## I. INTRODUCTION
Acne vulgaris (AV) is a widespread disease that causes damage to the sebaceous glands and associated hair follicles due to several genetic and environmental factors [13]. Epidemiological studies have shown that people between the ages of 12 and 24 are more susceptible to acne, with about $85\%$ of the population suffering from it. Almost $80\%$ of first-degree relatives suffer from acne and the disease occurs earlier and is more severe in people with a positive family history. In $15 - 20\%$ of acne vulgaris patients, the disease is moderate to severe (Bhate K., Williams H.C., 2013; Mahto A. 2017; Janani, S., Sureshkumar, R. A., 2019; Heng AHS, Chew FT. 2020). It has been established that both endogenous and exogenous factors, such as antimicrobial peptides, in particular the expression of cathelicidin (LL-37), defensin-2, cytokines (IL-1 $\alpha$, TNF- $\alpha$, IL-1 $\beta$, IL-8, IL-10, etc.), vitamin D and matrix metalloproteinases, interferon $\gamma$, P. Acnes, St. Aurous, et al. (Dreno B., 2017; Ibrahim AA et all, 2018; Xu H. Li H. 2019; Ebrahimi A et all. 2019; Swelam MM et all. 2019; Ragab M, 2019; Aydingoz IE et al. 2021).
Along with exogenous and endogenous factors, genetic predisposition plays a leading role in the formation of the disease. At present, enough data have been accumulated on the involvement of various candidate genes, the protein products of which are directly or indirectly involved in the regulation of the immune response during inflammation, in the formation of a predisposition to acne. Despite this, the molecular genetic basis of acne production has not been well investigated.
Tumor necrosis factor- $\alpha$ (TNF- $\alpha$ ) is a powerful pro-inflammatory cytokine, a mediator of inflammatory processes plays a central role in the initiation and regulation of the cytokine cascade during the inflammatory and immune response.
The TNF- $\alpha$ gene encoding this cytokine is located on the short arm of chromosome 6 (locus 6p21.1 6p21.3). To date, several functional polymorphic loci have been identified, among which the G-308A variant (replacement of guanine for adenine at nucleotide position -308, international code: rs1800629) is the most common polymorphism. The influence of this locus on the synthesis and level of concentration in the body of the cytokine TNF- $\alpha$ has been proven [Kroeger KM, 1997; Wilson AG 2003].
In studies of hereditary predisposition to various multifactorial diseases, namely the 308A allele and the G/A and A/A genotypes of the rs1800629 polymorphism of the TNF- $\alpha$ gene are considered as a risk factor for the development of various pathological processes associated with an impaired immune response.
The research aims to study the association of rs1800629 polymorphism of the TNF- $\alpha$ gene with the development and clinical course of acne.
## II. MATERIALS AND METHODS
The study was conducted on a sample of 165 acne patients and 161 conditionally healthy donors (control group). The clinical picture of acne vulgaris is characterized by the appearance of comedones, papules, pustules, nodes, cysts on areas of the facial skin (forehead, cheeks, chin), front and back of the back containing sebaceous glands (Tan A.U., Schlosser B.J., Paller A.S. 2018). The degree of activity of the process, the planned method of therapy is determined by the severity of the course of acne. Practical dermatology therefore often uses the classification proposed by the Global Alliance to Improve Acne Outcomes, which distinguishes between mild, moderate, or severe acne (Thiboutot D., Gollnick H., Bettoli V., et al; 2009.).
The analysis of associations was carried out by comparing two samples according to the "case-control" type. The sample "case" was formed from patients with different severity of acne (165 patients). 59 of them suffered from mild, 64 - moderate, and 42 - severe forms of acne. The "control" group included 97 conditionally healthy individuals without any dermatological or other diseases. To isolate DNA from peripheral blood, we used the standard phenol-chloroform extraction method and the RNA/DNA-sorb kit from InterLabService LLC (Russia) (Fig. 1). For genotyping rs1800629 of the TNF- $\alpha$ gene, test systems of NPF "Litekh" (Russia) were used according to the manufacturer's instructions. PCR detection was performed using a GeneAmp PCR-system 2720 thermal cycler (Applied Biosystems, USA).
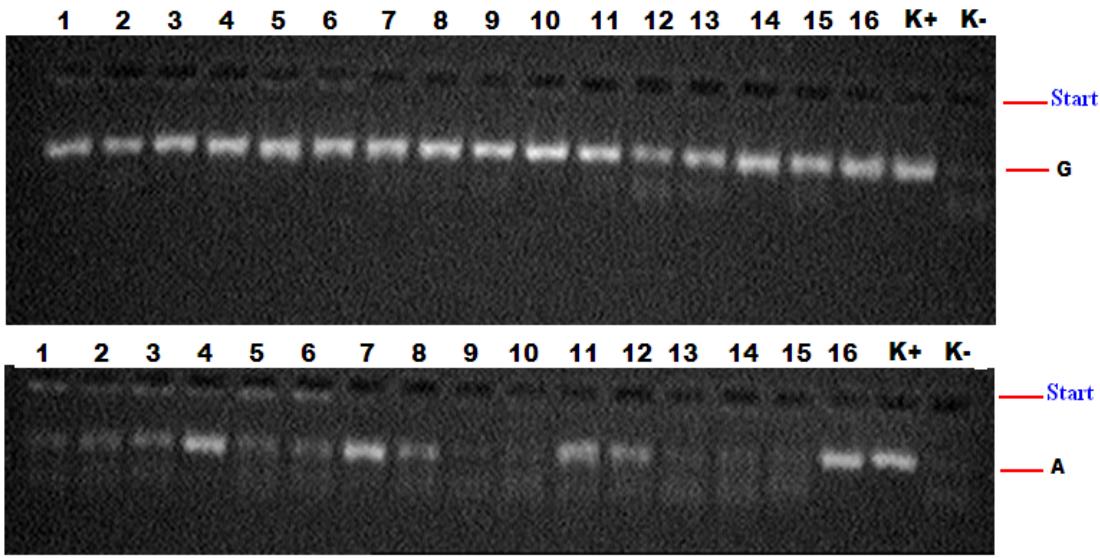 Fig. 1: Electropherogram for TNFα gene polymorphism detection.
K-negative control; $K +$ positive control;
1,2,3,5,6,9,10,13,14,15 - wild genotype G/G;
4,7,8,11,12,16 - heterozygous genotype G/A;
Statistical processing of the obtained data was carried out using the statistical software package OpenEpi 2009, Version 2.3.
## III. RESULTS AND DISCUSSION
The frequency distribution of alleles and genotypes of the rs1800629 polymorphism of the TNF- $\alpha$ gene and their statistical difference among patients and the control sample are shown in Table 1, where statistically significant differences were found between the compared groups. As can be seen from Table 1, the frequency of distribution of G and A alleles in the combined group of patients and the control sample was $79.4\%$ and $20.6\%$ versus $86.6\%$ and $13.3\%$, respectively. Rare allele A $(20.6\%)$ of this polymorphism was found to be statistically significantly more common in the combined patients' group than in the comparison group $(13.3\%)$ with odds ratio OR=1.7 $(\chi^2 = 6.1; p = 0.01; 95\% \text{ CI: } 1.10 - 2.55)$ (Table 2).
The frequency of the wild homozygous genotype G/G was lower in respondents with acne (60.4%), in the control group (73.3%; $\chi^2 = 6.5$; $p = 0.01$;
OR=0.5; CI: 95% 0.342-0.87) and the proportion of carriers of heterozygote G/A was significantly more frequent in patients with AV (38.8%) than in the comparison group (26.7%). According to the odds ratio for carriers of this genotype, the risk of developing acne is significantly increased by 1.7 times ( $\chi^2 = 5.4$; $p = 0.02$; OR=1.7; CI95% 1.08-277). Along with this, no reliable association of the homozygous genotype (A/A) with the formation of acne was found. This genotype was detected only in the group of AV patients (1.2%), while in the control group this genotype was not detected ( $\chi^2 = 2.0$; $p = 0.2$ ).
Table 1: Frequency distribution of alleles and genotypes of the rs1800629 (G308A) polymorphism of the TNF-α gene in the group and subgroups of patients with acne and the healthy control
<table><tr><td rowspan="3">No</td><td rowspan="3">Group</td><td rowspan="3">n*</td><td colspan="4">Allele frequency</td><td colspan="6">Genotype distribution frequency</td></tr><tr><td colspan="2">G</td><td colspan="2">A</td><td colspan="2">G/G</td><td colspan="2">G/A</td><td colspan="2">A/A</td></tr><tr><td>n</td><td>%</td><td>n</td><td>%</td><td>n</td><td>%</td><td>n</td><td>%</td><td>n</td><td>%</td></tr><tr><td>1</td><td>Main group n=165</td><td>330</td><td>262</td><td>79.4</td><td>68</td><td>20.6</td><td>99</td><td>60.0</td><td>64</td><td>38.8</td><td>2</td><td>.2</td></tr><tr><td>a</td><td>Severe form n=42</td><td>84</td><td>61</td><td>72.6</td><td>23</td><td>27.4</td><td>21</td><td>50.0</td><td>19</td><td>45.2</td><td>2</td><td>4.8</td></tr><tr><td>b</td><td>Moderate severity n=64</td><td>128</td><td>97</td><td>75.8</td><td>31</td><td>24.2</td><td>33</td><td>51.6</td><td>31</td><td>48.4</td><td>0</td><td>0.0</td></tr><tr><td>c</td><td>Mild severity n=59</td><td>118</td><td>104</td><td>88.1</td><td>14</td><td>11.9</td><td>45</td><td>76.3</td><td>14</td><td>23.7</td><td>0</td><td>0.0</td></tr><tr><td>2</td><td>Control group n=161</td><td>322</td><td>279</td><td>86.6</td><td>43</td><td>13.3</td><td>118</td><td>73.3</td><td>43</td><td>26.7</td><td>0</td><td>0.0</td></tr></table>
In the next stage of the work, a comparative analysis of the distribution of the frequencies of alleles and genotypes of this locus, depending on the severity of acne, was carried out.
It was found that among patients with severe acne severity, the proportion of carriers of alleles G and A was $72.6\%$ and $27.4\%$, and in the comparison group $86.6\%$ and $13.3\%$, respectively. And the value of the relative risk, equal to 2.4 with the confidence interval CI95% 1.374- 4.35 ( $\chi^2 = 9.6$ and $P = 0.002$ ), also confirms the significant association of the unfavorable allele A in the formation of acne (Table 2). The frequencies G/G, G/A and A/A in the studied subgroup of patients and the control sample were: $50.0\%$, $45.2\%$ and $4.5\%$ versus $73.3\%$ and $26.7\%$, respectively. The heterozygous G/A genotype is a determinant of an increased risk of developing severe acne ( $45.2\%$ versus $26.7\%$, $\chi^2 = 5.4$; $p = 0.02$; OR=2.3; CI: $95\%$ 1.12-4.56), while the favorable A/A genotype, on the contrary, a marker of a reduced risk of developing this form of the disease ( $50.0\%$ versus $73.3\%$, respectively; $\chi^2 = 8.4$; $p = 0.004$; OR=0.4; CI: $95\%$ 0.181- 0.73). The proportion of carriers of the homozygous genotype in the subgroup of patients was $4.5\%$, while in the healthy control this genotype was not detected ( $\chi^2 = 7.7$; $p = 0.005$ ) (Table 3).
Table 2: Differences in the frequency of alleles and genotypes of the rs1800629 polymorphism of the TNF- $\alpha$ gene in the study and control groups
<table><tr><td>Alleles and genotypes</td><td>Main group</td><td>Control group</td><td>x2</td><td>P</td><td>OR</td><td>95% CI</td></tr><tr><td>G</td><td>262</td><td>279</td><td rowspan="2">6.1</td><td rowspan="2">0.01</td><td rowspan="2">1.7</td><td rowspan="2">1.109-2.556</td></tr><tr><td>A</td><td>68</td><td>43</td></tr><tr><td>G/G</td><td>99</td><td>118</td><td>6.5</td><td>0.01</td><td>0.5</td><td>0.3424- 0.8727</td></tr><tr><td>G/A</td><td>64</td><td>43</td><td>5.4</td><td>0.02</td><td>1.7</td><td>1.08- 2.77</td></tr><tr><td>A/A</td><td>2</td><td>0</td><td>2.0</td><td>0.2</td><td>-</td><td>-</td></tr></table>
Table 3: Differences in the frequency of alleles and genotypes of the rs1800629 polymorphism of the TNF-
$\alpha$ gene in the subgroup of patients with mild acne and the control group
<table><tr><td>Alleles and genotypes</td><td>Severe form</td><td>Control group</td><td>x2</td><td>P</td><td>OR</td><td>95% CI</td></tr><tr><td>G</td><td>61</td><td>279</td><td rowspan="2">9.6</td><td rowspan="2">0.002</td><td rowspan="2">2.4</td><td rowspan="2">1.374- 4.35</td></tr><tr><td>A</td><td>23</td><td>43</td></tr><tr><td>G/G</td><td>21</td><td>118</td><td>8.4</td><td>0.004</td><td>0.4</td><td>0.181- 0.73</td></tr><tr><td>G/A</td><td>19</td><td>43</td><td>5.4</td><td>0.02</td><td>2.3</td><td>1.12- 4.56</td></tr><tr><td>A/A</td><td>2</td><td>0</td><td>7.7</td><td>0.005</td><td>-</td><td>-</td></tr></table>
A similar pattern was detected when the allele and genotype frequencies of this locus were compared between a subgroup of patients with average acne severity and a population sample. The frequency of distribution of alleles and genotypes in the studied subgroups of patients with moderate severity of acne was: $G = 75.8\%$ and $A = 24.2\%$, $G / G = 51.6\%$, $G / A = 48.4\%$ and $A / A = 0.0$. The risk of developing moderate acne is significantly higher in carriers of the functionally unfavorable allele A of genotype G/A 2.1 and 2.6 times than in carriers of other genotypes (2=7.9; p=0.005; OR=2.1; Cl: 95% 1.237-3.475 and 2=9.8; p=0.002; OR=2.6; Cl: 95% 1.412-4.706, respectively). At the same time, among patients with moderate severity, the carriage of the wild G/G genotype was significantly less frequent compared to the control group (P < 0.05), i.e., on the contrary, this genotype is likely to have a protective effect on the formation and development of acne (χ2=9.8; p=0.002; OR=0.4; Cl: 95% 0.21-0.70).
Table 4: Differences in the frequency of alleles and genotypes of the rs1800629 polymorphism of the TNF-α gene in the subgroup of patients with moderately severe acne and the control group
<table><tr><td>Alleles and genotypes</td><td>Moderate severity</td><td>Control group</td><td>x2</td><td>P</td><td>OR</td><td>95% CI</td></tr><tr><td>G</td><td>97</td><td>279</td><td rowspan="2">7.9</td><td rowspan="2">0.005</td><td rowspan="2">2.1</td><td rowspan="2">1.237- 3.475</td></tr><tr><td>A</td><td>31</td><td>43</td></tr><tr><td>G/G</td><td>33</td><td>118</td><td>9.8</td><td>0.002</td><td>0.4</td><td>0.21-0.70</td></tr><tr><td>G/A</td><td>31</td><td>43</td><td>9.8</td><td>0.002</td><td>2.6</td><td>1.412- 4.706</td></tr><tr><td>A/A</td><td>0</td><td>0</td><td>-</td><td>-</td><td>-</td><td>-</td></tr></table>
In a comparative analysis, no statistically significant differences were found between the samples of patients with mild acne and controls (Table 5, p>0.05). The frequency distribution of alleles and genotypes of this genetic marker in the studied group was: $G = 88.1\%$ and $A = 23.7\%$, $G / G = 76.3\%$, $G / A = 23.7\%$ and $A / A = 0.0\%$ in the patient subgroup, $G = 86.6\%$ and $A = 13.3\%$, $G / G = 73.3\%$, $G / A = 26.7\%$ and $A / A = 0.0\%$, in the control group. The odds ratios were $OR = 0.9$, $OR = 1.2$ and $OR = 0.8$, respectively.
Table 5: Differences in the frequency of alleles and genotypes of the rs1800629 polymorphism of the TNF-α gene in the subgroup of patients with severe AV.
<table><tr><td>Alleles and genotypes</td><td>Mild severity</td><td>Control group</td><td>x2</td><td>P</td><td>OR</td><td>95% CI</td></tr><tr><td>G</td><td>104</td><td>279</td><td rowspan="2">0.2</td><td rowspan="2">0.7</td><td rowspan="2">0.9</td><td rowspan="2">0.4588- 1.663</td></tr><tr><td>A</td><td>14</td><td>43</td></tr><tr><td>G/G</td><td>45</td><td>118</td><td>0.2</td><td>0.6</td><td>1.2</td><td>0.58-2.345</td></tr><tr><td>G/A</td><td>14</td><td>43</td><td>0.2</td><td>0.6</td><td>0.8</td><td>0.42- 1.709</td></tr><tr><td>A/A</td><td>0</td><td>0</td><td>-</td><td>-</td><td>-</td><td>-</td></tr></table>
A comparison of the frequencies of allelic and genotypic variations of the rs1800629 polymorphism of the TNF- gene in the subgroups of individuals with severe and mild AV demonstrated no significant differences.
Table 5 shows the distribution of the allele and genotype frequencies of the rs1800629 polymorphism of the acne TNF- $\alpha$ gene. In subgroups of patients with severe and mild forms of acne, a statistically significant difference in the frequency of occurrence of alleles and genotypes of this locus was revealed. So, the frequency of G and A alleles in the studied subgroups of patients corresponded to $72.6\%$ and $27.4\%$ versus $88.1\%$ and $11.9\%$. The odds ratio was 2.7 (OR=2.7; 95% CI: 1.34-
5.84) with $\chi 2 = 7.9$; $p = 0.005$, indicating an association between the minor A allele and clinically severe acne.
The G/G, G/A and A/A genotypes occurred at $50.0\%$, $45.2\%$ and $4.8\%$ in the subgroup with severe acne, and $76.3\%$, $23.7\%$ and $0.0\%$ in the subgroup with a mild form of acne respectively. The frequency of the G/G genotype in the subgroup of patients with severe acne was significantly lower than in the subgroup of patients with mild acne ( $50.0\%$ versus $76.3\%$, respectively). The odds ratio was 0.3 (OR=0.3; $\chi^2 = 7.4$; p=0.006; 95% CI: 0.13-0.72), which indicates a protective effect of this genotype against the development of severe acne.
Differences in the frequency of the heterozygous genotype were found between patients with severe and mild acne (45.2% versus 23.7%, respectively). The risk of developing a severe form with this genotype of acne was OR=2.6 ( $\chi^2 = 5.2$; $p = 0.02$; 95% CI: 1.13-6.23). These data also support the adverse effect of this genotype on the development of severe acne. The homozygous genotype A/A was found only in two patients with severe acne (4.8% versus 0.0%, with $\chi^2 = 2.9$; $p = 0.09$ ).
When comparing the frequencies of alleles and genotypes between subgroups of patients with moderate and mild forms of diseases, significant differences were also revealed. The unfavourable allele A predominated in the moderately severe group compared with the mild acne group (24.2% versus 11.9%, respectively; $\chi^2 = 6.3$, $p = 0.01$, RR=2.0; 95% CI: 1.14-3.64, OR=2.4; 95% CI: 1.19-4.73). Heterozygosity for the minor allele was markedly more frequent in patients with moderate acne than in the comparison group (48.4% and 23.7%, respectively; $\chi^2 = 3.84$, P=0.05; OR=0.3; 95% CI: 0.15-0.71). Also, the G/G genotype (51.6% vs 76.3%, respectively; $\chi^2 = 8.1$, P=0.004; 95% CI: 1.2-3.4, OR=3.0; 95% CI: 1.3-6.5) was protective against the formation of moderate acne.
Thus, the data obtained at this stage of the study allow us to conclude that the carriage of the rs1800629 polymorphism of the TNF- $\alpha$ gene is correlated with a more severe course of acne (with severe and moderate variants). At the same time, the heterozygous G/A genotype is the molecular genetic marker of an increased risk of acne formation and development, while the homozygous G/G genotype is a marker of a reduced risk of diseases.
Interestingly, Turkish colleagues (Baz K., et all (2008)) reported no significant association $(P < 0.001)$ between this genotype and acne severity, although there was a significant increase in the frequency of carriers of the unfavorable G/A genotype compared to healthy controls $(P > 0.05)$.
The results of most studies on the contribution of the rs 1800629 polymorphism of the TNF- $\alpha$ gene to the development of acne in different populations are rather inconsistent. For example, two meta-analyses of the associations of the rs1800629 polymorphism of the TNF- $\alpha$ gene with acne risk, performed by Chinese authors, also did not give unambiguous results [Yang JK et all 2014, Wang and Y. 2018]. Nevertheless, in the world literature, the vast majority of case-control design studies confirm a significant association between unfavorable genotypes of the rs1800629 polymorphism of the TNF- $\alpha$ gene with acne formation [Al-Shobaili HA 2012, A. Agodi, 2012 Grech I 2014 Aisha NM 2016, Szabo, K. 2011, Heng AHS et all. 2021]. Our findings for the rs1800629 polymorphism of the TNF- $\alpha$ gene also do not contradict these global studies.
However, along with studies that have identified an association between acne and this polymorphic locus, there are studies that do not support this association. Thus, according to Sobjanek M, et al. (2009), Irawan A. Et all. (2013) and Yu J., et all. (2011) in the Polish, Indonesian and Chinese populations studied, no significant association of this polymorphism with acne formation was found. Perhaps, this is due to the fact that, according to most authors, this polymorphic locus has a high degree of heterogeneity in the frequencies of allelic and genotypic variants depending on the ethnicity or population affiliation of the studied groups [Parra-Rojas I., et al. 2006, Lee Y.H. 2007].
## IV. CONCLUSION
Based on the findings and comparison results, it is possible to conclude that acne development and advancement are connected with the rs1800629 polymorphism of the TNF- gene, depending on the patient's ethnic background. Despite rather contradictory results obtained by researchers, there is no doubt that, at least in most European and Asian populations, unfavourable genotypic variants of the rs1800629 polymorphism of the TNF- $\alpha$ gene make a significant modifying and sometimes direct contribution to the predisposition to form various clinical forms of acne.
Acknowledgements
We thank all the participants in this study.
Author's contributions
Nilufar N. Malikova led the study design, data collection and planning, analysis plan and interpretation of findings and drafted the manuscript. Khamid Y. Karimov and Saidkarim S. Arifov were actively involved in the study conception, design and led the statistical analyses and interpretation. Kodirjon T. Boboev conceived the study, participated in its design, helped draft and revised the manuscript and led the team. The author(s) read and approved the final manuscript.
### Funding
There is no found to support of this study.
#### Declarations
#### Ethics approval and consent to participate
Ethics approval was obtained for the study from the Ethics Committee of Tashkent State Dental Institute. All participants provided written informed consent for the study. All methods were performed in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
#### Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Generating HTML Viewer...
References
30 Cites in Article
J Harper (2020). Acne vulgaris: What's new in our 40th year.
Anthony Wilson,Julian Symons,Tarra Mcdowell,Hugh Mcdevitt,Gordon Duff (1997). Effects of a polymorphism in the human tumor necrosis factor α promoter on transcriptional activation.
Karen Kroeger,Kylie Carville,Lawrence Abraham (1997). The −308 tumor necrosis factor-α promoter polymorphism effects transcription.
K Bhate,H Williams (2013). Epidemiology of acne vulgaris.
S Janani,R Sureshkumar (2019). In Vitro Models for the Evaluation of Anti-acne Technologies.
B Dréno (2017). What is new in the pathophysiology of acne, an overview.
Haoxiang Xu,Huiying Li (2019). Acne, the Skin Microbiome, and Antibiotic Treatment.
Jian-Kang Yang,Wen-Juan Wu,Jue Qi,Li He,Ya-Ping Zhang (2014). TNF -308 G/A Polymorphism and Risk of Acne Vulgaris: A Meta-Analysis.
B Wang,Y-L He (2019). Association of the <i>TNF-α</i> gene promoter polymorphisms at nucleotide -238 and -308 with acne susceptibility: a meta-analysis.
N Aisha,J Haroon,S Hussain,C Tahir,M Ikramullah,H Rahim,N Kishwar,S Younis,M Hassan,Q Javed (2016). Association between tumour necrosis-α gene polymorphisms and acne vulgaris in a Pakistani population.
Kornélia Szabó,Gábor Tax,Dragos Teodorescu-Brinzeu,Andrea Koreck,Lajos Kemény (2011). TNFα gene polymorphisms in the pathogenesis of acne vulgaris.
M Sobjanek,M Zabłotna,B Nedoszytko,M Sokołowska‐wojdyło,A Włodarkiewicz (2009). Lack of association between the promoter polymorphisms at positions –238 and –308 of the tumour necrosis factor alpha gene and acne vulgaris in Polish patients.
A Irawan,I Agusni,M Nasrum,I Yusuf (2013). The immunogenetic analysis of acne vulgaris.
A Agodi,M Barchetta,G Valenti (2012). TNFa‐308G/A GENE POLYMORPHISM IN SUSCEPTIBILITY TO PULMONARY TUBERCULOSIS AMONG INDONESIAN POPULATION.
I Grech,S Giatrakos,G Damoraki (2014). Impact of TNF Haplotypes in the Physical Course of Acne vulgaris.
Al-Shobaili Ha,T Salem,A Alzolibani (2012). Tumor necrosis factor-α -308 G/A and interleukin 10 -1082 A/G gene polymorphisms in patients with acne vulgaris.
I Parra-Rojas,B Ruíz-Madrigal,E Martínez-López,A Panduro (2006). Influence of the −308 TNF-α and −174 IL-6 polymorphisms on lipid profile in Mexican subjects.
Y Lee,J Ji,G Song (2007). Tumor necrosis factoralpha promoter -308 A/G polymorphism and rheumatoid arthritis susceptibility: a meta-analysis.
J Yu,T Yang,L Tian,Y Hu,W Wang (2011). Detect and analyze the TNF-a-308 G/A polymorphism of acne vulgaris in Han Chinese.
A Tan,B Schlosser,A Paller (2018). A review of diagnosis and treatment of acne in adult female patients.
D Thiboutot,H Gollnick,V Bettoli (2009). Global Alliance to Improve Outcomes in Acne. New insights into the management of acne: an update from the Global Alliance to Improve Outcomes in Acne Group.
A Ebrahimi,Z Rahimi,Z Ghadami,E Shakiba,Z Rahimi,M Akbari (2019). Association between CYP19A<G rs700518 Polymorphism with Acne vulgaris and its severity: influence on sex hormones level.
M Swelam,Rah El-Barbary,W Saudi,M Fathi,D Soliman,Abd Elrehem,H (2019). Associations among two vitamin D receptor (VDR) gene polymorphisms (ApaI and TaqI) in acne vulgaris: a pilot susceptibility study.
Ikbal Aydingoz,Gulsen Tukenmez Demirci,Deniz Agirbasli,Devrim Oz‐arslan,Guven Yenmis (2021). The investigation of the amounts and expressions of epidermal growth factor, epidermal growth factor receptor, and epidermal growth factor receptor gene polymorphisms in acne vulgaris.
M Ragab,E Hassan,D Elneily,N Fathallah (2019). Association of interleukin‐6 gene promoter polymorphism with acne vulgaris and its severity.
Adel Ibrahim,Rehab Salem,Ola El‐shimi,Shaymaa Baghdady,Shimaa Hussein (2019). <scp>IL</scp>1A (‐889) gene polymorphism is associated with the effect of diet as a risk factor in Acne Vulgaris.
Anna Heng,Yee-How Say,Yang Sio,Yu Ng,Fook Chew (2021). Gene variants associated with acne vulgaris presentation and severity: a systematic review and meta-analysis.
Anjali Mahto (2017). Acne vulgaris.
Anna Heng,Fook Chew (2020). Systematic review of the epidemiology of acne vulgaris.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Surayyo Z. Yuldasheva. 2026. \u201cPolymorphism of Rs1800629 Gene of TNF-α in the Pathogenesis of Acne Vulgaris\u201d. Global Journal of Medical Research - F: Diseases GJMR-F Volume 23 (GJMR Volume 23 Issue F10): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.