There has been an abundance of evidence from clinical, animal, and in vitro studies that progesterone (P4) is important for establishing, maintaining, and terminating a pregnancy. P4 exerts its primary action via its two receptors: progesterone receptor A (PGR-A) and B (PGR-B). Analyses of transcriptome and cistrome genome have unearthed novel members and modifiers of the P4 signaling pathway. The increase in serum P4 levels and down-regulation of PGR-B are important in the development of pinopodes, thus marking implantation. Additionally, it promotes the quiescent myometrial cell phenotype, and the inhibition of its production in myometrial cells induces labor and is the key physiologic initiator of parturition. Through genomic and non-genomic intracellular mechanisms involving these PGR isoforms, various physiologic states can be determined such as the quiescent and contractile myometrium during pregnancy.
### 20α-HSD: 20α-hydroxysteroid dehydrogenase
## I. INTRODUCTION
The pregnant state has baffled scientists since the beginning of time. The process of implantation, all the way through parturition has been the subject of extensive studies, as complex pathways at the neuroendocrine and immunological levels interact to establish and sustain a complex fetus. Alongside the hormonal changes that typically occur upon implantation and parturition, gene expression profiling studies have attempted to identify the signals that trigger these occurrences to provide insight into causative pathophysiological conditions and suggest treatment modalities to aid conception and prevent preterm births.
The common denominator between the above two states of pregnancy appears to be progesterone (P4). P4, an essential hormone in the regulation of female reproduction, acts primarily in the uterus: in particular, through preparing the endometrium for implantation after the ovulation and fertilization of an oocyte, and the maintenance of an ensuing pregnancy by promoting uterine growth and suppressing myometrial contractility [1, 2]. The ovary is the major site of synthesis and secretion of progesterone in the mammal, which in turn induces cyclical fluctuations in the levels of P4 in circulation [1].
The effects of P4 are facilitated by the nuclear progesterone receptor (PGR), which operates through nuclear transcription regulators that are ligand-activated [1, 2]. They are similar to other receptors for steroids, thyroid hormones, retinoids, and vitamin D3. The human PGR, a member of the NR3C3 subfamily of the nuclear receptor super family, is comprised of two isoforms: the full-length PGR-B and the truncated (by 164 N terminal amino acids) PGR-A [3]. The two receptor proteins are encoded by a single gene [located on human chromosome 11 (11q22-q23)] under the control of different promoters, each of which gives rise to a diverse subgroup of PGR messenger ribonucleic acid (mRNA) species [4].
The function of both receptors, similar to other steroid hormone receptors, is mainly via ligand-activated transcription factors. It appears that when the PGR-A level exceeds that of PGR-B, the latter will get suppressed. It is well known that PGR-B dominates throughout pregnancy and that P4 promotes myometrial relaxation via PGR-B-mediated genomic actions in myometrial cells to ensure myometrial quiescence. However, this is inhibited near term and during labor as myometrial cell expression of PGR-A increases leading to a PGR-A: PGR-B ratio of 3 or more. This leads to the conclusion that PGR-A is an endogenous repressor of PGR-B, and that genomic P4 responsiveness is inversely related to the PGR-A: PGR-B ratio [1, 3]. This differential expression of the progesterone receptor isoforms, PGR-A and PGR-B, contributes to a functional withdrawal of P4 and the onset of labor.
Furthermore, the role of a repressor that PGR-A plays appears to extend beyond its actions on PGR-B. It has been shown that PGR-A reduces the response of additional hormone receptors such as androgen, mineralocorticoid, glucocorticoid, and estrogen receptors to their appropriate ligands [5]. In the human myometrium, transcriptomic data reaffirmed that overexpression of PGR-B resulted in relaxed myometrium not responding to oxytocin stimulation, while the overexpression of myometrial PGR-A demonstrated a twofold increase in contractility in response to uterotonic agents [6].
The expression of PGR has also been described in other tissues which have been known to be responsive to P4, for example, the uterus, the ovary, and preovulatory granulosa cells [1]. Moreover, the expression of the P4 receptors, and therefore its sensitivity to progestins, seems to be controlled by estrogen, which increases the expression of PGR in most target tissues as opposed to P4, which decreases its expression [1].
Recent advances in the understanding of the process of parturition in humans have revealed a monumental finding. In 2003, Girotti and Zingg reported that concerning the expression of certain genes, the parturition phase represents a mirror image of the implantation phase [7]. In this narrative review, we hypothesize that the quiescent phase of pregnancy is a mirror image of parturition, whereas the implantation phase is not quite the opposite of it. While the common denominator between all phases is P4 and its receptor, there are many known and unknown mechanisms and pathways that operate differently in these phases.
## II. ROLE OF PROGESTERONE AND ITS RECEPTOR IN IMPLANTATION
A fruitful implantation of the embryo in the uterus necessitates an apt embryo, an amenable uterus, and complex harmonization between the two. For most mammals, the receptiveness of the uterus is time-sensitive; a window of receptivity lasting around 24 hours in mice and 2 to 3 days in humans is tightly controlled by two ovarian hormones: estrogen and progesterone [8].
Preparing the uterus for implantation is initiated by an epithelium-stroma crosstalk that has been proven to be regulated by PGR in several studies [9, 10]. The PGR in the endometrial epithelium mediates the signal from P4 to transcriptionally amplify the levels of Indian hedgehog (Ihh) signaling proteins before the embryo has been implanted. This increased epithelial Ihh then initiates the stromal hedgehog pathway to stimulate the expression of the downstream effector chicken ovalbumin upstream promoter transcription factor II (COUP-TFII). On the stromal cell side, COUP-TFII decreases estrogen signaling and stops epithelial proliferation, most likely via the heart and neural crest derivatives expressed transcript 2 (HAND2) orchestrated reduction of the fibroblast growth factor-extracellular signal-regulated kinase (FGF-ERK) pathway. However, it is unclear whether COUP-TFII mediates HAND2 directly or through PGR in the stroma, though COUP-TFII does directly promote PGR expression in the stroma and P4 does increase stromal HAND2 levels. At the physiological level, this initiation of crosstalk by epithelial PGR leads to a shift in the state of epithelial cells; from being proliferative into a state of differentiation, and hence ready for ensuing embryo implantation [8].
Additionally, the above epithelial pathway prepares the stroma for decidualization via COUP-TFII and in combination with epidermal growth factor receptor (EGFR) signaling in the stroma. Studies have found that in mouse models and cultured human endometrial cells, stromal COUP-TFII stimulates the expression of wingless-type MMTV integration site(WNT) family member 4 (WNT4) and thus stimulates decidualization. Furthermore, studies in mice with a uterus that is deficient in COUP-TFII have reported results of a positive regulatory function for COUP-TFII on the expression of bone morphogenetic protein 2 (BMP2) [11]. Downstream of BMP2, WNT4 is positively regulated by PGR and a member of the O-class of forkhead box (FOX) proteins, FOXO1 (Figure 1). It appears that stromal EGFR signals converge with the PR-IHH axis at BMP2 and WNT4 to affect decidualization [12]. Further downstream, researchers have speculated that the familiar serine/threonine proteininkinase: With-No-Lysine (K) 1 (WNK1) - mitogen-
activated protein kinase -7 (MAPK-7) axis may act subsequently of the EGFR pathway for decidualization in the stroma; however this role is still unclear[13]. Both PGR isoforms are important for pregnancy success in humans, with the PGR-B isoform playing a predominant role in decidualization [14]. Moreover, the endometrial epithelial PGR expression markedly decreases in the mid-secretory phase to enable normal uterine receptivity to occur. At the same time, it continues its abundance in stromal cells to maintain decidualization. Epithelial FOXO1 expression, on the other hand, is extremely high at this stage. This reciprocal expression relationship between PGR and FOXO1 is considered a critical step in the establishment of endometrial receptivity [2].
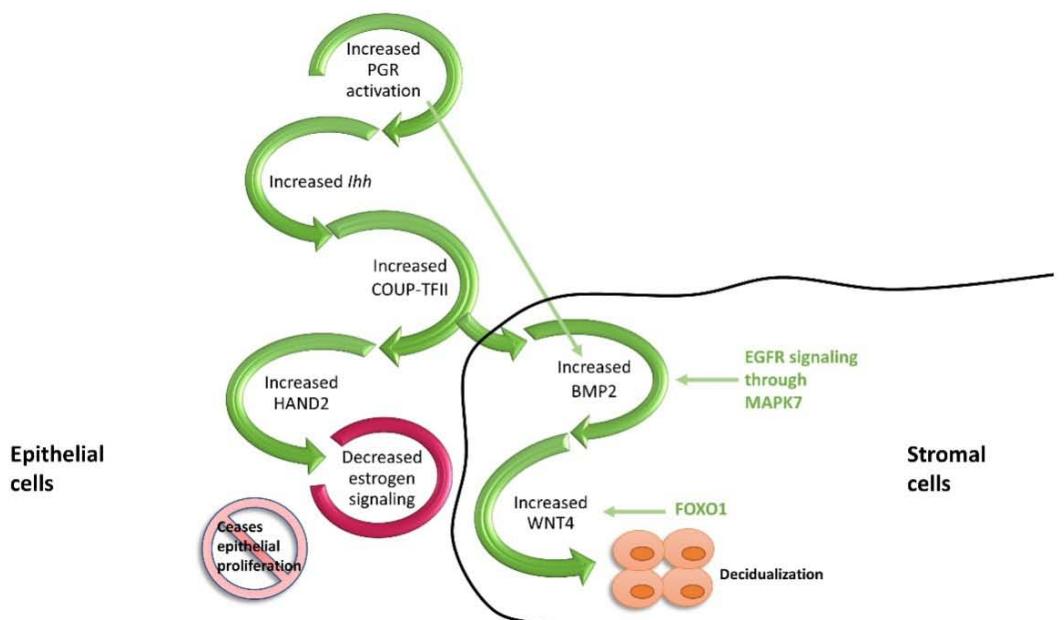 Figure 1. PGR-mediated crosstalk between the epithelial and stromal cells during implantation. P4 through its action in the epithelial cells initiates the Ihh axis which, in turn signals COUP-TFII and HAND2 to decrease estrogen-dependent proliferation. While at the stromal level, increased FOXO1 and hence increased WNT4 and BMP2 leads to decidualization.
Studies assessing the multiple processes involved in implantation and promoting receptivity of the uterus have recently highlighted the role of the pinopode in favorable fertility outcomes. The pinopode or the uterodome consists of small microvilli protrusions, flowerlike shapes, which develop on the apical surface of the luminal epithelium of the endometrium that is hormonally regulated [15]. Uterodomes are characterized by the presence of cell-adhesion molecules (integrins). P4 is responsible for the timely downregulation of estrogen receptors, an effect linked to the timely expression of integrin $\alpha v\beta 3$, which plays a role in blastocyst adhesion to the uterus. This is why several studies have stressed the dependency of pinopode formation on the two most studied hormones contributing to fertility, estrogen, and progesterone [16, 17]. The $\alpha v\beta 3$ integrin is regulated by a homeobox protein, the HOXA10 transcription factor, whose regulation is also controlled by P4.
The science so far has shown that other hormones such as adrenomedullin lead to an increase in pinopode growth; testosterone however leads to their decrease [18, 19]. It is therefore important to understand how hormones regulate pinopode growth and further investigate these relations.
## III. ROLE OF PROGESTERONE AND ITS RECEPTOR IN QUIESCENCE
Human uterine quiescence is characterized by an anti-inflammatory atmosphere generated through the myometrial PGR-B, the dominant isoform during this stage. It maintains the quiescent state throughout gestation by suppressing the transcription of genes encoding contraction-associated proteins (CAPs) [such as connexin43 (GJA1/CX43), prostaglandin receptor (F2a), and oxytocin receptor (OXTR)]; and by suppressing inflammatory genes, like nuclear factor kappa-light-chain-enhancer of activated B cells $(\mathsf{NF}_{-k}\mathsf{B})$
and interleukin-1(IL-1). Maintaining the quiescent state during pregnancy requires multifaceted and complex mechanisms that involve P4-PGR mediated interactions through genomic and non-genomic pathways.
Simply these pathways will lead to first of all the inhibition of the well-known contractile genes like CX43 and OXTR, by upregulating the expression of the transcriptional inhibitor zinc finger E-box-binding homeobox 1 (ZEB1) that binds to their promoters and suppress their expression to ensure the quiescent stage of pregnancy [20]. Additionally, these pathways will lead to the antagonizing of the proinflammatory transcription factors by direct interaction of PGR with $\mathrm{NF}_{-k}\mathrm{B}$ or activator protein-1 (AP-1). Evidence exists to support the notion that progesterone may directly or indirectly impair the $\mathrm{NF}_{-k}\mathrm{B}$ -mediated inflammatory cascade leading to labor. Experimental work has demonstrated that a mutual negative interaction exists between the PGR and $\mathrm{NF}_{-k}\mathrm{B}$ p65. Moreover, in human lower uterine segment fibroblasts and amnion epithelial cells, P4 was found to repress $\mathrm{NF}_{-k}\mathrm{B}$ transcriptional activity [21]. On the other hand, the repressor effect of PGR coupling with jun proto-oncogene (JUN)protein, a subfamily of the AP-1, to maintain myometrial quiescence has been suggested strongly by researchers [22]. Activated AP-1 is known to increase multiple pro-inflammatory cytokines [IL-1, interleukin-6 (IL-6), and interleukin-8(IL-8)] in the laboring human myometrium[23].
Furthermore, these pathways will influence the expression of genes that block proinflammatory transcription factors such as the $\mathrm{NF} - \kappa \mathrm{B}$ inhibitor, nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor, alpha $(\mathsf{l}_{\mathsf{k}}\mathsf{B}_{\alpha})$ - which prevents activation and nuclear translocation of NF- $\kappa \mathbb{B}$ p50 and p65 - and the mitogen-activated kinase (MAPK) inhibitor, MAPK phosphatase-1 (MKP-1 or DUSP1); which inhibits p38 MAPK activation and NF- $\kappa \mathbb{B}$ p65 nuclear translocation. As a result, P4-activated PGR maintains the quiescent state by inhibiting the activation of inflammatory response pathways[20].
The progesterone block hypothesis, first proposed by George Corner in 1942 and later expanded by Arpad Csapo in the 1950s, posits that P4 maintains pregnancy by blocking labor and that withdrawal of the P4 block is a key trigger for parturition[3].
## IV. ROLE OF PROGESTERONE AND ITS RECEPTOR IN PARTURITION
Parturition is the result of complex maternal and fetal interactions, whereby the quiescent uterus builds synchronized contractions and the cervix dilates to allow passage of the fetus through the birth canal [24]. For a successful result, maturation of fetal organs is required to ensure survival outside the uterus, in addition to maternal changes necessary to initiate lactation postpartum. As such, synchronized stimuli from both the mature fetus and the active uterus is desirable, with most evidence suggesting that the fetus itself triggers these two events [24].
Inhibition of P4 production or its function elicits labor by inducing inflammation of the decidua, remodeling of the cervix, promoting rupture of the fetal membrane, and increasing myometrial contractility [25]. The "progesterone receptor isoform switch" concept was posited to explain the transition from a quiescent to a contractile myometrial phenotype in the presence of high levels of progesterone [6]. At parturition, the rise in PGR-A expression promotes labor by inhibiting the anti-inflammatory actions of PGR-B and stimulating proinflammatory gene expression in response to progesterone [3].
The interplay of several genes, receptors, and enzymes affects the process of contractions in the myometrium. Studies so far have agreed on 5 genes and maybe more, that regulate the shift from the quiescent state to parturition. These genes - cyclooxygenase-2 (COX-2); prostanoid FP receptor (PgF2αR); OXTR; CX43; and gap junction protein - either suppress or activate muscular contractions thus leading to labor.
The increase in local metabolism of P4 in the myometrium and decrease in PGR function in the uterus and cervix near term is critical for parturition to commence in all mammals. Findings by Renthal et al (2015) revealed that the pivotal role that ZEB1 serves in myometrial contractility during pregnancy and labor is through mediating the opposite actions of P4 and estrogen. During the quiescent state, the elevated levels of P4 and the increased function of its receptors promote the upregulation of myometrial ZEB1. This increase leads to the inhibition of expression of the microRNA-200 (miR-200) family and the suppression of both OXTR and CX43. The decreased levels of miR-200 up-regulates ZEB1 further and increase zinc finger E-box-binding homeobox 2 (ZEB2), thus binding response elements upstream of the miR-199a/214 clusters to enhance its expression, which eventually suppresses COX-2, and block the contractile prostaglandins biosynthesis. The low levels of miR-200s allow the upregulation of the transcription factor, signal transducer, and activator of transcription 5B (STAT5b), which inhibits expression of $20\alpha$ -hydroxysteroid dehydrogenase $(20\alpha$ -HSD), and as such permits local myometrium levels of P4 to remain elevated [20].
As pregnancy transitions to labor, the decreased function of P4/PGR in the myometrium and increased circulating levels of estrogen and the activity of its receptor cause the downregulation of ZEB1. This then induces the miR-200 family and hence causes further suppression of ZEB1 and ZEB2, which in turn allows the up-regulation of expression of OXTR and CX43. The decreased levels of ZEB1/2 also lead to a decrease in expression of the miR199a/214 cluster, which allows the up-regulation of COX-2 and increased synthesis of contractile prostaglandins. The high expression of miR-200 inhibits STAT5b and as such permits the increased transcription of $20\alpha$ -HSD, which metabolizes P4 into inactive products in the myometrium [20]. Collectively, these molecular events contribute to the induction of contractions in the uterus and therefore lead to labor (Figure 2).
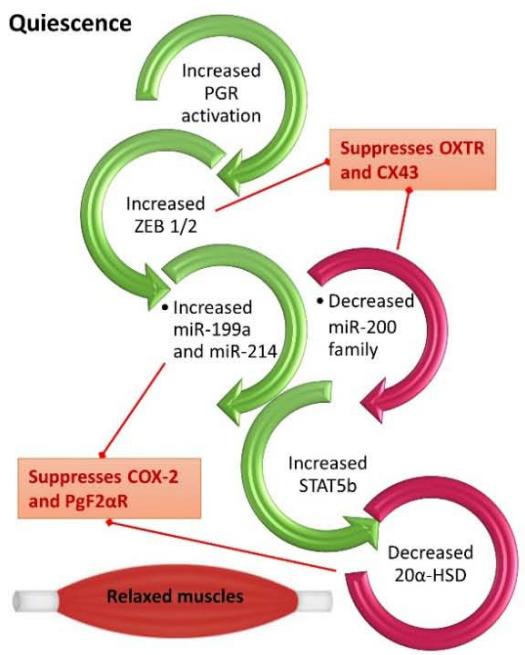
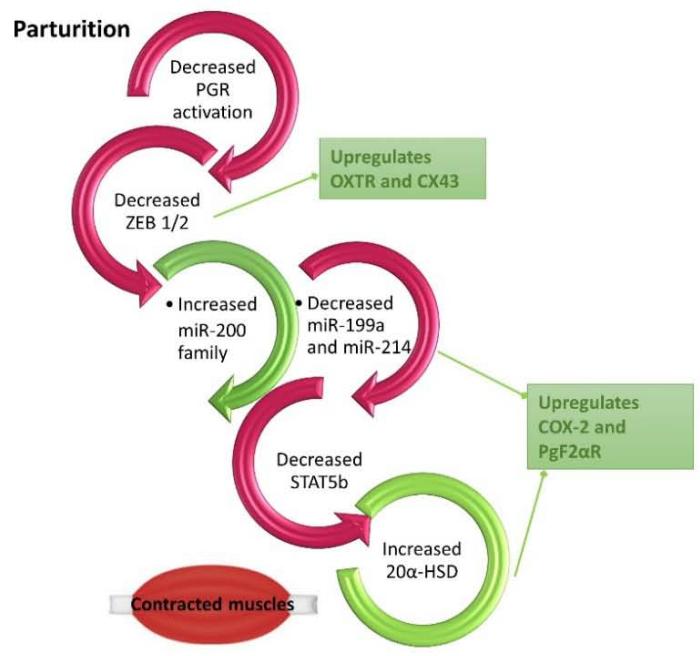 Figure 2. P4 and PGR mediation of quiescence and parturition. The regulation of PGR impacts ZEB1/2 and hence quiescence during pregnancy or contractility during labor.
Myometrial muscle cells contract together to generate a synchronized movement through the presence of intercellular bridges or gap junctions [26]. One protein that has been described when mediating the formation of gap junctions as term approaches is Gap junction alpha 1 (GJA1), also known as CX43. The reporter expression downstream of a synthetic GJA1 promoter is increased by co-expression of constructs encoding members of the AP-1 transcription factor; namely FBJ osteosarcoma oncogene (FOS) and JUN subfamilies. In humans, it appears that the levels of JUN protein remain relatively constant in the myometrium throughout gestation, whereas an increase in levels of FOS and Fos-like antigen 2 (FOSL2) proteins is reported during labor inside the nuclei of the myometrial cells. Several JUN subfamily members are present in the uterine smooth muscle during the quiescent phase of pregnancy. It is therefore possible that the JUN family plays a role in maintaining the quiescence, and that they require heterodimerization with a partner from the FOS subfamily to stimulate genes for the initiation of labor [26].
## V. CONCLUSION
While inflammatory changes are required for both implantation and parturition, the genetic process is totally different between the two phases. The studies conducted so far have not identified similar pathways when it comes to genetic mediation of implantation and labor. It appears that the only common denominator between these two phases of pregnancy is P4 and its receptors, PGR-A and PGR-B. Further research is needed to determine whether these two phases are indeed mirrored images as the data so far indicates that quiescence and parturition are the true mirror images.
Generating HTML Viewer...
References
26 Cites in Article
J Graham,Christine Clarke (1997). Physiological Action of Progesterone in Target Tissues*.
Francesco Demayo,John Lydon (2020). 90 YEARS OF PROGESTERONE: New insights into progesterone receptor signaling in the endometrium required for embryo implantation.
Huiqing Tan,Lijuan Yi,Neal Rote,William Hurd,Sam Mesiano (2012). Progesterone Receptor-A and -B Have Opposite Effects on Proinflammatory Gene Expression in Human Myometrial Cells: Implications for Progesterone Actions in Human Pregnancy and Parturition.
P Kastner,A Krust,B Turcotte,U Stropp,L Tora,H Gronemeyer,P Chambon (1990). Two distinct estrogen-regulated promoters generate transcripts encoding the two functionally different human progesterone receptor forms A and B..
D Wen,Y Xu,D Mais,M Goldman,D Mcdonnell (1994). The A and B isoforms of the human progesterone receptor operate through distinct signaling pathways within target cells..
M Peavey,S Wu (2021). Progesterone receptor isoform B regulate the Oxtr-Plcl2-Trpc3 pathway to suppress uterine contractility.
Milena Girotti,Hans Zingg (2003). Gene Expression Profiling of Rat Uterus at Different Stages of Parturition.
S Wu,R Li,F Demayo (2018). Progesterone Receptor Regulation of Uterine Adaptation for Pregnancy.
Jian Tan,Bibhash Paria,Sudhansu Dey,Sanjoy Das (1999). Differential Uterine Expression of Estrogen and Progesterone Receptors Correlates with Uterine Preparation for Implantation and Decidualization in the Mouse1.
Margeaux Wetendorf,San-Pin Wu,Xiaoqiu Wang,Chad Creighton,Tianyuan Wang,Rainer Lanz,Leen Blok,Sophia Tsai,Ming-Jer Tsai,John Lydon,Francesco Demayo (2017). Decreased epithelial progesterone receptor A at the window of receptivity is required for preparation of the endometrium for embryo attachment†.
Isao Kurihara,Dong-Kee Lee,Fabrice Petit,Jaewook Jeong,Kevin Lee,John Lydon,Francesco Demayo,Ming-Jer Tsai,Sophia Tsai (2007). COUP-TFII Mediates Progesterone Regulation of Uterine Implantation by Controlling ER Activity.
Michael Large,Margeaux Wetendorf,Rainer Lanz,Sean Hartig,Chad Creighton,Michael Mancini,Ertug Kovanci,Kuo-Fen Lee,David Threadgill,John Lydon,Jae-Wook Jeong,Francesco Demayo (2014). The Epidermal Growth Factor Receptor Critically Regulates Endometrial Function during Early Pregnancy.
Nyssa Adams,Yasmin Vasquez,Qianxing Mo,William Gibbons,Ertug Kovanci,Francesco Demayo (2017). WNK lysine deficient protein kinase 1 regulates human endometrial stromal cell decidualization, proliferation, and migration in part through mitogen-activated protein kinase 7.
Hatice Kaya,Alison Hantak,Lisa Stubbs,Robert Taylor,Indrani Bagchi,Milan Bagchi (2015). Roles of Progesterone Receptor A and B Isoforms During Human Endometrial Decidualization.
Kelsey Quinn,Brooke Matson,Margeaux Wetendorf,Kathleen Caron (2020). Pinopodes: Recent advancements, current perspectives, and future directions.
Alex Lopata,Ursula Bentin-Ley,Allen Enders (2002). “Pinopodes” and Implantation.
Brooke Matson,Stephanie Pierce,Scott Espenschied,Eric Holle,Imani Sweatt,Eric Davis,Robert Tarran,Steven Young,Trudy Kohout,Marcel Van Duin,Kathleen Caron (2017). Adrenomedullin improves fertility and promotes pinopodes and cell junctions in the peri-implantation endometrium†.
H Mokhtar (2014). Testosterone decreases the expression of endometrial pinopode and L-selectin ligand (MECA-79) in adult female rats during the uterine receptivity period.
Nora Renthal,Koriand’r Williams,Alina Montalbano,Chien-Cheng Chen,Lu Gao,Carole Mendelson (2015). Molecular Regulation of Parturition: A Myometrial Perspective.
D Hardy (2006). Progesterone Receptor Plays a Major Antiinflammatory Role in Human Myometrial Cells by Antagonism of Nuclear Factor-κB Activation of Cyclooxygenase 2 Expression.
Lubna Nadeem,Oksana Shynlova,Elzbieta Matysiak-Zablocki,Sam Mesiano,Xuesen Dong,Stephen Lye (2016). Molecular evidence of functional progesterone withdrawal in human myometrium.
David Macintyre,Yun Lee,Roberta Migale,Bronwen Herbert,Simon Waddington,Donald Peebles,Henrik Hagberg,Mark Johnson,Phillip Bennett (2014). Activator protein 1 is a key terminal mediator of inflammation‐induced preterm labor in mice.
J Challis (2000). Endocrine and Paracrine Regulation of Birth at Term and Preterm.
Virlana Shchuka,Luis Abatti,Huayun Hou,Nawrah Khader,Anna Dorogin,Michael Wilson,Oksana Shynlova,Jennifer Mitchell (2020). The pregnant myometrium is epigenetically activated at contractility-driving gene loci prior to the onset of labor in mice.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Hisham Arab. 2026. \u201cParturition is Not the Mirror Image of Implantation: The Role of Progesterone\u201d. Global Journal of Medical Research - E: Gynecology & Obstetrics GJMR-E Volume 23 (GJMR Volume 23 Issue E1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
There has been an abundance of evidence from clinical, animal, and in vitro studies that progesterone (P4) is important for establishing, maintaining, and terminating a pregnancy. P4 exerts its primary action via its two receptors: progesterone receptor A (PGR-A) and B (PGR-B). Analyses of transcriptome and cistrome genome have unearthed novel members and modifiers of the P4 signaling pathway. The increase in serum P4 levels and down-regulation of PGR-B are important in the development of pinopodes, thus marking implantation. Additionally, it promotes the quiescent myometrial cell phenotype, and the inhibition of its production in myometrial cells induces labor and is the key physiologic initiator of parturition. Through genomic and non-genomic intracellular mechanisms involving these PGR isoforms, various physiologic states can be determined such as the quiescent and contractile myometrium during pregnancy.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.