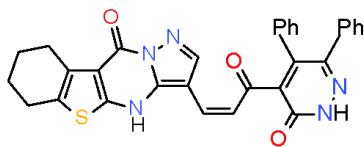
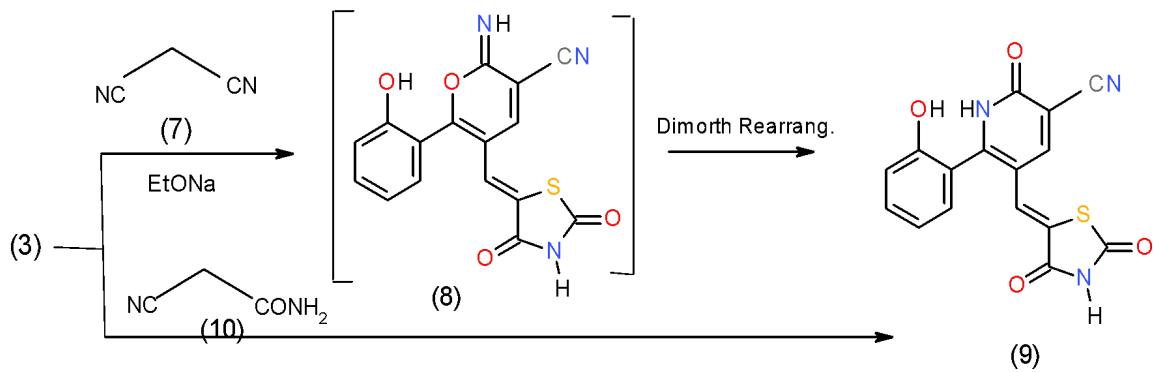 Scheme 2: Formation of oxopridinecarbonitrie When biheterocyclic enone 6 was subjected to react with malononitrile (7) in boiling ethanol containing ammonium acetate, cyclocondensation took place at $\alpha$, β-unsaturated carbonyl part yielding 4, 6-diheterocyclylpyridinecarbonitrile 11\[8\](Scheme 63).
 (6)
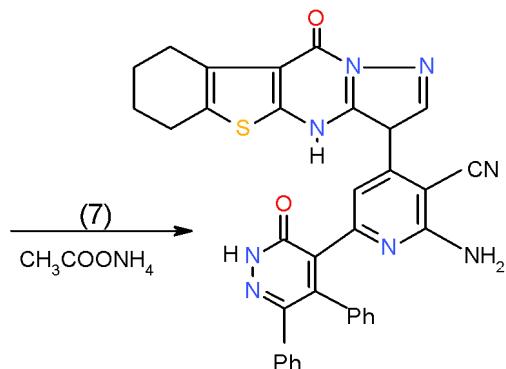 (11) Scheme 3: Formation of aminopridinecarbonitrie
Similarly, another two synthesized enones having quinolone in their structures was prepared, the enone 13 obtained by condensation of formylchromone cyanothioacetamide (16) and the enones 13 and 15 was carried out in ethanol containing piperidinium acetate, the respective 4,6-diheterocyclylpyridines 17 and 18, was obtained. But when the reaction between 13 and 16
took place in sodium ethoxide, ring opening of $\mathbf{y}$ -pyrone ring followed by ring closure took place giving rise to the thioxopyridinecarbonitrile 19 \[9, 10\](Scheme 4).
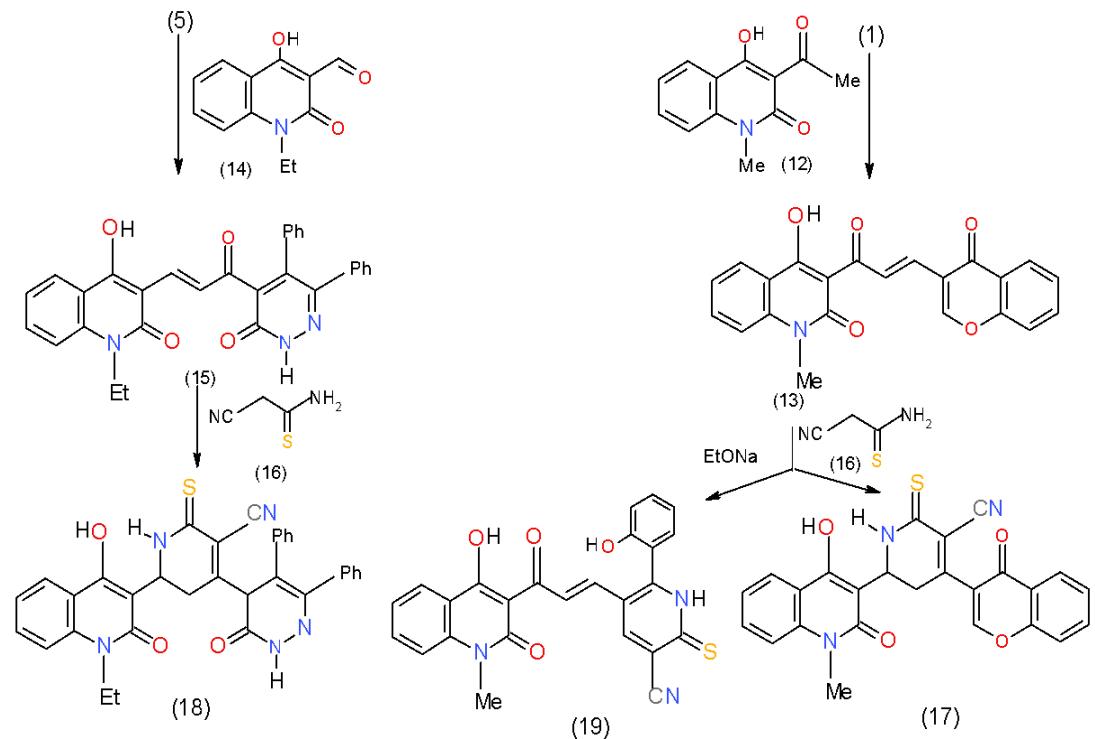 Scheme 4: Cyanothioacetamide in the formation of thioxopyridinecarbonitries
Moreover, acylation of 2-aminopyrimidinecarbonitrile (20) using acetic anhydride yielded 2-acetamidopyrimidinecarbonitrile (21), which underwent condensation with benzaldehyde in ethanolic sodium hydroxide giving 2-(N-cinnamoylamino) pyrimidine-carbonitrile (22). As reported for $\alpha$, $\beta$ -unsaturated ketones, the interaction of compound 22 with active methylene compounds afforded the substituted pyridine. Accordingly, when compound 22 was allowed to react with malononitrile (7) and ethyl cyanoacetate in the presence of ammonium acetate afforded 2-amino-6-pyrimidinylaminopyridinecarbonitrile (23) and 3-cyano-6-pyrimidinylamino-pyridone (24), respectively \[11\](Scheme 5).
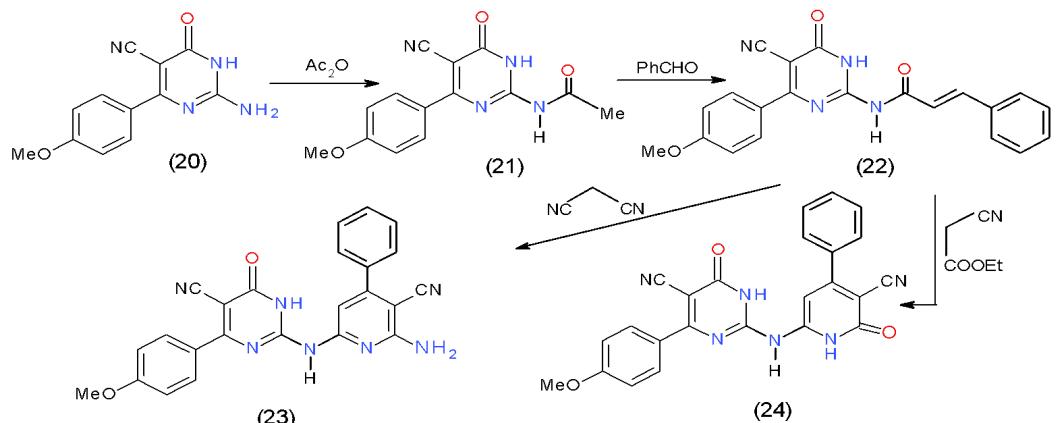 Scheme 5: Formation of pyrimidinylaminopyridines
### b) Using Acetylpyridazine
In addition to the use of acetylyridazine (5) in the formation of some enones, it could be converted into a new active methylene compound or enaminone and used them in the synthesis of pyridine. Accordingly, synthesized active methylene compound namely, pyridazinyl-butan-1,3-dione 27 obtained from the reaction of 4-acetylpyridazinone 5 with ethyl acetate (25) under clasein condensation [12]. Whereas, the enaminons 28 formed through condensation of 5 with dimethyl-formamide dimethylacetal, DMFDA, (26) in non-polar solvent [9]. Cyclocondensation of cyano
acetamide (10) with compound 27 in refluxing ethanol containing catalytic amount of triethyl amine afforded 4-pyridinylpyridazinone 29 [8]. Whereas, 2-pyridinyl 4- pyridazinyl ketone (30) synthesized on refluxing the enaminone 28 in acetic acid in the presence of ammonium acetate \[13\](Scheme 6).
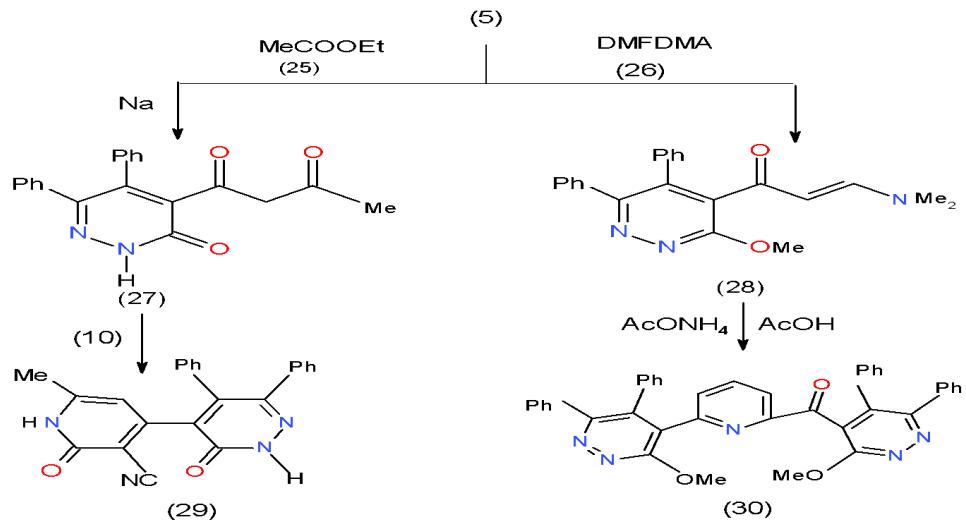 Scheme 6: Formation of pyridazinylpridines
### c) Using Arylidene Malononitriles
Condensation of malononitrile (7) with each of p-chlorobenzaldehyde (31), formyl derivative (4) and 3-formyl-6-methylchromone (32) afforded the respective arylidenemalononitriles 33a-c. In refluxing dimethyl formamide containing catalytic amount of piperidine cyanoacetic acid-hydrazide (35), the product of hydrazonolsis of ethyl cyanoacetate, (34) was added to arylidenemalononitriles 33a-c, and yielded the corresponding 1,6-diamino-4-aryl-2-oxo-1, 2-dihydropyridine-3,5-dicarbonitriles (36a-c) \[14,15\](Scheme 7). Compound 36 was considered as a suitable synthon for many fused pyridines.
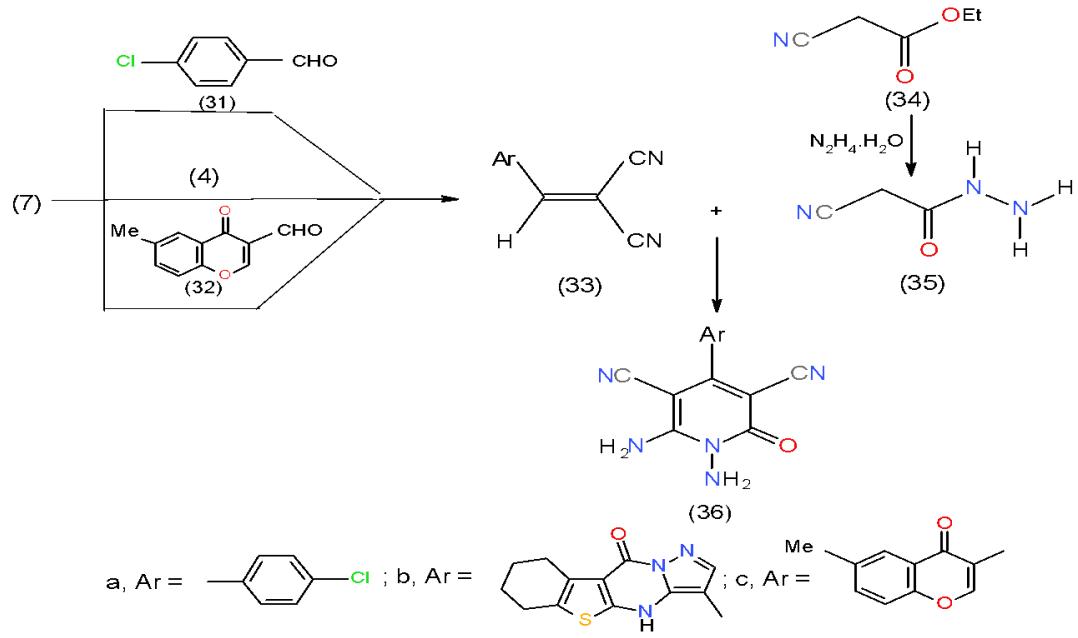 Scheme 7: Formation of diaminopridinedicarbonitrile
## III. FORMATION OF FUSED PYRIDINES
Incorporating pyridine with other heterocyclic systems such as thiazole, indole, pyrimidine, triazine and triazepines in one molecular frame-work enhance the biological activities of the produced condensed pyridine derivatives. Synthesis of fused pyridine could be carried out using some heterocyclic systems having active methylene such as thiazoldinones, 2-indolinone or heterocycles have active methine like 6-amino-1,3-dimethyluracil and with a heterocycle with a vicinal diamino groups as in compound 36.
### a) Synthesis of Thiazolopyridines
A number of methods to prepare thiazolopyridines have been documented in the literature. For example, methods that construct the bicycle by formation of a thiazole ring, include condensations of 3-amino-2-halopyridine, or 3-amino-2-pyridone derivatives, with thiocyanates, thioamides, or thioesters, condensations with 3-aminopyridin-2-thiones, and reactions of N-(2-pyridone-3-yl) acetamides with phosphorous pentasulfide, the oxidative ring-closure of 3-aminopyridine thioamides or thioureas, [16]. In our work, we annulated pyridineon thiazolidnone derivative using active methylene compounds. Thus, cyclocondensation of 5-arylidenethiazolidinone 36 with malononitrile (7) and ethyl cyanoacetate (34) in the presence of ammonium acetate yielded thiazolopyridine 37 and thiazolopyridone 38, respectively \[17\](Scheme 8).
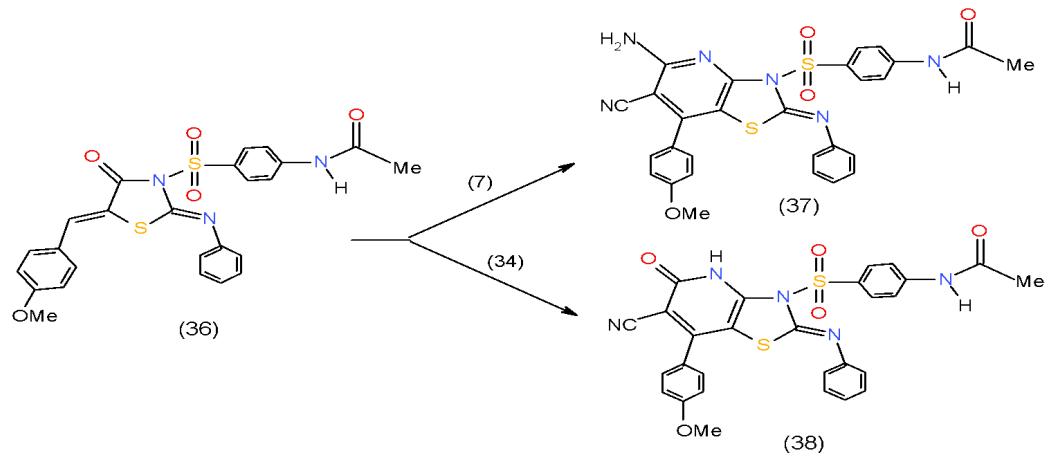 Scheme 8: Formation of thiazolopyrimidines
### b) Synthesis of Indolopyridines
In the last 30 years' hundreds of indoloquinoline analogues were synthesized and their biological activities evaluated as scaffolds for drug discovery. This fact aroused us to synthesize some bioactive indolopyridines starting with 2-indolinone (39) was studied. Thus, condensation of 2-indolinone (39) with DMFDA (26) afforded the respective enaminone (40), which on treating with $\mathrm{POCl}_3$ gave 2-chloroindole derivative 41. Cyclocondensation of 41 with two compounds having active methane site in their structures such as, enaminonitrile 42 and 6-aminoaminouracil 43, afforded the indolopyridine-carbonitrile 44 and indolopyridopyrimidinedione 45, respectively \[18\](Scheme 9).
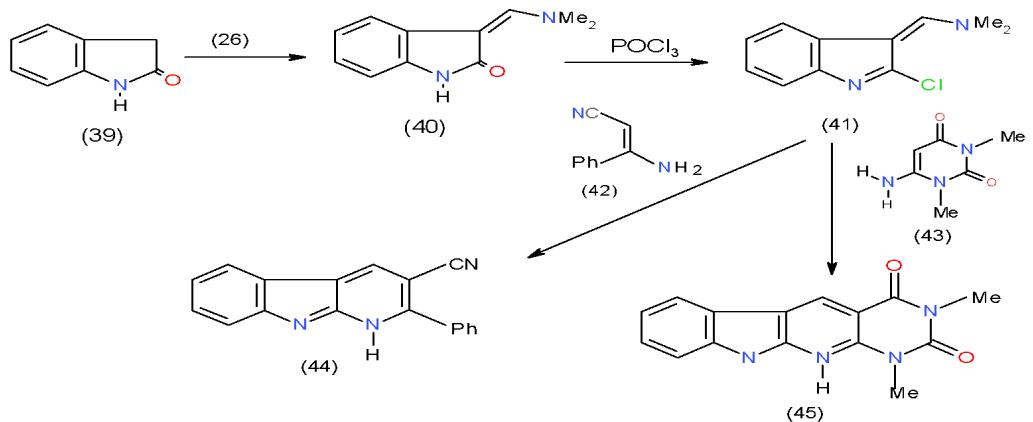 Scheme 9: Formation of indolopyridine derivatives On the other hand, treatment of 40 with 1-naphthylamine (46) yielded 3-N-naphthylamino derivative 47, which on boiling in poly phosphoric acid (PPA), dehydration took place giving rise to indolobenzoquinoline 48. Heating of indolinone (39) with enaminone 40 in acetic acid and ammonium acetate mixture furnished indolopyridoindole (49) \[18\](Scheme 10).
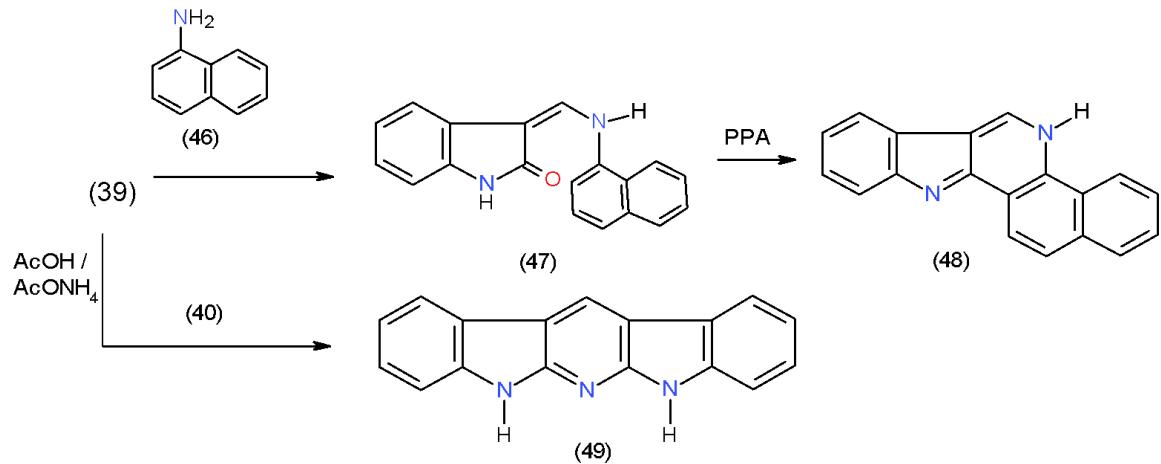 Scheme 10: Formation of poly cyclic system having pyridine
### c) Formation of Pyridopyrimidine Derivatives
Pyridopyrimidine derivatives are a privileged bicyclic ring system. Due to its potent and significant biological activities it has great pharmaceutical importance; synthesis of these compounds is considerable interest. The development of a practical method for the synthesis of various pyrido- pyrimidines, in view of their structure relation with peteridine, is of interest in the field of medicinal chemistry [19]. Most preparation of pyridopyrimidines concentrated on ring closure reactions of either pyridine or pyrimidine nucleus having appropriate substituents. Accordingly, chloropyridinecarbonitrile 50 reacted with some heterocycles having vicinal amino-cyano groups such as 2-aminopyridinecarbonitrile 51 and 3-aminopyrazolopyridinecarbonitrile 52 to afford the respective polycyclic systems 53 and 54 having pyridolopyrimidines in their structures [20](Scheme11).
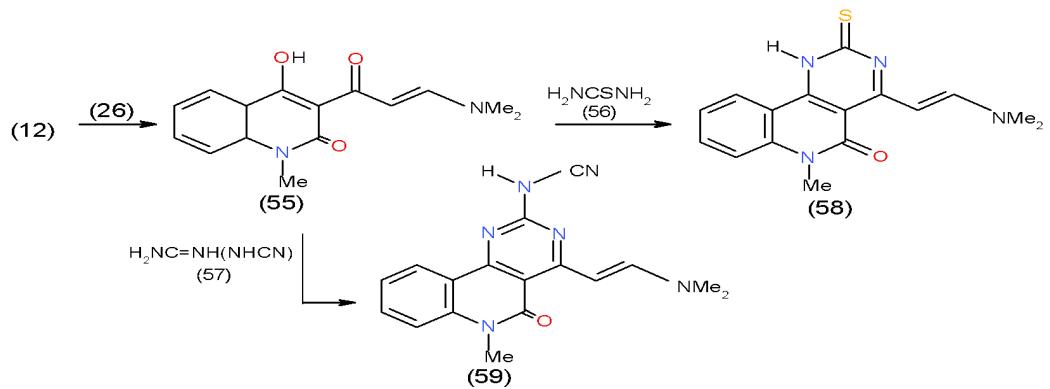
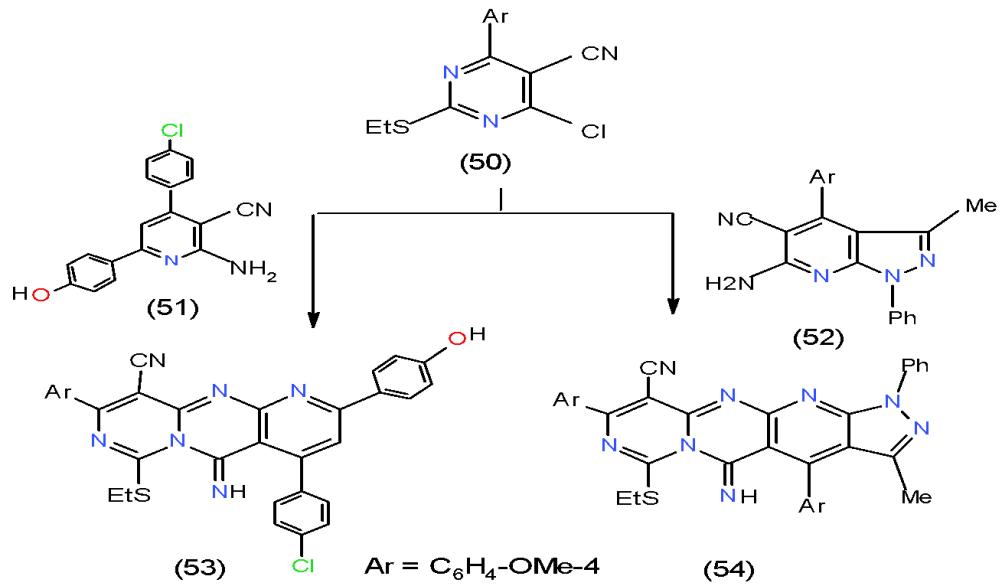 Scheme 11: Formation of pyridopyrimidopyrimidine derivatives On the other hand, Condensation of 3-acetyl quinolinone 12 with DMFDA (26) yielded the enaminone 55. The action of thiourea (56) and cyanoguanidine (57) upon the enaminone 55 gave rise to pyrimidoquinolinones 58 and 59, respectively \[21\](Scheme 12).
 Scheme 12: Formation of pyrimidoquinolinones
Nucleophilic substitution of the chlorine atom in chloropyrimidinecarbonitrile (60) using ethyl cyanoacetate (34) under basic condition afforded pyrimidinyl cyanoacetate derivative (61). Compound 61 is considered a good synthon for some interesting trinitrogenous heterocyclic systems having pyridine nucleus in their structures. Thus, cyclocondensation of 61 with acetami-dine hydrochloride (62a), guanidine hydrochloride (62b) and S-methylthiourea sulphate (62c) in sodium ethoxide solution furnished substituted pyrimidopyridopyrimidines 63a-c respectively. Similar behavior was observed when compound 61 also reacted with N-phenylthiourea (64) to afford pyrimidopyridopyrimidinethione 65. Moreover, treatment of 61 with aqueous ammonia at room temperature gave the corresponding amide 66, which reacted with formamide (67) in DMF to yield the substituted pyrimidopyridopyrimidines 68 \[22\](Scheme 13).
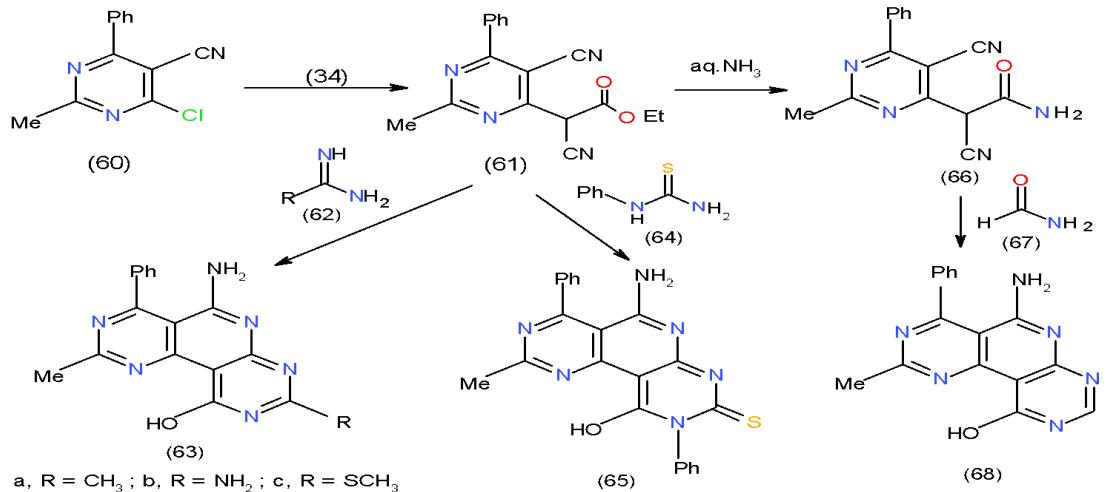 Scheme 13: Formation of pyrimidopyridopyrimidines
Recently, an analogous method for the synthesis of pyridopyrimidines was proposed via the reaction of 6-amino-2-thiouracil with activated olefinic system possessing a leaving group such as dimethyl-amino [23]. Motivated by this fact, we allowed ethyl cyanoacetate (34) to react with both 3,5-dimethylpyrazole (69) and tiethylorthoformate (70) to afford 3,5-dimethylpyrazol-1-yloxo) acetonitrile (71) and ethyl ethoxymethylenecyanoacetate (72), respectively. Treatment of 71 with DMFDA (26) yielded the enaminone 73, which reacted with 6-aminouracil 43 to give the pyridopyrimidine 75 via non-separable intermediate 74. Compound 75 was also obtained from the reaction of 6-aminouracil 43 with compound 72\[24\](Scheme 14).
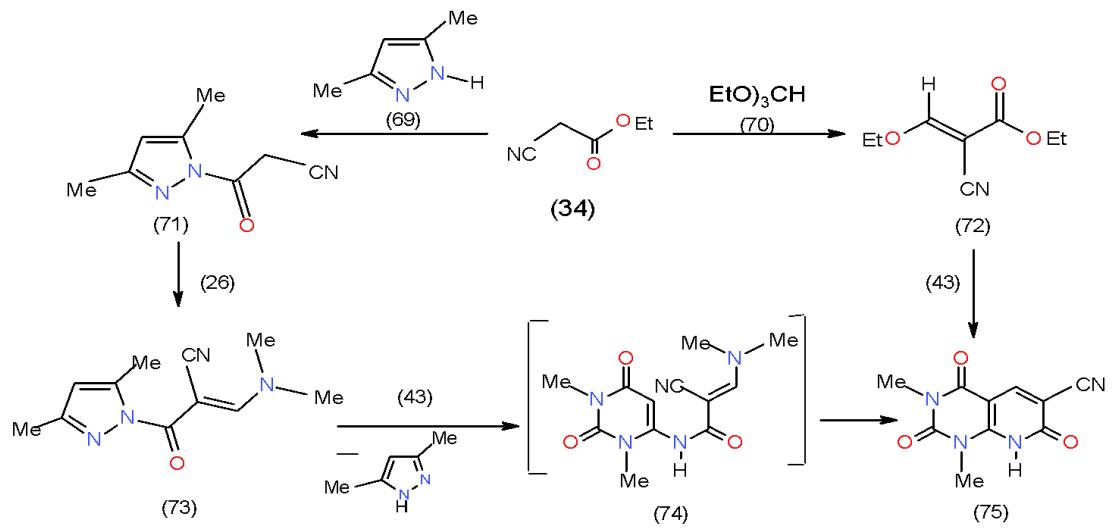 Scheme 14: Formation of pyrimidopyridines In conjunction to our interest in the chemistry of the enaminones, pyrazolopyrido- pyrimidindiones 77a, b synthesized on treatment of 6-aminouracil 43 with the cyclic enaminones 76a, b, while the pyrido
pyrimidinediones 80a, b and benzimidazolylpyridopyrimidinedione 81 obtained from the reaction of compound 43 with two enaminones 78a, b and enamine 79, respectively \[25\](Scheme 15).
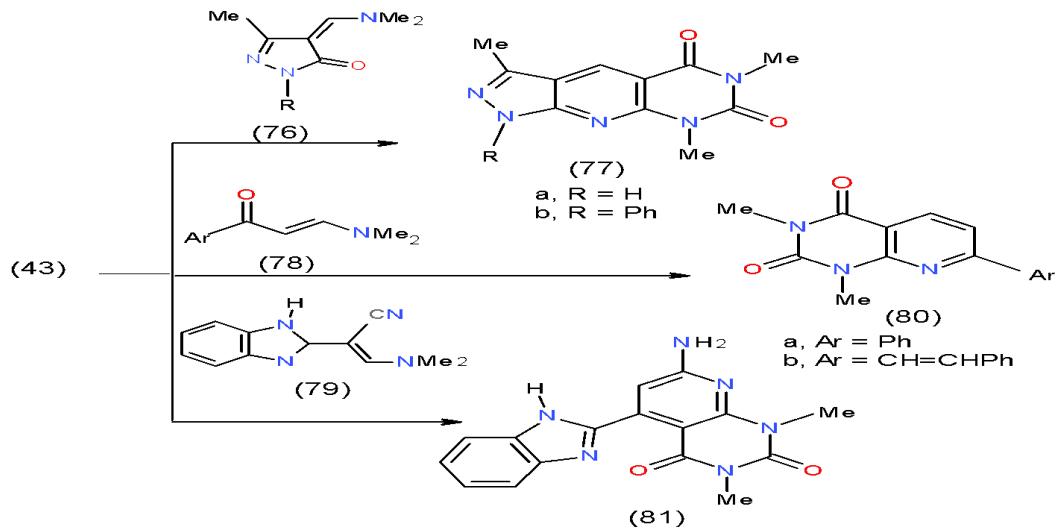 Scheme 15: Formation of pyridopyrimidinediones
Moreover, 6-amino-1,3-dimethyluracil (43) having an active methine group at position-5, reacted with some bifunctional heterocyclic systems having vicinal chloro-cyano groups in their structures such as 2-chloropyridinecarbonitrile 82, 3-chloropyridazinecarbonitrile 83 and 4-chloropyrimidinecarbonitrile 60 and afforded the novel triheterocyclic systems having pyridopyrimidines in their structures 84-86, respectively \[25\](Scheme 16).
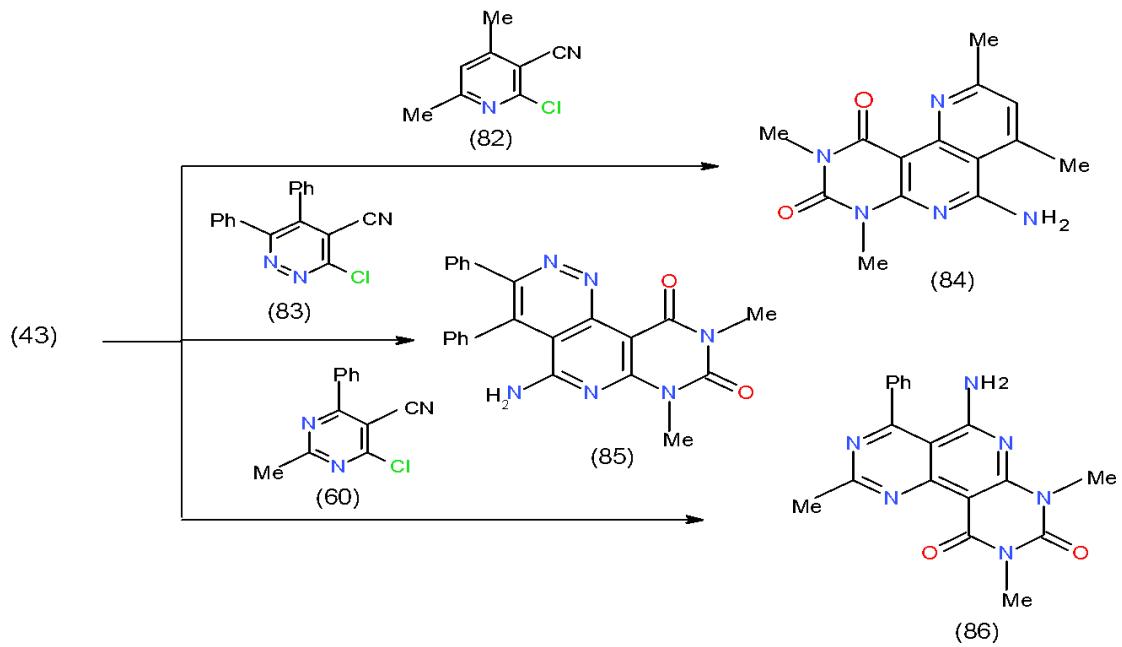 Scheme 16: Formation of pyridine in triheterocyclic systems
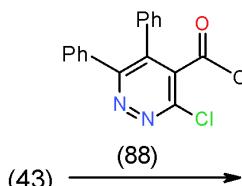
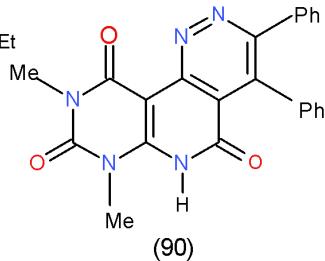
Furthermore, bifunctional heterocyclic system having vicinal chloro-acetyl groups (4-acetyl-3-chloropyridazine 87) or vicinal chloro-ethoxycarbonyl groups (4-carbethoxy-3-chloropyridazine 88) reacted with 6- amino-1,3-dimethyluracil (43) to yield the corresponding pyrimidopyrido- pyridazines 89 and 90 \[25\](Scheme 17).
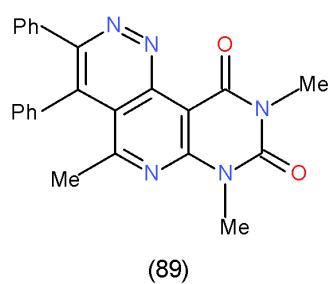
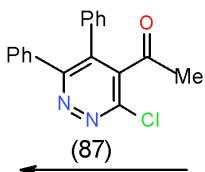
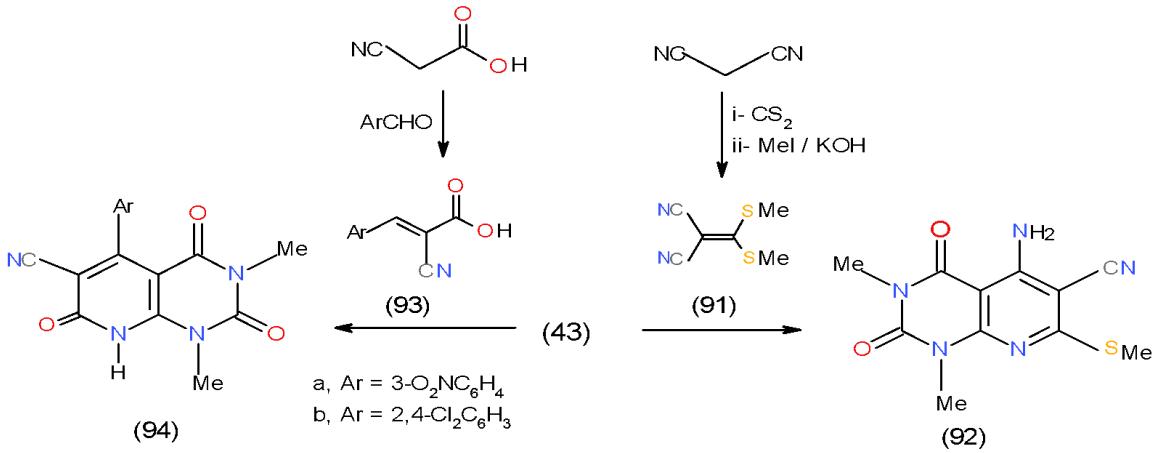
 Scheme 17: Formation of pyrimidopyridopyridazines On the other hand, the reaction of malononitrile (7) with carbon disulphide in basic medium followed by addition of methyl iodide afforded 2-cyano-3,3-bis (methylthio) acrylonitrile (91) [26], which reacted with 43 to yield pyridopyrimidinedione derivative 92 while, the
reaction of 43 with arylidenecyano acetic acids 93a, b, obtained from the condensation of cyanoacetic acid with aromatic aldehydes afforded pyrido- pyrimidine-triones 94a, b \[20\](Scheme 18).
 Scheme 18: Aminouracil in the formation of pyrimidopyridines
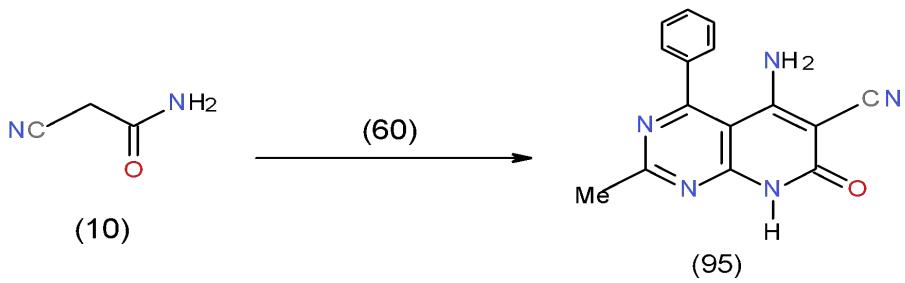
Also, the reaction of 4-chloropyrimidine-carbonitrile 60 with cyanoacetamide (10) in boiling dimethyl formamide gave aminopyridopyrimidinecarbonitrile 95 \[27\](Scheme 19).
 Scheme 19: Formation of aminopyridopyrimidinecarbonitrile
### d) Formation of Pyridotriazines Derivatives
The bicyclic heterocycles such as pyridotriazine and pyridopyridazine are useful for treating cell proliferative disorders, such as cancer, atherosclerosis, restenosis, angiogenesis, diabetic retinopathy, psoriasis, and endometriosis and immunological disorders [28]. Motivated by these facts we succeeded in the preparation of some pyridotriazines with the help of 1,6-diamino-4-aryl-2-oxo-1,2-dihydropyridine-3,5- dicarbonitriles (36a-c) as starting intermediate. Thus, the action of $\alpha$, $\beta$ - bifunctional electrophilic reagents such as chloroacetyl chloride, benzil, oxalyl chloride and sodium pyruvate upon compounds 36b, c was studied and yielded the corresponding nitrogen bridgehead pyrido 1,2,4-triazinones 96a, b- 99a, b, respectively, via cyclocondesation process \[14,15\](Scheme 20).
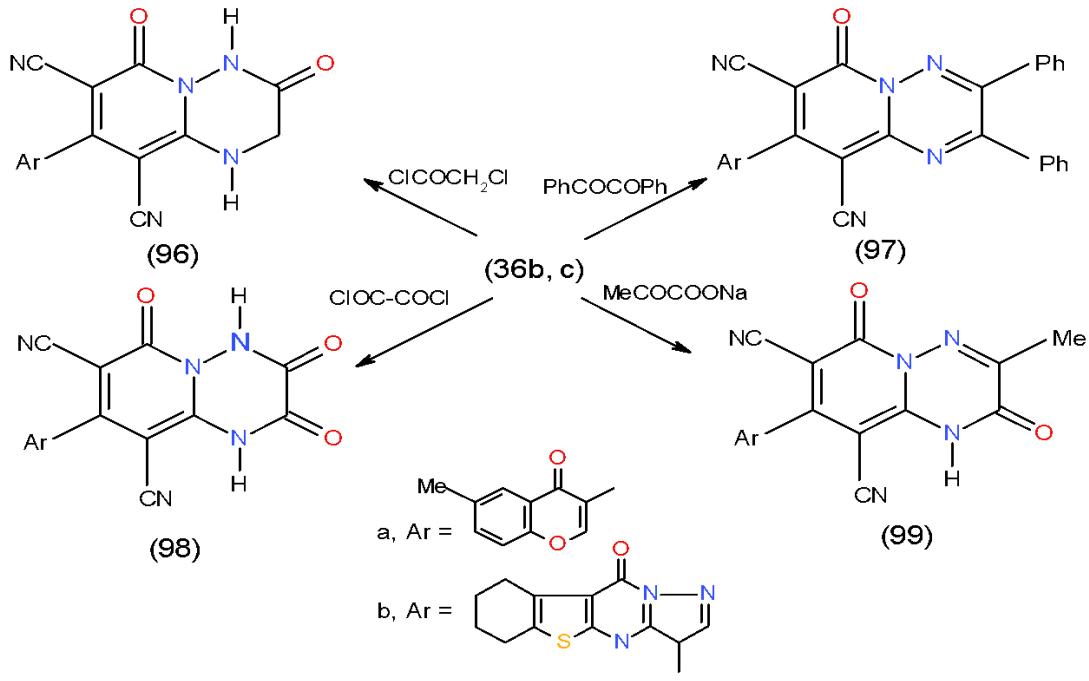 Scheme 20: Formation of pyrido1,2,4-triazinones
Moreover, the action of ethyl $\alpha$ -cyano- $\alpha$ -phenylazoacetate 101, obtained via the condensation of ethyl cyanoacetate (34) with benzenediazonium chloride 100 upon diaminopyridinedicarbo- dicabonitrile 36a afforded pyrido 1,2,4-triazinone 104 instead of aminopyrido 1,2,4-triazine 105. The formation of 104 could be explained via non-separable intermediate 102 not 103 \[11\](Scheme 21).
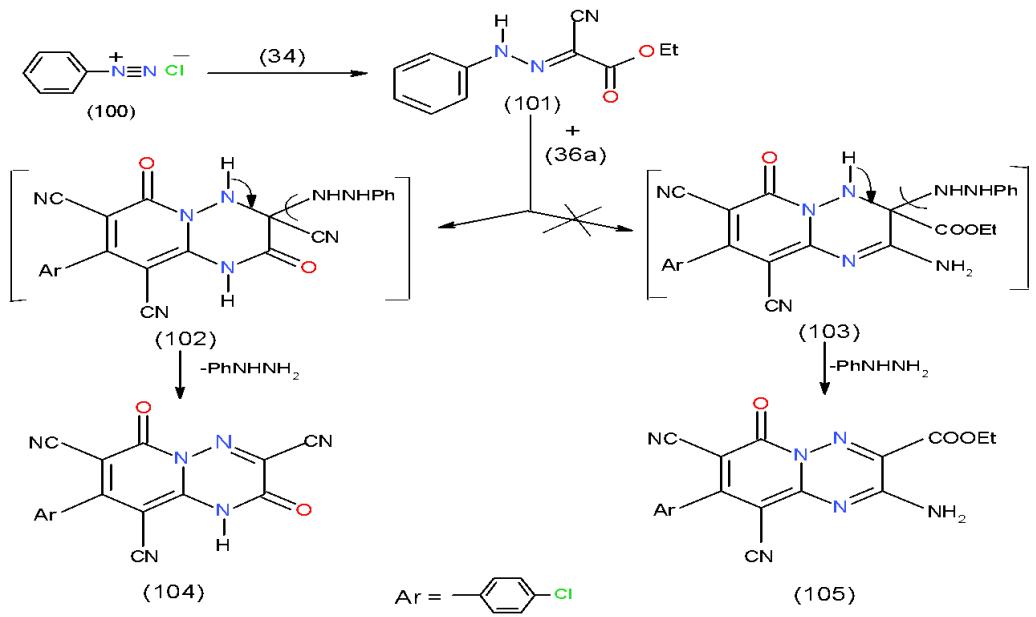 Scheme 21: Formation of pyrido1,2,4-triazinone
Also, condensation of 2,3-indoledione (isatin) (106) with diaminopyridonedicarbonitrile 36b, c in glacial acetic acid containing freshly fused sodium acetate afforded indolopyridotriazine derivative 107a, b.
Whereas, a different behavior was observed on treating 36b, c with N-acetylisatin 108 in glacial acetic acid as it furnished pyridotriazinone 110a, b. The formation of 110 took place via the intermediate 109 \[14\](Scheme 22).
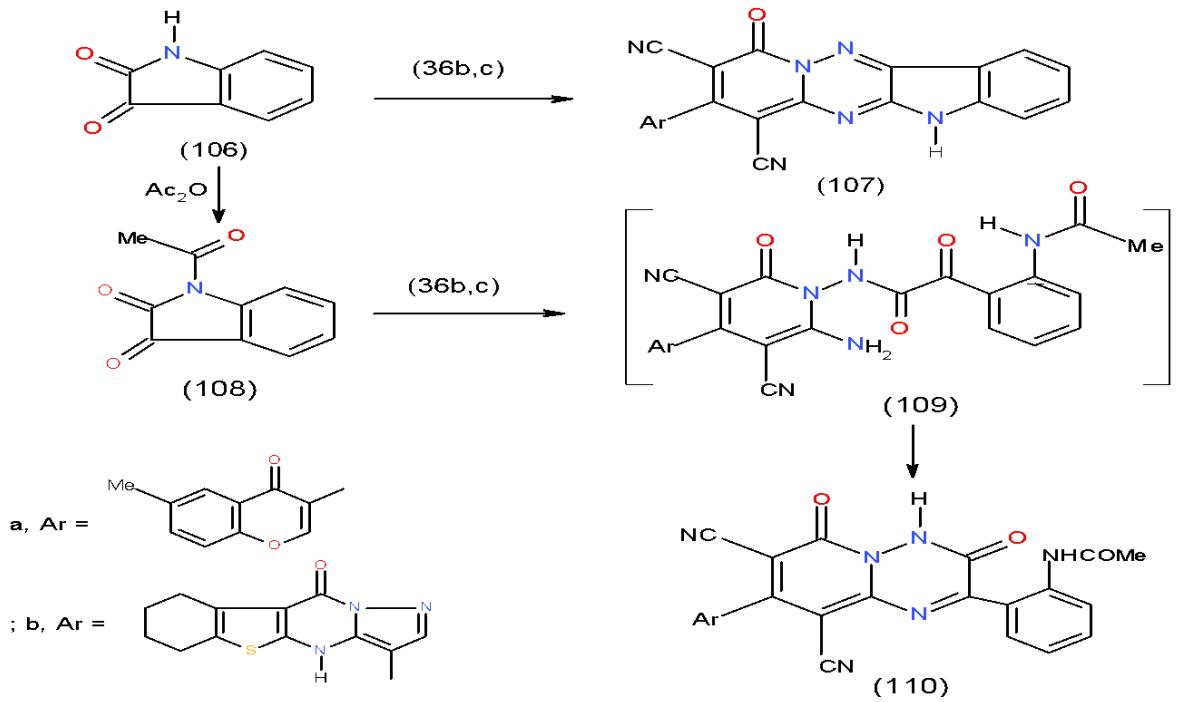 Scheme 22: Formation of pyrido1,2,4-triazinone derivatives On the other hand, for the preparation of pyridine bearing indotriazine we carried out the condensation of 2,6-diaminopyridine (111) with 2,3-indoledione (106) afforded 2,6-bis (indol-imino) pyridine (112). Alkylation of 112 with phenacyl bromide and chloro acetone gave 2,6-bis (1-substitutedindolimino) pyridine (113a, b), respectively. Hydrazinolysis of 113a, b with hydrazine hydrate yielded the respective 2,6-bis (1,2,4-triazinoindolimino) pyridine 114a, b, whereas,
alkylation of 112 with chloroacetamide and ethyl bromoacetate furnished 2,6-bis(1-substitutedindolimino) pyridine (113c, d), which on fusion with hydrazine hydrate produced 2,6-bis(1,2,4-triazinoindolimino) pyridinone 115 \[29\](Scheme 23).
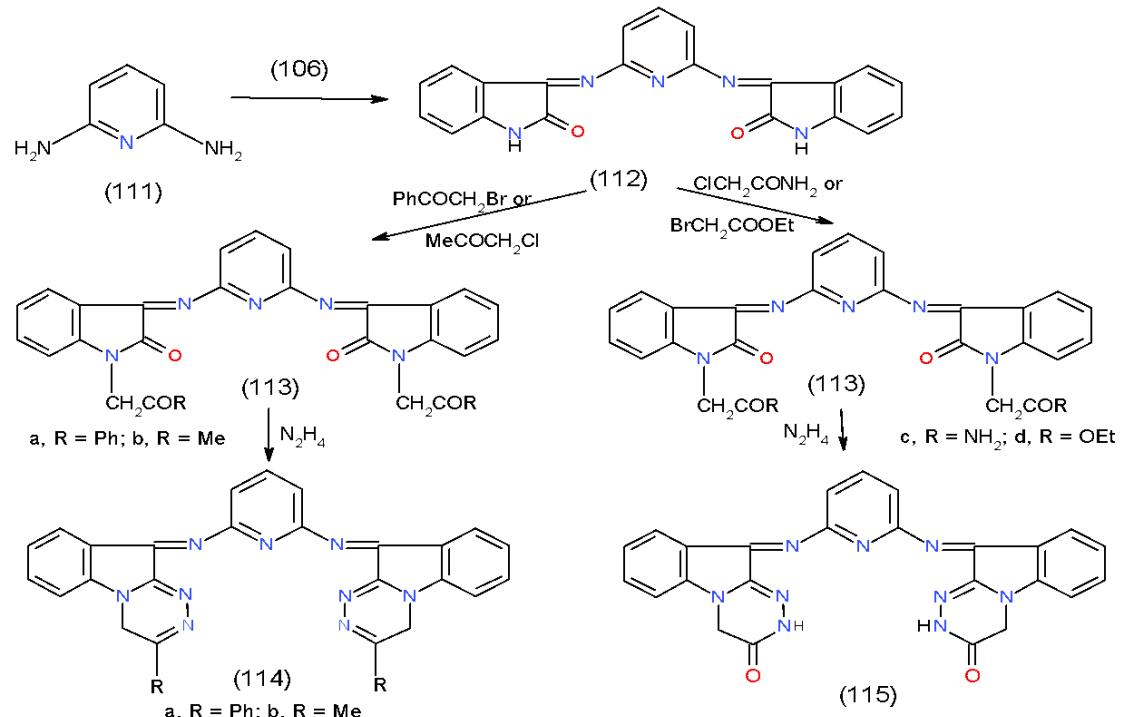 Scheme 23: Formation of bis indolo1,2,4-triazinone
Moreover, the formation of pyrazolopyridine in addition to pyridazinotriazine in one molecular frame was carried out by alkylation of compound 18 with ethyl chloroacetate to afford S- and N- alkylated product 116.
Treatment of 116 with hydrazine hydrate produced pyridazino1,2,4-triazinone 118 through the non-separable intermediate 117 [9]. (Scheme 24).
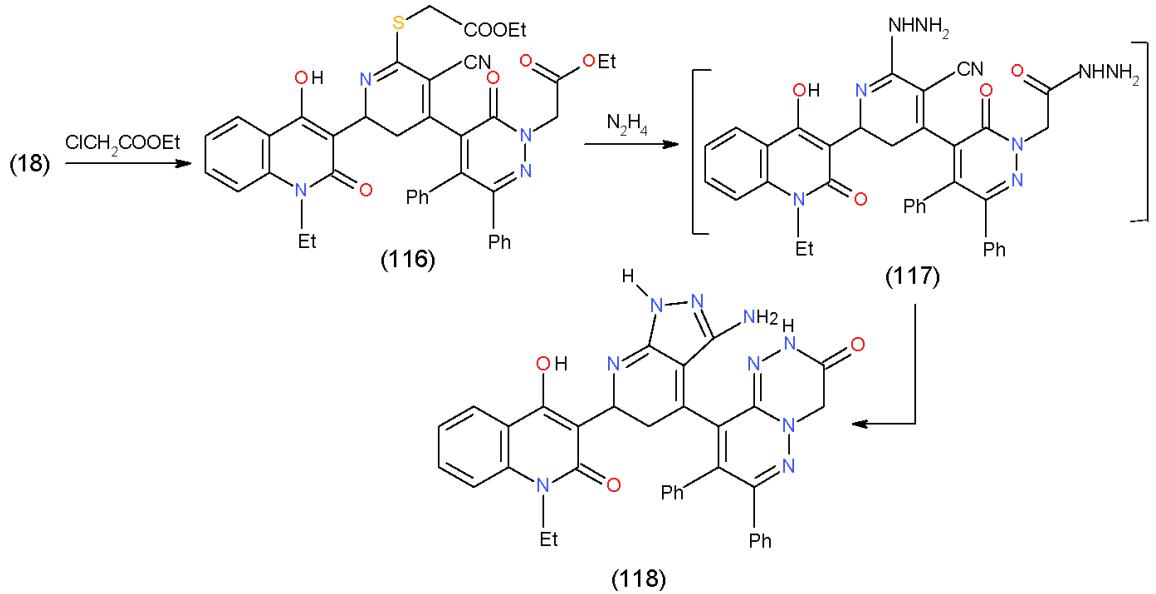 Scheme 24: Formation of and pyridzino1,2,4-triazinones
### e) Formation of Pyrido 1,2,4- Triazepines
The literature survey of 1,2,4-triazepines, synthesis and reactions of monocyclic and fused heterocycles incorporating 1,2,4-triazepines as well as their biological evaluation and synthetic applications was described [30]. As a part of our program directed for the synthesis of new polynuclear bioactive heterocyclic systems, one of our synthetic strategy is designed to utilize of 1,6-diamino-4-aryl-2-oxopyridine-3,5-dicarbonitrile 36a-c as a suitable synthon for the synthesis of nitrogen bridge-head pyrido[1,2,4]-triazepines. Accordingly, treatment of 36a with diethyl malonate (119), diazotized acetylacetone (120), dimethyl acetylenedicarboxylate (121) and dehydroacetic acid (122) furnished substituted pyrido1,2,4-triazepines 123-126, respectively \[31\](Scheme 25).
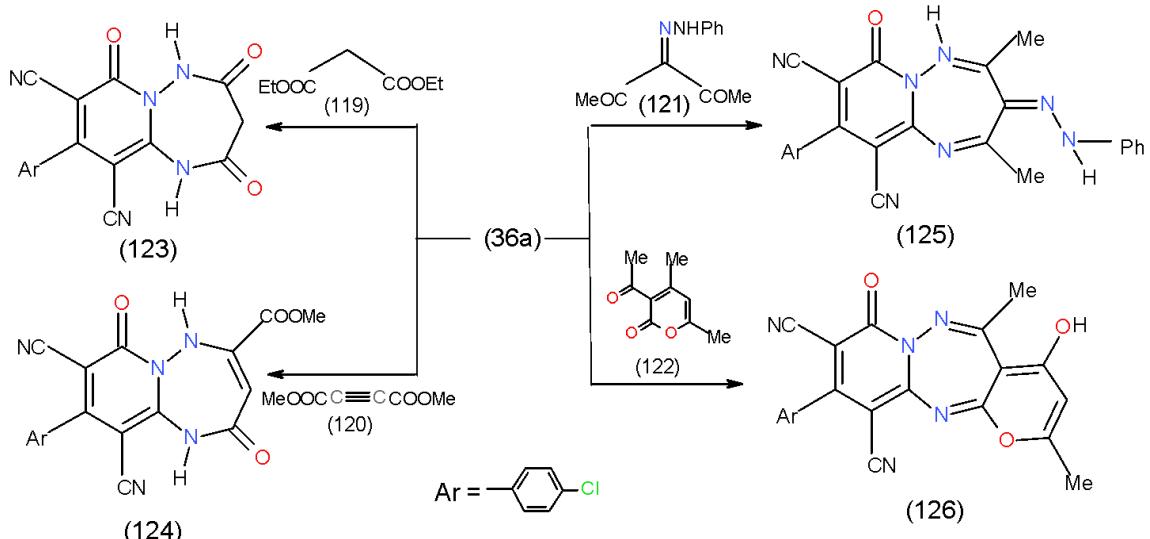 Scheme 25: Formation of pyridotriazepine derivatives
Moreover, the action of 2-cyano-3,3-bis (methylthio)acrylonitrile (91) and chromone-3-carbonitrile 127 upon 36a-c yielded pyrido1,2,4-triazepines 128a-c and 129a-c, respectively. Also, Treatment of 36a with ethyl exothymethylenecyanoacetate (72) gave pyrido1,2,4-triazepine- tricarbonitriles 130 while, the reaction of 36b, c with arylidene cyanoacetate (131) produced aminopyrido1,2,4-triazepinedicarbonitriles 132a, b, respectively \[14-31\](Scheme 26).
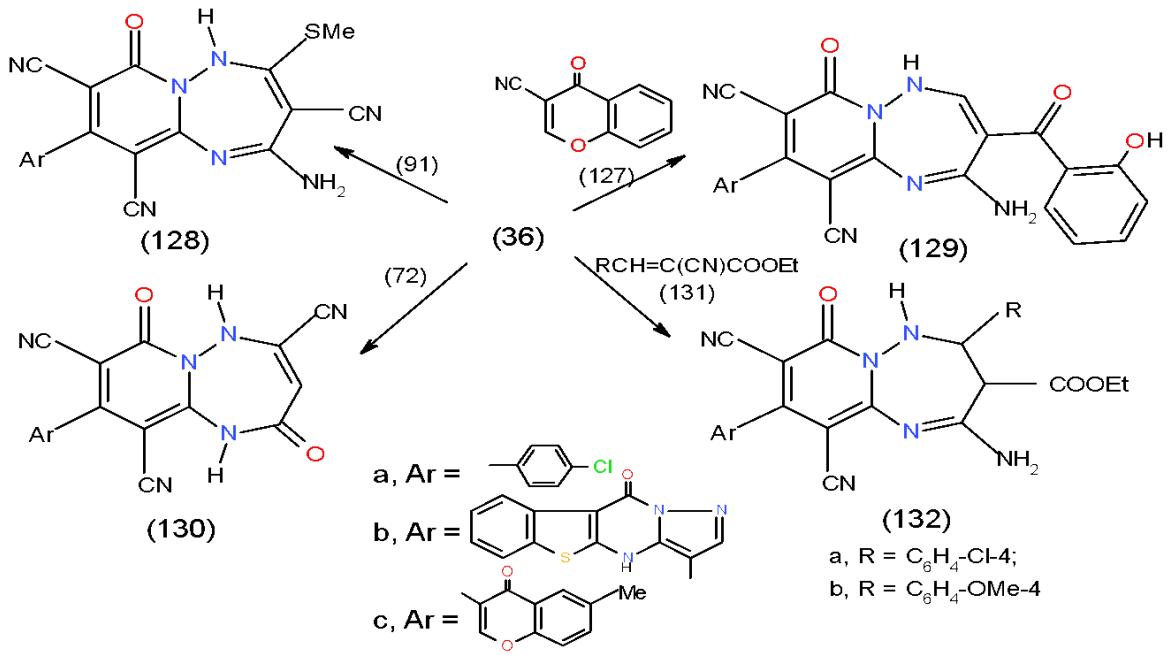 Scheme 26: Formation of substituted pyridotriazepine
Furthermore, cyclocondensation of 36b, c with arylided malonitrile (133), 2-cyano-3,3-bis(methyl thio) prop-2-enamide (134) and o-chloroaldehyde derivative 135 gave the corresponding pyrido- 1,2,4-triazepines 136a, b, 137a, b and 138a, b, respectively \[14,15\](Scheme 27).
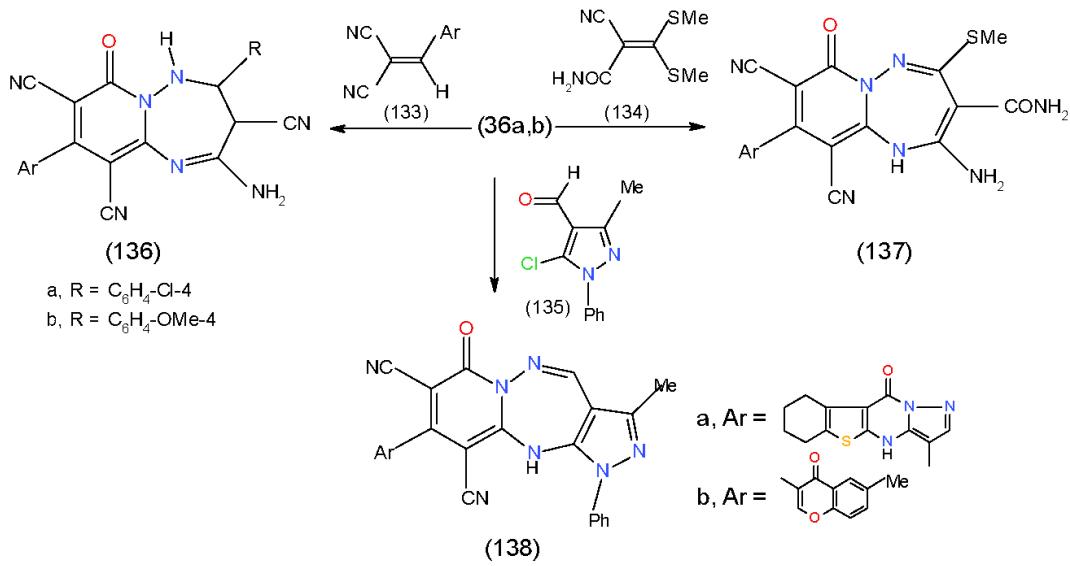 Scheme 27: Formation of substituted and condensed pyridotriazepines On the other hand, the action of 3-formylchromone (1) upon 36a and the effect of 2-chloro-3-formylquinoline (139) on 36c was studied and
furnished the respective pyrido1,2,4-triazepines 140 and 141 \[14,31\](Scheme 28).
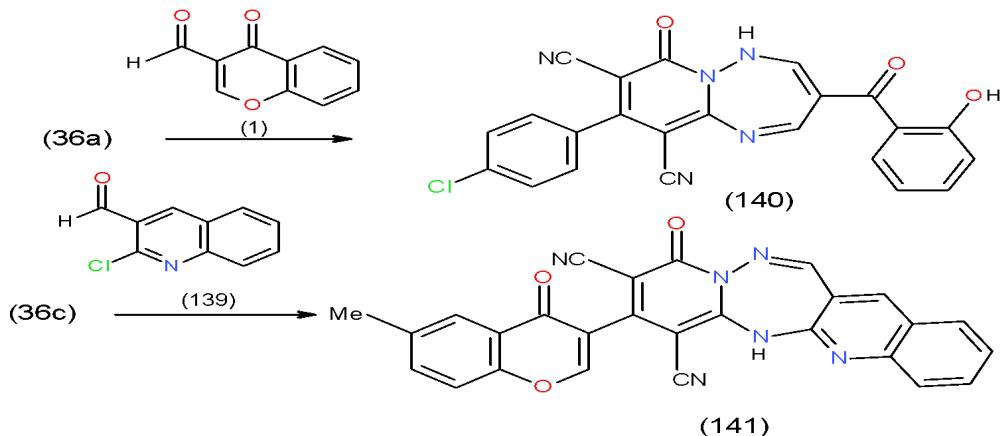 Scheme 28: Formation of pyridoquinolinotriazepine
## IV. BIOLOGICAL IMPORTANCE
### a) Antimicrobial Activities
Some synthesized heterocyclic compounds were tested their antimicrobial activity against some Gram-positive bacteria such as Bacillus subtilis, Staphylococcus aureus and Bacillus cereus and some Gram-negative bacteria namely, Escherichia coli, Salmonella typhimurium, Pseudomonas aeruginosa and Proteus vulgaris and some fungi for examples Candida albicans, Aspergillus fumigatus using standardized disc agar diffusion method [32] or Vincent filer paper disc method [33] with taken some antibiotics as reference. Herein we recorded the compounds exhibited strong inhibition effect against a certain microorganism.
## i. Action on Gram-positive bacteria
# a. Bacillus
A Gram-positive Bacterium Bacillus subtilis, like many microorganisms, can form most of the enzymes needed for the biosynthesis of the amino acid tryptophan. The primary role of tryptophan within living organisms is as a novel residue within many proteins. The efficient production of secreted enzymes B. subtilis is considered a key drivers of the successes in the enzyme industry [34]. The selected synthesized heterocyclic compounds were tested against Bacillus subtilis. The results showed that pyrido1,2,4-triazepinetricarbonitriles 130b, which carrying pyrazolobenzothienopyrimidinyl nucleus [15] exhibited the highest antimicrobial activity against Bacillus subtilis compared with the tested compounds in the study. (Bio-1 scheme).
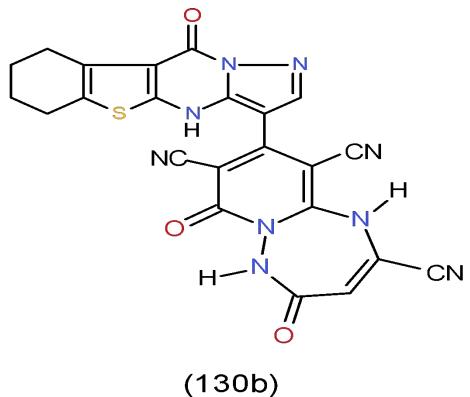 Bio-1 scheme: The highest antimicrobial activity one against Bacillus subtilis
### b. Staphylococcus areus
It is a Gram-positive, omnipresent bacterial pathogen that have the ability to adapt and live in various states. S. aureus is one of the major causes of spreading of the clinical infection such as bacteraemia and infective endocarditis, osteoarticular, skin and soft tissue, pleuropulmonary, and device-related infections [35]. The results showed that pyrazolopyridotroazepine 138a, which having pyrazolobenzothienopyrimidinyl moiety exhibited the highest antimicrobial activity against Staphylococcus areus [15]. Also, the pyridine carrying thiazolindione nuclei exhibited higher inhibition effect to Staphylococcus areus as in oxopyridine-carbonitrile 227 \[30\](Bio-2 scheme).
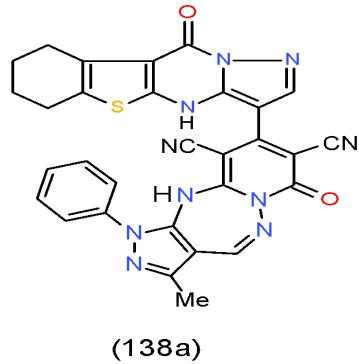 Bio-2 scheme: Compounds could use as antimicrobial agents against Staphylococcus aureus
## ii. Action on Gram-Negative Bacteria
# a. Escherichia
Escherichia coli is a large and various group of bacteria that is found naturally in the intestines of healthy humans and animals. Most types of Escherichia coli are harmless or cause relatively brief diarrhea, but some of the Escherichia coli can cause a disease for people which can be done by creating a toxin known as Shiga Toxin [36]. It can be seen clearly that pyridotriazepine 128b, which having pyrazolobenzothienopyrimidinyl moiety exhibited the highest antimicrobial activity against Escherichia coli compared with the tested 77 investigated compounds [8]. (Bio-3 scheme).
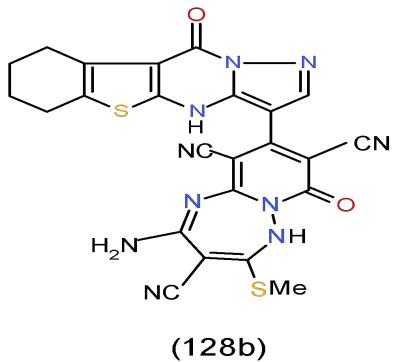 Bio-3 scheme: The highest antimicrobial activity compound against Escherichia coli
### b. Salmonella typhimurium
Infection of humans by the enteric pathogen Salmonella typhimurium generally results in severe abdominal cramping and diarrhea. These symptoms may largely result from the mucosal immune response elicited by this pathogen. Specifically, colonization of the human intestine by S. typhimurium leads to infiltration of polymorphonuclear leukocytes (PMNs) into the intestinal epithelium culminating in the formation of an intestinal crypt abscess [37]. Results obtained disclosed that pyrido1,2,4-triazinedione 110b [15], which having pyrazolobenzothieno- pyrimidinyl moiety exhibited the highest antimicrobial activity against Salmonella typhimurium compared with the tested compounds \[15\](Bio-4 scheme).
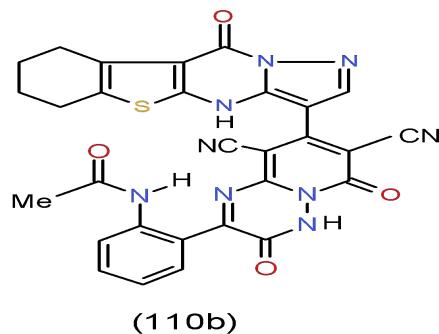 Bio-4 scheme: One of the highest antimicrobial activity against Salmonella typhimurium
## iii. Action on Fungi
# a. Candida
Candida albicans is one of the very few fungal species causing disease in humans—millions of others do not. It is a member of the healthy microbiota, asymptotically colonizing the gastrointestinal (GI) tract, reproductive tract, oral cavity, and skin of most humans [38]. The results showed that pyrido1,2,4-triazinedione 96b and aminopyrido1,2,4-triazepine 129b that having pyrazolobenzothienopyrimidinyl moiety in their structures exhibited the highest antimicrobial activity against Candida albicans compared with the tested compounds \[15\](Bio-5 Scheme).
 (96b)
 (129b) Bio-5 scheme: Compounds with the highest antimicrobial activity against Candida albicans
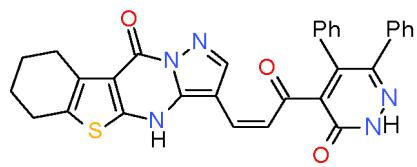
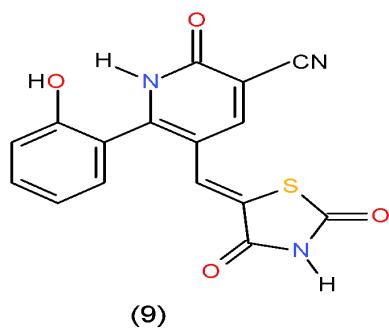
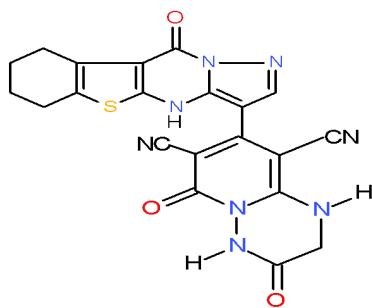
Also, the compound carrying thiazolidinedione nucleus in their structures such as pyridinecarbonitrile 9 and pyrido1,2,4-triazepine 142, obtained from reaction of the enone 3 with diaminopyridinedicarbonitrile 36a, may be used as antimicrobial agent against Candida albicans from tested compounds \[36\](Bio-6 scheme).
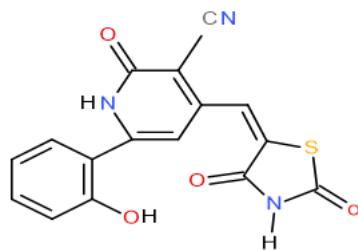 (9)
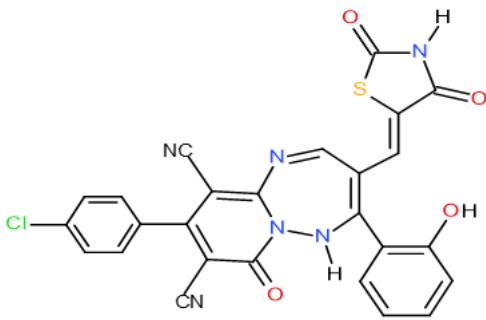 (142) Bio-6 scheme: Compounds may be used as antimicrobial agent against Candida albicans
### b. Aspergillus fumigatus
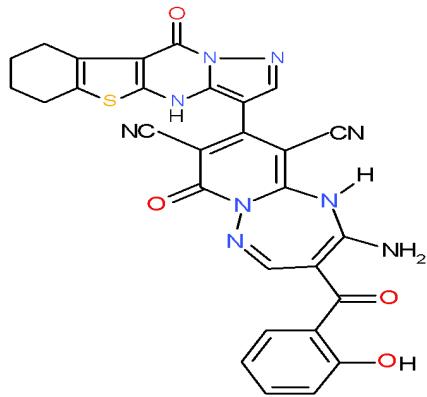
Aspergillus fumigatus is an opportunistic fungus causing allergic and invasive aspergillosis in humans and animals. It secretes an array of complex biologically active glycoprotein antigens and allergens, that have been implicated in human respiratory allergic disorders [39]. The compounds carrying pyrazolobenzothienopyrimidinyl moiety in their structures such as pyrido 1,2,4-triazepine 136a and pyrazolopyrido1,2,4-triazepine 138a may be used as antimicrobial agent against Aspergillus fumigates as they shown the highest inhibition zones \[15\](Bio-7 scheme).
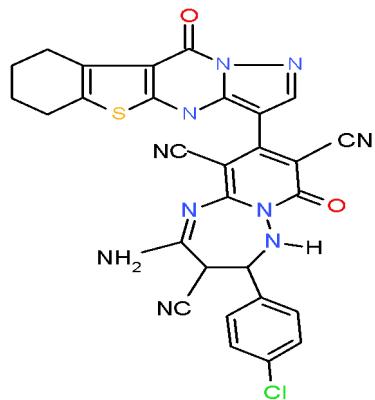 (136a)
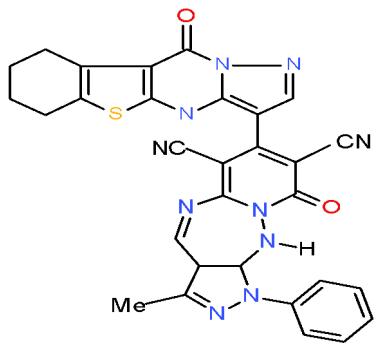 (138a) Bio-7 scheme: Compounds with the highest antimicrobial activity against Aspergillus fumigatus
## V. CONCLUSION
Pyridine derivatives are very important chemicals with tremendous biological application. In this study effort to optimize the synthetic procedures for the preparation of various bioactive heterocycles. Herein, we explain how active acyclic and cyclic methylene compounds as well as heterocyclic having active methyl or methine sites were used to synthesize wide varieties of pyridines and its fused systems by choosing certain starting materials and different synthesizing reagents. This study will help researchers in the fields of organic and medicinal chemistry to design and implement new procedures for the constructions of novel biological components.
Generating HTML Viewer...
References
39 Cites in Article
Darrin Hopper,Kristina Kutterer,Aimee Crombie,Jeremy Clemens (2009). Chapter 6.1: Six-membered ring systems: pyridine and benzo derivatives.
Mohamed Abass (2005). Fused Quinolines. Recent Synthetic Approaches to Azoloquinolines. A Review.
Mohamed Abdel-Megid (2021). Part–I: Utilities of active methylene compounds and heterocycles bearing active methyl or having an active methine in the formation of bioactive heteroarylpyrimidines and pyrimidopyrimidines.
Mohamed Abdel-Megid (2020). Part—II: Utilities of active methylene compounds and heterocycles bearing active methyl or having an active methine in the formation of bioactive pyrazoles and pyrazolopyrimidines.
Mohamed Abdel-Megid (2020). Unknown Title.
Mohamed Abdel-Megid (2021). Part –IV: Utilities of active methylene compounds and heterocycles bearing active methyl or having an active methine in the synthesis of triazoles, pyridazines, triazines and diazepines.
A Magdy,M Ibrahim,Abdel-Megid,M Naser,El-Gohary (2011). Unknown Title.
M Kamelia,El-Mahdy,M Azza,M El-Kazak,M Abdel-Megid,O Seada,Farouk (2013). Unknown Title.
M Abdel-Megid,M Ismail (2002). Synthesis of some new nitrogen bridge-head pyrido[1,2,4]triazepines.
Mohamed Abass,Mohamed Abdel‐megid,Mohamed Hassan (2007). Substituted Quinolinones, Part 12: Heterocyclization Reactions of 3‐(3‐Chromonyl)acryloylquinolinone with Some Bifunctional Nucleophiles.
M Abdel-Megid,M Awas,M Seada,K El-Mahdy,M El-Sayed (2010). Unknown Title.
M Abdel-Megid,Y Gabr,M Awas,N Abdel-Fatah (2009). Synthesis of some new hetarylpyranopyridazines, cinnolines, and hetarylpyridazine derivatives.
M Abdel-Megid (2007). A New Route for the Synthesis of Substituted Pyridazines.
M Abdel-Megid,M Ibrahim,Y Gabr,N El-Gohary,E Mohamed (2013). Synthesis of Some New Nitrogen Bridge-head Triazolopyridines, Pyridotriazines, and Pyridotriazepines Incorporating 6-Methylchromone Moiety.
M Abdel-Megid,M Azza,El-Kazak,M Kamelia,M El-Mahdy,Osama Seada,Farouk (2017). Unknown Title.
Kamelia El-Mahdy,Azza El-Kazak,Mohamed Abdel-Megid,Magdy Seada,Osama Farouk (2016). of Some New Heterocyclic Schiff Bases Derived from Thiocarbohydrazide.
Mohamed Abdel‐megid,Mohamed Abass,Mohamed Hassan (2007). Substituted quinolinones. Part 13 a convenient route to heterocyclization reactions with 3‐substituted 4‐hydroxyquinolin‐2(1<i>H</i>)‐one.
M Abdel-Megid (1995). Synthesis of some new nitrogen bridge-head pyrido[1,2,4]triazepines.
Jairo Quiroga,Braulio Insuasty,Henry Insuasty,Rodrigo Abonia,Antonio Ortíz,Adolfo Sánchez,Manuel Nogueras (2001). Reactions of 6‐aminopyrimidines with 2‐dimethylaminomethylenetetralone. Regiospecific Synthesis Of 5,6‐Dihydrobenzo [<i>h</i>]pyrimido [4,5‐<i>b</i>] quinolines.
M Seada,M Fawzy,H Jahine,M Abd El‐megid,R Saad (1989). Synthesis and Biological Activities of Some New Pyridazine Derivatives.
M Abdel-Megid (2010). A convenient route for the synthesis of some new bi- and triheterocondensed uracils.
M Seada,S Ibrahim,A Abdel-Halim,M Abdel-Megid (1992). Unknown Title.
M Seada,A Abdel-Halim,S Ibrahim,M Abdel-Megid (1992). Unknown Title.
James Kramer,Howard Hollis (2002). Potential Changes in US Patent Laws: The Publication of Patent Applications.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Mohamed Abdel-Megid. 2026. \u201cPart –III: Utilities of Active Methylene Compounds and Heterocycles Bearing Active Methyl or having an Active Methine in the Formation of Substituted and Fused Pyridines\u201d. Global Journal of Science Frontier Research - B: Chemistry GJSFR-B Volume 22 (GJSFR Volume 22 Issue B1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Part –III: Utilities of Active Methylene Compounds and Heterocycles Bearing Active Methyl or having an Active Methine in the Formation of Substituted and Fused Pyridines