## I. INTRODUCTION
Lipoproteins have been an important intersectional carrier between cardiac and hormonal functions. Lipopeptide LPK (L-PEG $_{12}$ -(KIAALKE) $_3$ ) is involved in membrane destabilizing with the electrostatic activity of LPE (L-PEG $_{12}$ -(EIAALEK) $_3$ ) in membrane fusion activities (1); cell entry of the hepatitis C virus, for example, is aided by apolipoprotein C-I (ApoC-I) for membrane fusion (2). High-density lipoprotein (HDL) is differentiated to cell-penetrating peptides' mutants and fusogenic lipids at mildly acidic pH, with the latter showing higher affinity to the cell plasma membrane during the process (3). Endocytic uptake of SARS-CoV-2 S2 protein induces fusogenic activities (4, 5), which is key to the pathogenic severities not only limited to SARS-COV-2 (6, 7). The correlational study of low-density lipoprotein - cholesterol (LDL-C) to COVID-19 severity (8) and clinical results of NCT05711810 trial led to the hypothesis on S2 proteins' hibernation concentration being on LDL-C.
HDL and LDL's steroidogenesis capacities (9) further justified a sebaceous immunobiological review. While Apolipoprotein B (apoB)-containing lipoproteins, including chylomicrons, very-low-density lipoprotein (VLDL), and LDL, carry and deliver triglycerides and cholesterol, HDL mediates Reverse Cholesterol Transport (RCT), with apoA-I for anti-atherogenic properties (10). An LDL and a molecule of apolipoprotein B100 covalently bound, via disulfide bonds, to a plasminogen-like particle apolipoprotein(a) [Apo(a)], compose of the plasma lipoprotein lipoprotein(a) [lp(a)](11). Insulin levels play a critical role in lp(a) degradation through the hepatic synthesis of Apo(a), whereby lp(a) particles have the capacity to cross the endothelial barrier and the arterial intima in promoting atherogenesis and delivering LDL-C (11).
The internal mitochondrial synthesis paths have thus become the focus of the review. Mitochondrialfunctions include DNA segregation, oxidative phosphorylation efficiency, reactive oxygen species (ROS) production, and apoptosis ${ }^{(12)}$. ROS is considered the key physiological response biomechanism in the review ${ }^{(13)}$. From the clinical trial experience, it is hypothesized that SARS-CoV-2 S2 protein competes for ROS with host physiology, and it is thereafter anchored for practical safety concerns in the review with the potential pharmacokinetic targeting solution purpose.
## II. REVIEW
The review is structured from the opening on interactions between melatonin and melanin. Melatonin actions are mediated through its cognate membrane bound type 1 and 2 (MT1 and MT2) receptors or through receptor-independent mechanisms; they can affect the phenotype of normal human melanocytes (14). Melatonin can downregulate the paracrine factors Endothelin-1 (ET-1) and prostaglandin synthase 2 (PTGS2) in the keratinocytes by inhibiting the janus kinase 2/signal transducer and activator of transcription 3 (JAK2/STAT3) pathway, which reduces melanogenesis and melanin production in pigment cells (15, 16).
With the neuronal-sebaceous ROS dynamics, the second subsection reviews the mitochondria-driven adenosine triphosphatase (ATP) synthesis. Endocytotic pathways are evaluated after the downstream mitochondrial activities revolving around the ROS. The third subsection examines the upstream steroidogenesis, specifically focusing on macropinocytosis. RCT in the fourth subsection concentrates on SARS-CoV-2's main pathogenesis by the complexities in autophagy and the autophag-lysosome pathway. With the sebaceous immunobiological pathways, SARS-CoV-2's vaccination poisoning trails in human hosts are analyzed. The fifth subsection reviews the melanin involvement in adrenocorticotropic hormone (ACTH) $^{(17,18)}$.
a) The Downstream Basis of Sebaceous Immunobiology
Melatonin synthesis involves both hydroxylation and decarboxylation with the aromaticL-aminoacid, and acetylserotonin O-methyltransferase (ASMT) (19); it occurs in the mitochondrial matrix in some species, where chloroplasts are also capable of (20). Melatonin secretion is reported to have direct and varying associations with leukocytes and leukocyte levels (21), and melatonin nuclear binding receptors are identified in human lymphocytes and monocytes (22). Statistical experiment exhibits a circadian effect on human melatonin inhibition of leukocyte levels (23). Its impact on neutrophils is involved in apoptosis in carp and zebrafish (24, 25).
The chemo-physiological associations between melatonin and sebaceous immunobiology are intertwined with the circadian effect. Ex vivo study evidenced melatonin and its metabolites' protection function on melanocytes from ultraviolet B radiation (UVB), including its adverse impact on keratinocytes (14). Methoxytryptamine receptor independent protective effects against UVB-induced DNA-damaging ROS generation from epidermal melanocytes stimulate UVB irradiated melanocytes' expression of major antioxidant enzymes, including catalase (CAT), glutathione peroxidase (GPx), glutathione reductase (GR), glutathione-S-transferase (GSTP1), superoxide dismutase (Cu/Zn-Sod and Mn-SOD), glutamylcysteine synthetase (GCS), and NAD(P)H dehydrogenase, quinine 2 (NQO2) (14). Albeit melatonin promotes melanin synthesis in the human SK-MEL-1 melanoma cells, it also inhibits, with a different chemical process, melanin synthesis from UVB irradiation induced melanin pigmentation (26).
### b) Mitochondrial Pathway in Adenosine Triphosphatase
The phenomenon of mitochondrial flashes (mitoflashes) is considered a biomarker for mitochondrial energy metabolism under certain conditions, and protons produced by photolysis or electroneutral proton ionophores act as a powerful mitoflash trigger (27). Combined with my previous proton-production review, where protons are made in reactions in which ATP is hydrolyzed or the oxidized form of nicotinamide-adenine dinucleotide $(\mathrm{NAD}^{+})$ is reduced $(\mathrm{RH}_{2} + \mathrm{NAD}^{+} \rightarrow \mathrm{NADH} + \mathrm{H}^{+} + \mathrm{R})$ (28), and cardiac mitochondria regulate and energize ATP levels (29);
anions inhibit mitochondrial ATP production and decrease mitoflash frequencies, and vice versa, cations trigger mitofilases (27). Electron transport chain (ETC) from proton-coupled electron transfer (PCET) provides energy with heat generation to ATP synthesis; less concise is the evidence for ROS interactions with proton-motive force (PMF) and cation fluxes, possibly contributed by the rapidly shifting dipole fields (27, 29).
Hydrogenic (cation-based) and oxidative (anion-based) phosphorylation differences provide further insights. Even though cations, in general, promote mitofilases, phosphine $(\mathsf{PH}_3)$ toxicity exists with its functions on lipid peroxidation from its superoxidization of ROS, by intoxicating mitochondria and by changing the latter's homeostasis (30). The phenomena in comparison to the Fe anions in oxidative phosphorylation suggest that ETC-mediated ROS generation from mitofilases originates from the proton-gating process, wherein the gating pore's depolarization cut-off ionizes the hydrolysis chain for ROS production catalyzed by the PMF (13, 27, 29, 31). The mitochondrion is considered an energy-producing organelle that generates ROS and heat as byproducts of oxidative phosphorylation (10).
### c) Sebaceous Pathway in Upstream Mitochondria
While HDL-C mediates RCT, LDL-C is internalized into cells through LDL receptor (LDLR)-mediated endocytosis, an important source for steroidogenesis (10, 32). The LDL is degraded frequently in lysosomes, and its cholesterol is released for use in the synthesis of membranes, steroid hormones, and bile acids (33). Coated pits in discrete regions of the surface membrane where receptors cluster, invaginate into the cell to form endocytic vesicles, enclosed by extracellular ligand; the LDL receptor enters the cell together with LDL, after which it recycles to the surface, strongly up-regulated by estrogen in pituitary somatolactotropic $\mathrm{GH}_3$ cells (33, 34). Indirect evidence shows that ezrin phosphorylation to phosphoezrin is involved in the receptor-mediated endocytotic process, corroborating with the mitochondrial pathway analysis in section 2.2 (34, 35).
Lipoproteins primarily manifest in plasma forms during endocytosis. Macropinocytosis is not only actively involved in the uptake of $\mathrm{lp(a)}$, but it also represents an important endocytic uptake mechanism for native LDL in macrophages during atherogenesis (36, 37). A relational chart among clathrin-mediated endocytosis, clathrin-independent endocytosis, macropinocytosis, and transcytosis is seen in Figure 1 (37).
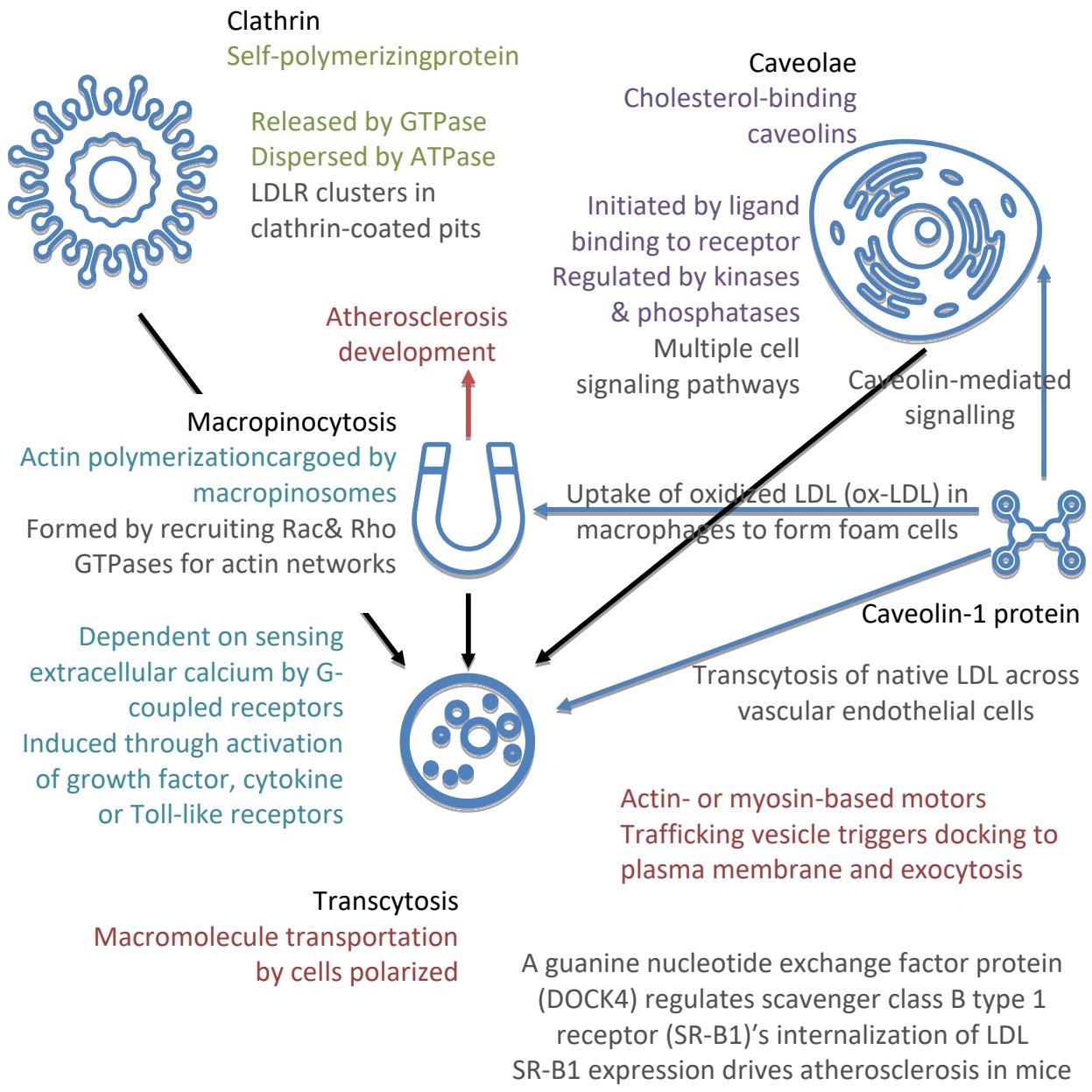 Figure 1: Main Endocytosis Forms of LDL-Related Lipoproteins and Atherosclerosis Development in the Endocytotic Chains.
Dynamin both Catalyzes Mature Clathrin-Coated Vesicle Releases and Promotes Membrane Fissionin Release of Caveolin-Coated Vesicles. SR-B1 Receptor is Traditionally Thought of as an HDL Receptor.
While caveolin-1 protein is mainly responsible for ox-LDL in endocytosis, possibly contributed by the ATP dispersion effects, macropinocytosis, initiated by extracellular calcium, is mainly responsible for $\mathrm{lp(a)}$ uptake by intracellular engulfment of cargoes and $\mathrm{lp(a)}$ cell surface binding (36). No direct evidence in the archival research is obtained on macrophage infection by SARS-CoV-2, albeit the calcium channel infection path and amelioration of $\mathrm{lp(a)}$ -induced atherosclerotic burden by selective serotonin reuptake inhibitors (SSRIs) suggest a plausible correlative risk during the innate immune response (36, 38, 39). Further in vitro experiments are needed to determine the concise pathological depths beyond the autophagy-lysosome pathway, especially concerning the confounding variables of electronegative LDL (L5) and ApoE $^{(40-42)}$.
### d) Reverse Cholesterol Transport In Sebaceous Immune Chains
RCT links hematology with lipid metabolism by autophagy and the autophagy-lysosome pathway. The three main lipid metabolism pathways are exogenous, endogenous, and RCT, with the last crucial for homeostasis (43). SARS-CoVs' immune escape capabilities concentrate on the autophagy-lysosome pathway, by hijacking autophagy initiation to form unique double-membrane vesicles (DMVs) and by hijacking host cell endoplasmic reticulum associated degradation (ERAD) machinery (42). The risen HDL-C indicator to the decreased prognosis of SARS-CoV-2 severe and critical cases, therefore, is relatively significant if macrophage infection potentials are eliminated (8).
Lipolysis is critical in SARS-CoV-2 vaccines' lethal adverse symptoms, besides carcinogenesis. Since macropinocytosis is induced through activation of growth factor, cytokine, or Toll-like receptors, carcinogenesis is mainly contributed by DMVs' formation (37, 42). With its blocking of autophagy, namely RCT, epithelial homeostasis is broken in the exogenous pathway for lipid metabolism mediated by epidermal growth factor (EGF) (43, 44). It is therefore hypothesized that rapid acidification is not the cause/precondition, but the effect of SARS-CoV-2 infection from the cholesterol cell locking, resulting in host cell lysis (43, 45, 46). The subsequent pathway blocked is the mitochondrial ATP synthesis (43). This is why even when SARS-CoV-2 vaccines targeted the S1 protein's binding to angiotensin-converting enzyme 2 (ACE2), myocardial adverse events still happen with the S2's influence (47).
Even if there's no direct SARS-CoV macrophage infection, macrophages' functions in the subsidiary caveolin-1 protein's intracellular lipid metabolism are still compromised seen in Figure 1. LipidpoorApoA-I, secreted by the liver and intestines, is lipidated in the circulation via cholesterol efflux by transporters located in peripheral tissues and macrophages, generating mature HDLs, which are hydrolyzed or returned to circulation for relipidation $^{(43)}$. The upstream ROS and steroidogenesis are blocked, resulting in endocrine and neurological symptoms after SARS-CoV-2 vaccination. Macrophage apoptosis from host cell lysis creates further risks of atherosclerosis and secondary necrosis ${ }^{(43)}$.
### e) Autophagy Induction and the Upstream ROS
The RCT's mitochondria-protective functions are reflected in HDL. Lipid peroxides in oxLDL stimulate ROS formation and impair oxygen consumption at Complexes I, II/III, and IV of the respiratory chain, resulting in mitochondrial dysfunction (10). Antioxidization by hydrolyzing cholesteryl esters and phospholipids in oxidized lipoproteins functions via the HDL-associated protein Paraoxonase 1 (PON1); the lysosphingolipid Sphingosine 1-Phosphate (S1P) of HDL, synthesized in hematopoietic and endothelial cells, induces Reperfusion Injury Salvage Kinase (RISK) and Survivor Activating Factor Enhancement (SAFE) pathways, with the STAT3-mediated attenuation of mitochondrial injury (10). The symbiotic upstream ROS dynamics amongst mitochondria, ATP, and melanin are thus interlinked by macroautophagy in sebaceous immunobiology (10, 48).
ATP signaling is mediated by binding to purinergic receptors type P2, expressed by human keratinocytes, Langerhans cells, and fibroblasts (48). The P2X receptor family consists of seven different ligand-gated ion channels that elicit the flow of cations $(\mathrm{Na}^{+}, \mathrm{K}^{+}$, and $\mathrm{Ca}^{+2}$ ) when activated by extracellular ATP; $\mathrm{P2Y}_{1}$ and $\mathrm{P2Y}_{2}$ receptors are expressed by proliferating keratinocytes in the basal layer of the epidermis, and keratinocyte cell numbers in vitro can be increased by low concentrations of ATP, uridine $5^{\prime}$ -triphosphate, and adenosine $5^{\prime}$ -diphosphate (ADP) (48). Further detailed research in the correlations to clathrin-independent endocytosis may shed new light on the complexities in sebaceous immunobiology, including PMF's roles in Complex I & III ROS production currently concentrated on iron-sulfur clusters (13, 31, 37, 48).
Extra-adrenal and extra gonadalsteroidogenesis is critical for upstream signaling of sebaceous immunobiology. Toll-like receptors are the key element in innate sebaceous immunology, compared to the T cell and B cell adaptive immunity (49). While vitamin D levels' photochemical synthesis by the absorption of UVB energy by 7-dehydrocholesterol in the epidermis can be hydrolyzed at positions other than C25 by CYP11A1 in steroidogenic organs, the assumption on the reverse correlation between the activity of the cutaneous pigmentary system and vitamin D production is widely accepted by experts in the field of vitamin D (50). Extra-adrenal and extra gonadalsteroidogenesis take place where CYP11A1 is expressed, such as in both breast tumors and surrounding normal breast tissue, in the human gut and colon cancer cells, and in the heart, skin and cells of the immune system, with pathways that commence from cholesterol (49). The hypothalamic-pituitary-adrenal (HPA) axis in the psychiatric focus of suicidology regulates localsteroidogenic activity by phenotypes (49).
The HPA axis, therefore, can also be influenced by mitochondrial ROS dynamics in the heart. The two major endogenous ROS sources across the endocytotic spectra, i.e., oxidative phosphorylation and proton gradient across mitochondrial membrane potential, correlate to cardiac polarity and depolarization (31). Myocardial infarction might occur by ischemia conversion of ATP to ADP and adenosine monophosphate (AMP), impacting cyclic AMP (cAMP) and damaging innate immunity (31, 51).
## III. RESULT
Albeit further research and review are needed to determine the endocrine dysfunctions caused by SARS-CoV infections, the causal infection path from $\text{lp(a)}$ is inferentially falsified in the study. Lipoprotein indicators in COVID-19 severity are phenomenological to ROS damages in the sebaceous immunobiological chain. The plausibility elimination of the steroidogenesis pathway has narrowed down the neuronal infection paths to stem cell migration pathways for the unvaccinated, and stem cell and leukocytes for the vaccinated.
## IV. CONCLUSIONS
Ischemia-induced AMP homeostasis dysregulation is the key lethal element in COVID-19 vaccination poisoning. With the viral infection potentials on human embryonic stem cells, COVID-19 vaccination diffuses viral concentration while increasing its productive infection capacities (47, 52, 53). Not only the coercive mandates on COVID-19 vaccination and tracking need to be lifted, but also it is medically recommended against COVID-19 vaccination.
Stem-targeting $\mathsf{H}_2\mathsf{O}_2$ may treat stem cell infection by apoptosis, given the clearance on intact upstream sebaceous pathways and AMP homeostasis (52). Apart from the concentration on SARS-CoV-2 therapeutic explorations, the review has roughly mapped sebaceous immunobiology in relation to the earth's environment. Further research study on the centromeres' roles will be published separately.
### ACKNOWLEDGMENTS
I thank the Global Journals in the support in publication and in making the review publicly available through Open Access publication.
#### FUNDING
The research/review is not funded.
### CONFLICT OF INTEREST
The author declares no conflict of interest.
### DATA AVAILABILITY
No data associated with the review.
Generating HTML Viewer...
References
53 Cites in Article
Tingting Zheng,Yun Chen,Yu Shi,Huanhuan Feng (2019). High efficiency liposome fusion induced by reducing undesired membrane peptides interaction.
M Dreux,B Boson,Ricard Blum,S Molle,J Lavillette,D Bartosch,B (2007). The exchangeable apolipoprotein ApoC-I promotes membrane fusion of hepatitis C virus.
O Petersen,O Gerasimenko,J Gerasimenko (2020). Endocytic uptake of SARS-CoV-2: the critical roles of pH, Ca2+, and NAADP.
Xiaojie Xu,Guangle Li,Bingbing Sun,Yi Zuo (2022). S2 Subunit of SARS-CoV-2 Spike Protein Induces Domain Fusion in Natural Pulmonary Surfactant Monolayers.
Wei Qiang,Yan Sun,David Weliky (2009). A strong correlation between fusogenicity and membrane insertion depth of the HIV fusion peptide.
A Saito,T Irie,R Suzuki,T Maemura,H Nasser,K Uriu (2021). Enhanced fusogenicity and pathogenicity of SARS-CoV-2 Delta P681R mutation.
Mengmeng Zhao,Zhen Luo,Hua He,Bo Shen,Jinjun Liang,Jishou Zhang,Jing Ye,Yao Xu,Zhen Wang,Di Ye,Menglong Wang,Jun Wan (2021). Decreased Low-Density Lipoprotein Cholesterol Level Indicates Poor Prognosis of Severe and Critical COVID-19 Patients: A Retrospective, Single-Center Study.
E Reaven,J Boyles,M Spicher,S Azhar (1988). Evidence for surface entrapment of cholesterol-rich lipoproteins in luteinized ovary..
C White,G Datta,S Giordano (2017). High-Density Lipoprotein Regulation of Mitochondrial Function.
Angelo Silverio,Francesco Cancro,Marco Di Maio,Michele Bellino,Luca Esposito,Mario Centore,Albino Carrizzo,Paola Di Pietro,Anna Borrelli,Giuseppe De Luca,Carmine Vecchione,Gennaro Galasso (2022). Lipoprotein(a) levels and risk of adverse events after myocardial infarction in patients with and without diabetes.
S Clayton,L Macdonald,M Kurowska-Stolarska,A Clark (2021). Mitochondria as Key Players in the Pathogenesis and Treatment of Rheumatoid Arthritis.
B Berry,A Trewin,A Amitrano,M Kim,A Wojtovich (2018). Use the Protonmotive Force: Mitochondrial Uncoupling and Reactive Oxygen Species.
Z Janjetovic,S Jarrett,E Lee,C Duprey,R Reiter,A Slominski (2017). Melatonin and its metabolites protect human melanocytes against UVB-induced damage: Involvement of NRF2-mediated pathways.
Mengie Ayele,T,Tilahun Muche,Z,Behaile Teklemariam,A,Bogale Kassie,A,Chekol Abebe,E (2022). Role of JAK2/STAT3 Signaling Pathway in the Tumorigenesis, Chemotherapy Resistance, and Treatment of Solid Tumors: A Systemic Review.
S Yang,H Zeng,L Jiang,C Fu,L Gao,L Zhang (2023). Melatonin reduces melanogenesis by inhibiting the paracrine effects of keratinocytes.
J Blalock,C Harp (1981). Interferon and adrenocorticotropic hormone induction of steroidogenesis, melanogenesis and antiviral activity.
Christopher Natale,Elizabeth Duperret,Junqian Zhang,Rochelle Sadeghi,Ankit Dahal,Kevin O'brien,Rosa Cookson,Jeffrey Winkler,Todd Ridky (2016). Sex steroids regulate skin pigmentation through nonclassical membrane-bound receptors.
M Rath,S Coon,F Amaral,J Weller,M Møller,D Klein (2016). Melatonin Synthesis: Acetylserotonin O-Methyltransferase (ASMT) Is Strongly Expressed in a Subpopulation of Pinealocytes in the Male Rat Pineal Gland.
Dake Zhao,Yang Yu,Yong Shen,Qin Liu,Zhiwei Zhao,Ramaswamy Sharma,Russel Reiter (2019). Melatonin Synthesis and Function: Evolutionary History in Animals and Plants.
Kenji Obayashi,Keigo Saeki,Norio Kurumatani (2015). Higher melatonin secretion is associated with lower leukocyte and platelet counts in the general elderly population: the HEIJO‐KYO cohort.
V Srinivasan,Gjm Maestroni,D Cardinali,A Esquifino,Sr Perumal,S Miller (2005). Melatonin, immune function and aging.
Roberto Zivieri,Fabio Borziani,Angela Strazzanti,Angela Fragomeni,Nicola Pacini (2018). Effect of Indolic-Amide Melatonin on Blood Cell Population: A Biophysical Gaussian Statistical Analysis.
M Kepka,E Szwejser,L Pijanowski,Bml Verburg-Van Kemenade,M Chadzinska (2015). A role for melatonin in maintaining the pro-and anti-inflammatory balance by influencing leukocyte migration and apoptosis in carp.
D-L Ren,A-A Sun,Y-J Li,M Chen,S-C Ge,B Hu (2015). Exogenous melatonin inhibits neutrophil migration through suppression of ERK activation.
Li-Ping Ma,Meng-Meng Liu,Fang Liu,Bo Sun,Si-Nian Wang,Jie Chen,Hui-Jie Yu,Juan Yan,Mei Tian,Ling Gao,Qing-Jie Liu (2023). Melatonin inhibits senescence-associated melanin pigmentation through the p53-TYR pathway in human primary melanocytes and the skin of C57BL/6 J mice after UVB irradiation.
Xianhua Wang,Xing Zhang,Di Wu,Zhanglong Huang,Tingting Hou,Chongshu Jian,Peng Yu,Fujian Lu,Rufeng Zhang,Tao Sun,Jinghang Li,Wenfeng Qi,Yanru Wang,Feng Gao,Heping Cheng (2017). Mitochondrial flashes regulate ATP homeostasis in the heart.
W-Z Zeng,T-L Xu (2012). Proton production, regulation and pathophysiological roles in the mammalian brain.
(2023). Unknown Title.
A Sciuto,B Wong,M Martens,H Hoard-Fruchey,M Perkins (2016). Phosphine toxicity: a story of disrupted mitochondrial metabolism.
Y-R Chen,J Zweier (2014). Cardiac Mitochondria and Reactive Oxygen Species Generation.
Yu-Xia Zhou,Jian Wei,Gang Deng,Ao Hu,Pu-Yu Sun,Xiaolu Zhao,Bao-Liang Song,Jie Luo (2023). Delivery of low-density lipoprotein from endocytic carriers to mitochondria supports steroidogenesis.
Joseph Goldstein,Richard Anderson,Michael Brown (1982). Receptor‐Mediated Endocytosis and the Cellular Uptake of Low Density Lipoprotein.
P Smith,A Cowan,B White (2004). The Low-Density Lipoprotein Receptor Is Regulated by Estrogen and Forms a Functional Complex with the Estrogen-Regulated Protein Ezrin in Pituitary GH3 Somatolactotropes.
J Goldstein,M Brown (2009). The LDL Receptor.
Gmi Redpath,N Deo,H Siddiqui,G Madani,N Kapoor-Kaushik,N Ariotti (2021). Serotonin and common antidepressants regulate lipoprotein(a) macropinocytosis via enhanced cell surface binding.
Halima Siddiqui,Nikita Yevstigneyev,Golnoush Madani,Sally Mccormick (2022). Approaches to Visualising Endocytosis of LDL-Related Lipoproteins.
Mai Abdelmoaty,Pravin Yeapuri,Jatin Machhi,Katherine Olson,Farah Shahjin,Vikas Kumar,You Zhou,Jingjing Liang,Kabita Pandey,Arpan Acharya,Siddappa Byrareddy,R Mosley,Howard Gendelman (2021). Defining the Innate Immune Responses for SARS-CoV-2-Human Macrophage Interactions.
Sascha Berlansky,Matthias Sallinger,Herwig Grabmayr,Christina Humer,Andreas Bernhard,Marc Fahrner,Irene Frischauf (2022). Calcium Signals during SARS-CoV-2 Infection: Assessing the Potential of Emerging Therapies.
Wei-Yu Chen,Yun-Fang Chen,Hua-Cheng Chan,Ching-Hu Chung,Hsien-Yu Peng,Yu-Cheng Ho,Chu-Huang Chen,Kuan-Cheng Chang,Chih-Hsin Tang,An-Sheng Lee (2020). Role of apolipoprotein E in electronegative low-density lipoprotein-induced mitochondrial dysfunction in cardiomyocytes.
W He,Y Gao,J Zhou,Y Shi,D Xia,H Shen (2022). Friend or Foe? Implication of the autophagy-lysosome pathway in SARS-CoV-2 infection and COVID-19.
Haowei Liang,Dan Luo,Hai Liao,Shun Li (2022). Coronavirus Usurps the Autophagy-Lysosome Pathway and Induces Membranes Rearrangement for Infection and Pathogenesis.
A Remmerie,C Scott (2018). Macrophages and lipid metabolism.
Christos Zouboulis (2013). Epidermal growth factor receptor and the sebaceous gland.
C Morrison,C Edwards,K Shaffer,K Araba,J Wykoff,D Williams (2022). SARS-CoV-2 infection of airway cells causes intense viral and cell shedding, two spreading mechanisms affected by IL-13.
Y Pachankis (2023). Theoretical Strategies in SARS-CoV-2 Human Host Treatment.
Abdel-Malek Za (2019). Fueling Melanocytes with ATP from Keratinocytes Accelerates Melanin Synthesis.
A Slominski,B Zbytek,G Nikolakis,P Manna,C Skobowiat,M Zmijewski (2013). Steroidogenesis in the skin: implications for local immune functions.
Andrzej Slominski,Arnold Postlethwaite (2015). Skin Under the Sun: When Melanin Pigment Meets Vitamin D.
Carlos Serezani,Megan Ballinger,David Aronoff,Marc Peters-Golden (2008). Cyclic AMP.
Y Sui,Z Zhao,R Liu,B Cai,Fan (2014). Adenosine monophosphate-activated protein kinase activation enhances embryonic neural stem cell apoptosis in a mouse model of amyotrophic lateral sclerosis.
W Zeng,F Xing,Ji Yang,S Xu,T Huang,S (2022). Evidence of Infection of Human Embryonic Stem Cells by SARS-CoV-2.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Yang I. Pachankis. 2026. \u201cPlausibility Review on Lipoprotein(a) Infection Path of S2 Autoimmune Pathogen\u201d. Global Journal of Medical Research - C: Microbiology & Pathology GJMR-C Volume 23 (GJMR Volume 23 Issue C3): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.