and their composition was investigated using GC-FID and GC/MS. Determination of antioxidant capacity, and urease and xanthine oxidase inhibitions of the methanolic extracts were performed with HPLC-DAD and spectrophotometry. 34 compounds were identified constituting 89.2% of the total essential oil compounds. The major components were determined as T-cadinol (31.1%), γmuurolene (14.4%) and 14-nor-cadin-5-en-4-one isomer A (11.0%) in the oil. Mainly rosmarinic acid, chlorogenic acid, and caffeic acid derivatives were quantified together with apigenin, luteolin and tangeretin derivatives in the extracts by HPLC-DAD. The total phenolics of the extract from leaf and flower parts, 50.81 mg GAE.g-1, was higher than the extract from stem part, and the radical scavenging activity of this extract was also stronger. While, the leaf and flower extract had significant urease and xanthine oxidase inhibitory activities (62.47 and 48.48 µg.mL-1), stem extract had low inhibition on both enzymes.
## I. INTRODUCTION
The genus Nepeta is distributed over a large part of Central and Southern Europe, and West, Central, and Southern Asia as a multi-regional genus of the Lamiaceae (labiatae or mint) family, consisting of approximately 300 taxa (1, 2). 34 species (40 taxa) of the genus Nepeta were recorded in the Flora of Turkey (3, 4). According to the revision of this genus, it is represented by 39 species (46 taxa) in Turkey with recent studies. Endemism rate on the basis of this species is $44\%$ (5). Nepeta species are usually named as catmint or catnip due to the sedative effects on cats and they are commonly used as diuretic, spasmolytic, diaphoretic, bronchodilator, antitussive, anti-asthmatic and sedative agents in Turkey. Due to their antiseptic properties, they are used topically in the treatment of children with skin rashes, and in snake and scorpion bites as well. Nepeta caes area, an endemic species in Turkey, has folkloric uses in southern Anatolia and is used as a herbal tea to treat gastric disorders (6,7). Some Nepeta species have been known their feline attractant activity since they have nepetalactone and its derivatives which are responsible for attractant properties (1,7). Nepetalactones have been reported to effect on insects compare to DEET (N, N-diethyl-m-toluamide) (8-10). Nepetoideae is essential oil-rich genera of the Lamiaceae, therefore has potential economic interest (11).
According to the various studies on the essential oil composition of Nepeta species the essential oil composition depends on the species, place of cultivation, climatic conditions and method of analysis (12-26). The most comprehensive study on 22 Nepeta species was performed by Baser et al. (2000). They were classified into two groups according to composition of essential oil of these species; nepetalactone-containing and nepetalactone-less. Nepetalactone-containing species have $4\alpha \alpha -7\alpha -7\alpha$ nepetalactone as the most frequently contained nepetalactone. Nepeta cadmea Boiss., Nepeta cataria L., Nepeta caesarea Boiss. and Nepeta pilinux P.H. Davis contained $4\alpha \alpha -7\alpha -7\alpha$ nepetalactone while Nepeta racemosa Lam. contained $4\alpha \alpha -7\alpha -7\beta$ nepetalactone as major compound in their oils. Caryophyllene oxide or 1,8-cineole/linalool were identified in the essential oils of Nepetalactone-less species as the major components. As main compounds, $\beta$ -pinene, $\mathbf{a}$ -terpineol, germacrene-D, and spathulenol were respectively determined in the oil of Nepeta phyllochlamys P.H. Davis, Nepeta viscida Boiss., Nepeta sorgerae Hedge et Lamond, and Nepeta trachonitica Post which are out of the two groups (1).
Being a type of the most popular antioxidant secondary metabolites, phenolic compounds were investigated in the Nepeta species as well. Rosmarinic acid, epicatechin, chlorogenic acid, caffeic acids, quercetin, rutin, ellagic acid, thymusin, luteolin, and apigenin which are well-known as antioxidant compounds were found in the extracts of Nepeta cadmea Boiss., Nepeta nuda subsp. albiflora, Nepeta asterotricha Rech., Nepeta rtanjensis Diklic & Milojevic (27-30).
Ureases, a nickel-dependent metalloenzymes, are synthesized by plants, some bacteria, algae and fungi. The jack bean urease (urea amidohydrolase EC 3.3.1.5) catalyzes the hydrolysis of urea to form ammonia and carbon dioxide (31). Helicobacter pylori, a gram-negative microaerophilic pathogen survivable in a limited pH (4.0-8.2) range. This pathogen can successfully colonize and persist in the mucous layer of the human stomach since its urease activity which produces ammonia to reduce stomach acidity. Since antibiotic-resistant strains of H. pylori can emerge against antibiotics, it is believed that plant-derived urease inhibitors would be more beneficial against gastroduodenal disease associated with this pathogen (32, 33).
Xanthine oxidase (EC 1.2.3.2.) produces hydrogen peroxide and superoxide anion, which are reactive oxygen species (ROS) during the oxidation of hypoxanthine and xanthine to uric acid. Under physiological conditions, ROS is kept at a low level by the antioxidant system. Disruption of the balance of ROS and antioxidants due to some diseases causes tissue and DNA damage due to the increase in metabolism of ROS. Inhibition of xanthine oxidase reduces the amount of uric acid and ROS in the bloodstream to prevent both hyperuricemia and oxidation stress (34). Inhibitory effects of some flavonoids such as diosmetin, luteolin, chrysoeriol, apigenin, kaempferol on xanthine oxidase have been reported in various in vitro studies (35-37).
Nepeta pilinux P. H. Davis in Kew Bull. is an endemic species of Nepeta genus growing in the Southwestern Anatolia (Antalya: Alanya) (3). However, in recent flora research, Nepeta pilinux was encountered in Sanliurfa, Birecik district and was recorded as new endemic species for Sanliurfa flora. Nepeta pilinux is named as 'top pisik otu'. In Sanliurfa, the fresh aerial parts of the plant are used to heal mouth sores (38).
There is no knowledge about the essential oil and phenolic composition of Nepeta pilinux endemic species from Šanliurfa. Chemical compositions of the polar and apolar extracts from Nepeta pilinux were determined by HPLC-DAD and GC/MS respectively for the first time in this study. Radical scavenging activities against DPPH and ABTS radicals and enzyme inhibition activities on urease and xanthine oxidase were investigated in vitro to elucidate the bioactivities which may have developed by this species depending on its chemical composition.
## II. EXPERIMENTAL
### a) Materials
Nepeta pilinux was collected from Sanliurfa: Birecik, Kelaynak area in Turkey in 5 May 2018. The voucher specimen has been deposited at the Herbarium in the Recep Tayyip Erdoğan University (RTEUB 6079), Rize, Turkey (Voucher specimen no: FABAK 1702). The plant material was identified by Prof. Dr. Vagif ATAMOV (Recep Tayyip Erdoğan University, Faculty of Science and Literature, Department of Biology, Rize, Turkey). All standards of phenolic compounds were obtained from Sigma-Aldrich (St. Louis, MO, USA) but quercetin from Fluka Chemie GmbH (Switzerland). $\mathrm{Na_2CO_3}$ and $\mathrm{K_2S_2O_8}$ were provided from Sigma-Aldrich (St. Louis, MO, USA). HPLC grade acetonitrile, methanol, acetic acid, Folin ciocalteau, 2,2-Diphenyl-1-picrylhydrazyl (DPPH), and 2,2'-Azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS) were supplied by Merck (Darmstadt, Germany).
### b) Isolation of the Essential Oil
Aerial parts of the plant were water distilled for 3 h using a Clevenger-type apparatus. The essential oil was stored at $4^{\circ}\mathrm{C}$ in the dark until analyzed.
### c) Extraction of Phenolic Compounds
The aerial parts of the plant were divided into two parts. The leaves and flowers were combined in one part and the stems were separated into the other part. The part consisting of leaves and flowers was called NP-LF for short, and the part consisting of stem is called NP-S. These parts were finely ground and 0.5 grams of each were defatted by using $10\mathrm{mL}$ of hexane. The plant residues were dried at $40^{\circ}\mathrm{C}$ for $30\mathrm{min}$ after removing the hexane extract. $20\mathrm{mL}$ methanol were added to these residues for the extraction of phenolic compounds. Extraction was continued overnight at $37^{\circ}\mathrm{C}$ in the dry thermo-shaker cabinet at $352\mathrm{rpm}$, then the extracts were centrifuged at $5000\mathrm{rpm}$ and supernatants transferred into the falcon tube. This procedure was repeated by adding $10\mathrm{mL}$ of methanol in the residue. All extract was concentrated until $5\mathrm{mL}$ by using rotary evaporator at $35^{\circ}\mathrm{C}$. 500 and $1500\mu\mathrm{L}$ of these extracts were stored at $-20^{\circ}\mathrm{C}$ for HPLC-DAD analysis, antioxidant and enzyme inhibition tests. Remaining $3\mathrm{mL}$ of extract was evaporated to calculate the concentration of the extracts. NP-LF and NP-S extracts concentration were 23.17 and $23.50\mathrm{mg.mL}^{-1}$ respectively.
### d) Gas Chromatography (GC) and Gas Chromatography-Mass Spectrometry (GC/MS) analysis of essential oil
The oil was analyzed by capillary GC and GC/MS using an Agilent GC-MSD system.
## i. GC/MS analysis Conditions of Essential Oils
The oil was analyzed by capillary GC/MS using an Agilent GC-MSD system (Agilent Technologies Inc.,
Santa Clara, CA). HP-Innowax FSC column (Hewlett-Packard-HP, U.S.A.) $(60\mathrm{m}\times 0.25\mathrm{mm}$ i.d., with $0.25\mu \mathrm{m}$ film thickness) was used for separation of components in the oil and helium as a carrier gas $(0.8~\mathrm{mL / min})$. The GC oven temperature was kept at $60^{\circ}\mathrm{C}$ for 10 min and programmed to $220^{\circ}\mathrm{C}$ at a rate of $4^{\circ}\mathrm{C / min}$, and kept constant at $220^{\circ}\mathrm{C}$ for 10 min and then programmed to $240^{\circ}\mathrm{C}$ at a rate of $1^{\circ}\mathrm{C / min}$. The split flow was adjusted at $40~\mathrm{mL~min}^{-1}$ with 40:1 split ratio. The injector temperature was set at $250^{\circ}\mathrm{C}$. Mass spectra were taken at $70\mathrm{eV}$ with the mass range $m / z$ 35-450.
## ii. GC Analysis Conditions of Essential Oils
The GC analysis was done with Agilent 6890N GC system fitted with a FID detector set at a temperature of $300^{\circ}\mathrm{C}$. To obtain the same elution order with GC/MS, simultaneous auto-injection was done on a duplicate of the same column applying the same operational conditions. Relative percentage amounts of the separated compounds were calculated from FID chromatogram.
## iii. Identification of Essential Oils
Identification of essential oil components were performed by comparison of their mass spectra with those in the Baser Library of Essential Oil Constituents, Wiley GC/MS Library, Adams Library, MassFinder Library (39-41) and confirmed by comparison of their retention indices. A homologous series of $n$ -alkanes were used as the reference points in calculation of relative retention indices (RRI) (42). The relative percentages of the separated compounds were calculated from FID chromatograms. The analysis results are expressed as mean percentage as listed in Table 1.
### e) HPLC-DAD Analysis Conditions of Methanol Extracts
The chromatographic analyses were performed using a Dionex (Thermo scientific, Germering, Germany) Ultimate 3000 high performance liquid chromatography (HPLC) system equipped with an Ultimate 3000 diode array detector (DAD).
A Thermo acclaim C30 column (150mm. 3mm id. $3\mu \mathrm{m}$ pd) was used with Macherey Nagel (3mm id) guard column. Gradient elution was used with mobil phases; A: $2\%$ acetic acid in water and B: $70\%$ acetonitrile- $30\%$ water. Flow rate was $0.37~\mathrm{mL / min}$ and injection volume was $10~\mu \mathrm{L}$. Column temperature was $25^{\circ}C$. Following 24 phenolic standards were used to calibration and validation of HPLC-DAD analysis method: Gallic acid, protocatechuic acid, $p$ -hydroxy benzoic acid ( $p$ -OH benzoic acid), vanillic acid, catechin, chlorogenic acid, caffeic acid, syringic acid, vanillin, epigallocatechin gallate (EGCG), epicatechin, $p$ -coumaric acid, ferulic acid, chicoric acid, rutin, luteolin-7-glycoside, hesperidin, apigenin-7-glycoside, rosmarinic acid, luteolin, quercetin, hesperetin, apigenin, and tangeretin. They were diluted from their stock solution into nine different concentration at 0.3125; 0.625; 1.25; 5.0; 10.0; 25.0; 40.0 mg.L $^{-1}$ in 1:1 methanol-water solution. External calibration method was used and their regression coefficient were found at least 0.999. Repeatability of the retention time and peak areas were measured as coefficient of variation (CV) which was under 0.93 for retention times and 6.02 for areas of the peaks. Limit of detection and quantification values of the peaks were under 0.11 and $0.37 \mu \mathrm{g.mL}^{-1}$ for all standards. Chromatograms were processed at 254, 280, 315, and 370 nm with DAD which operated 200-400 nm. The identification of the peaks was carried out by comparing the retention times and UV spectra with those of standard phenolic compounds. Some peaks had the same or very similar UV spectra as some standards, but with different retention times. They were defined as derivatives of standards with similar UV spectrum and quantified as equivalent of those standards.
### f) Determination of Total Phenolic Content
Total phenolic content was determined by using the yellow colored Folin-Ciocalteu's phenol reagent, which was reduced to its blue complex in the presence of reducing agent such as phenolic compounds (43). Gallic acid and quercetin were used as phenolic standards to generate standard curves in a range of 0.0156 and $0.500\mathrm{mg/mL}$ at 6 concentration levels $(r^2 = 0.998)$. The optical density of the extracts with phenol reagent in the alkaline solution was measured at $760\mathrm{nm}$ with a UV-Vis detector (Thermo Scientific Multiskan Go, USA). The results were expressed in mg of gallic acid (GAE) and quercetin equivalent (QE) per gram of extracts. All concentration point of the extracts was analyzed in triplicate.
### g) DPPH Free-Radical Scavenging Activity Assay
The free-radical scavenging activity was determined based on the reduction of the purple colored 2,2-diphenyl-1-picrylhydrazyl (DPPH $\bullet$ ) radical to the yellow colored DPPH-H form by the effect of an antioxidant species such as phenolic compounds in the extracts. It was spectrophotometrically performed at 517 nm (44). Briefly, $0.15 \mathrm{~mL}$ of plant extract was mixed with $0.15 \mathrm{~mL} 0.1 \mathrm{mM}$ daily prepared DPPH in methanol and incubated for $30 \mathrm{~min}$ in the dark. Gallic acid and quercetin were used as standards to compare with the methanol extracts. Results are reported as $\mathrm{SC}_{50}$ values, demonstrating the concentration of extract ( $\mu \mathrm{g}$ extract per mL methanol) necessary to scavenge $50\%$ of DPPH $\bullet$. All concentration point of the extracts was analyzed in triplicate.
### h) ABTS Radical Scavenging Assay
the $\mathrm{ABTS}^{\bullet +}$ radical. Obtained radical solution was then diluted with methanol 25 times to obtain an $\mathrm{ABTS}^{\bullet +}$ solution has optical density of $0.700\pm 0.01$ at $734~\mathrm{nm}$ (45). $50~\mu \mathrm{L}$ plant extracts were allowed to react with 250 $\mu \mathrm{L}$ of the $\mathrm{ABTS}^{\bullet +}$ radical solution and the absorbance was measured at $734~\mathrm{nm}$ after 30 min using a spectrophotometer. The $\mathrm{ABTS}^{\bullet +}$ scavenging capacity of the extracts were compared with that of gallic acid and quercetin and reported with $\mathrm{SC}_{50}$ values ( $\mu \mathrm{g}$ extract/ mL methanol). All concentration point of the extracts was analyzed in triplicate.
### i) Urease Inhibitory Assay
Urease inhibition of the extracts were performed according to the phenol-hypochlorite method developed by Weatherburn (1967) (46). Jack bean urease was used as a model enzyme. Optical density of the resulting blue-navy colored mixture at 625 nm were recorded on a spectrophotometer (1601UV-Shimadzu, Australia). To calculate the $\mathrm{IC}_{50}$ values of the polar extracts, different concentrations of the extracts or inhibitory compounds were prepared. Acetohydroxamic acid, well-known inhibitor of urease, were used as positive control.
### j) In Vitro Anti-Xanthine Oxidase Assay
The inhibition of xanthine oxidase was measured by UV spectroscopy technique at $295~\mathrm{nm}$ which is attributed to the released uric acid from xanthine. The inhibitory activity of the extract was determined using a slight modification of the reference methods (34). Briefly, the reaction mixture consisted of $500~\mathrm{mL}$ of the extract solution, (diluted in DMSO), 770 mL of phosphate buffer $(\mathsf{pH}7.8)$ and $70~\mathrm{mL}$ of bovine milk xanthine oxidase $(0.4~\mathrm{U / mL}$, Sigma Aldrich, St. Louis, USA) was prepared. The reaction was initiated by the addition $660~\mathrm{mL}$ of xanthine solution $(0.4~\mathrm{mM})$ into the mixture after incubation at $25^{\circ}\mathrm{C}$ for $15\mathrm{min}$. The assay mixture was incubated at $25^{\circ}\mathrm{C}$ for $15\mathrm{min}$ again. The reaction was stopped by adding $200~\mathrm{mL}$ of $0.5\mathrm{N}$ HCl and the absorbance was measured at $295~\mathrm{nm}$ using UV/vis spectrophotometer (1601UVShimadzu, Australia). A well-known XO inhibitor (XOI), allopurinol (Sigma Aldrich, St. Louis, USA) was used as a positive control. XO activity was expressed as percent inhibition of xanthine oxidase, calculate as $(1 - \mathrm{B / A})\times 100$ where A is the change in absorbance of the assay without the test samples. ( $\Delta$ abs with enzyme - $\Delta$ abs without enzyme), and B is the change in absorbance of the assay with the test sample ( $\Delta$ abs with enzyme - $\Delta$ abs without enzyme). The assay was done in triplicate. The $\mathrm{IC}_{50}$ value was determined as the concentration of the extract that gave $50\%$ inhibition of maximal activity.
## III. RESULTS AND DISCUSSION
### a) GC And GC/MS Analysis of Essential Oils
Essential oil yield in the sample was calculated as $0.26\%$. Thirty-four compounds comprising about $89.2\%$ of the essential oil were identified. Identified essential oil components were compared with literature polar column retention times. The major components were determined as T-cadinol $(31.1\%)$, $\gamma$ -murolene $(14.4\%)$ and 14-nor-cadin-5-en-4-one isomer A $(11.0\%)$ in the oil. Nepeta pilinux essential oil has oxygenated sesquiterpenes $(44.6\%)$, sesquiterpenes hydrocarbons $(17.3\%)$, oxygenated monoterpenes $(10.1\%)$, monoterpene hydrocarbons $(2.3\%)$ and others $(14.9\%)$. The analysis results were shown in Table 1.
Our results were not similar to the classification of Nepeta species reported by Baser et al. (2000) (1). Nepeta pilinux which was collected from Antalya, Alanya district was in the group containing nepetalactone, an iridoid monoterpene according to their report. Although $89.2\%$ of the essential oil was determined in Nepeta pilinux from Sanliurfa, nepetalactone was not detected. The determination of different essential oil compositions from Nepeta pilinux from different regions may have resulted from the difference in locality.
The major constituent of water-distilled essential oils of Nepeta heliotropifolia and Nepeta congesta subsp. cryptantha was determined by GC/MS and GC-FID and found to be germacrene D (36.7% and 38.5%, respectively). Their main aroma component was determined as eucalyptol (48.0% and 24.7%, respectively) (47). Although the major essential oil and aroma compounds of these two species were quite different from the main essential oils of Nepeta pilinux, the compositions of these three species had lots of common compounds such as $\alpha$ and $\beta$ -pinene, $\gamma$ -murolene, myrtenal, pinocarveol, caryophyllene oxide, cubenol, T and $\alpha$ -cadinol ect. The presence of more or less components may be due to differences between species, as well as environmental conditions and the harvesting time of the plant.
Table 1: Composition of the Essential Oil of Nepeta Pilinux
<table><tr><td>RRIa</td><td>RRIb</td><td>Components</td><td>%</td><td>IM</td></tr><tr><td>1032</td><td>1032c</td><td>α-Pinene</td><td>2.3</td><td>tR, MS</td></tr><tr><td>1118</td><td>1118c</td><td>β-Pinene</td><td>tr</td><td>tR, MS</td></tr><tr><td>1213</td><td>1213d</td><td>1,8-Cineole</td><td>tr</td><td>tR, MS</td></tr><tr><td>1376</td><td></td><td>trans-Muurola-3,5-diene</td><td>0.3</td><td>MS</td></tr><tr><td>1499</td><td>1499c</td><td>α-Campholenal</td><td>0.7</td><td>MS</td></tr><tr><td>1535</td><td>1535e</td><td>Pinocamphone</td><td>0.1</td><td>MS</td></tr><tr><td>1553</td><td>\(1553^d \)</td><td>Linalool</td><td>2.3</td><td>\(t_R, MS \)</td></tr><tr><td>1577</td><td>\(1577^{e,g} \)</td><td>α-Cedrene</td><td>0.3</td><td>MS</td></tr><tr><td>1586</td><td>\(1586^c \)</td><td>Pinocarvone</td><td>0.7</td><td>MS</td></tr><tr><td>1617</td><td>\(1613^g \)</td><td>β-Cedrene</td><td>tr</td><td>MS</td></tr><tr><td>1648</td><td>\(1648^e \)</td><td>Myrtenal</td><td>0.6</td><td>MS</td></tr><tr><td>1670</td><td>\(1670^d \)</td><td>trans-Pinocarveol</td><td>1.3</td><td>\(t_R, MS \)</td></tr><tr><td>1684</td><td>\(1684^e \)</td><td>trans-Verbenol</td><td>4.4</td><td>MS</td></tr><tr><td>1694</td><td>\(1693^g \)</td><td>β-Acoradiene</td><td>tr</td><td>MS</td></tr><tr><td>1704</td><td>\(1704^{c,d} \)</td><td>γ-Muurolene</td><td>14.4</td><td>MS</td></tr><tr><td>1706</td><td>\(1706^d \)</td><td>α-Terpineol</td><td>tr</td><td>\(t_R, MS \)</td></tr><tr><td>1726</td><td>\(1725^e \)</td><td>Verbenone</td><td>tr</td><td>\(t_R, MS \)</td></tr><tr><td>1747</td><td>\(1740^e \)</td><td>p-Mentha-1,5-dien-8-ol</td><td>tr</td><td>MS</td></tr><tr><td>1751</td><td>\(1751^d \)</td><td>Carvone</td><td>tr</td><td>\(t_R, MS \)</td></tr><tr><td>1797</td><td>\(1804^d \)</td><td>Myrtenol</td><td>tr</td><td>MS</td></tr><tr><td>1845</td><td>\(1845^d \)</td><td>trans-Carveol</td><td>tr</td><td>\(t_R, MS \)</td></tr><tr><td>1853</td><td>\(1849^d \)</td><td>cis-Calamene</td><td>1.8</td><td>MS</td></tr><tr><td>2008</td><td>\(2008^c \)</td><td>Caryophyllene oxide</td><td>0.6</td><td>\(t_R, MS \)</td></tr><tr><td>2050</td><td>\(2050^e \)</td><td>(E)-Nerolidol</td><td>0.4</td><td>\(t_R, MS \)</td></tr><tr><td>2080</td><td>\(2080^d \)</td><td>Cubenol</td><td>3.7</td><td>MS</td></tr><tr><td>2089</td><td></td><td>6-Methyl-5 (3-methyl phenyl)-2-heptanone</td><td>0.8</td><td>MS</td></tr><tr><td>2187</td><td>\(2187^d \)</td><td>T-Cadinol</td><td>31.3</td><td>MS</td></tr><tr><td>2256</td><td></td><td>epi-α-Bisabolol</td><td>0.3</td><td>\(t_R, MS \)</td></tr><tr><td>2257</td><td>\(2233^f \) \(2256^e \)</td><td>Cadalene</td><td>0.5</td><td>MS</td></tr><tr><td>2258</td><td>\(2219^c \) \(2255^d \)</td><td>α-Cadinol</td><td>1.4</td><td>\(t_R, MS \)</td></tr><tr><td>2264</td><td>\(2264^e \)</td><td>4,7-dimethyl-1-tetralone</td><td>0.8</td><td>MS</td></tr><tr><td>2320</td><td>\(2324^c \)</td><td>14-Nor-cadin-5-en-4-one isomer A</td><td>11.0</td><td>MS</td></tr><tr><td>2349</td><td>\(2349^e \)</td><td>Cadina-4, 10 (15)-dien-3-one</td><td>6.1</td><td>MS</td></tr><tr><td>2931</td><td>\(2931^c \) \(2913^f \)</td><td>Hexadecanoic acid</td><td>3.1</td><td>MS</td></tr><tr><td></td><td></td><td></td><td></td><td></td></tr><tr><td></td><td></td><td>Monoterpene hydrocarbons</td><td>2.3</td><td></td></tr><tr><td></td><td></td><td>Oxygenated monoterpenes</td><td>10.1</td><td></td></tr><tr><td></td><td></td><td>Sesquiterpenes hydrocarbons</td><td>17.3</td><td></td></tr><tr><td></td><td></td><td>Oxygenated sesquiterpenes</td><td>44.6</td><td></td></tr><tr><td></td><td></td><td>Others</td><td>14.9</td><td></td></tr><tr><td></td><td></td><td>Total%</td><td>89.2</td><td></td></tr></table>
$\mathsf{RRI}^{\mathrm{a}}$: RRI Relative retention indices experimentally calculated against $n$ -alkanes; $\mathsf{RRI}^{\mathrm{b}}$: RRI from literature [c (48); d (49); e (50); f (51); g (52)] for polar column values;% calculated from FID data; tr; Trace (\<0.1%); Identification Method (IM): tR, Identification based on comparison with co-injected with standards on a HP Innowax column; MS, identified on the basis of computer matching of the mass spectra with those of the libraries.
### b) HPLC-DAD Analysis of Phenolic Compounds in the Methanol Extracts
The list of standard phenolic compounds has a wide range of phenolic standards such as 5 benzoic acids (gallic, protocatechuic, $p$ -OH benzoic, vanillic, and syringic acid), 1 hydroxybenzaldehyde (vanillin), 5 cinnamic acids (caffeic, $p$ -coumaric, ferulic, chicoric and chlorogenic acid), 3 flavanols (catechin EGCG, epicatechin), 1 flavonol with its 1 glycoside (quercetin and rutin), 3 flavones with 2 sugar attached derivatives, (luteolin, apigenin, tangeretin, luteolin-7-glucoside, apigenin-7-glucoside), 1 flavanone with its 1 glycoside (hesperidin and hesperetin), and rosmarinic acid which is a caffeic acid ester (supp. Table 1).
A total $6.9\mathrm{g}$ and $2.1\mathrm{g}$ of phenolic compounds per $100\mathrm{g}$ of NP-LF and NP-S, respectively, were quantified by HPLC-DAD (Table 2). The stem part of the plant had at least 3 times less amount of phenolic compounds than the leaf and flower parts (NP-LF). Rosmarinic acid (RA) was found to be the major compound at $3.1\mathrm{g}$ per $100\mathrm{g}$ of NP-LF $(3.1\%)$ as expected from Lamiaceae family members. The peaks which had same UV spectrum as RA but eluted earlier were identified as RA derivatives (der.) and quantified as equivalent of RA (Fig. 1). They could be the sugar or other functional groups attached to the RA causing this early elution. Total ratio of RA derivatives in the extracts were $4.3\%$ for NP-LF and $0.3\%$ for NP-S (Table 2). Caffeic acid and derivatives including RA covered $4.5\%$ of the NP-LF.
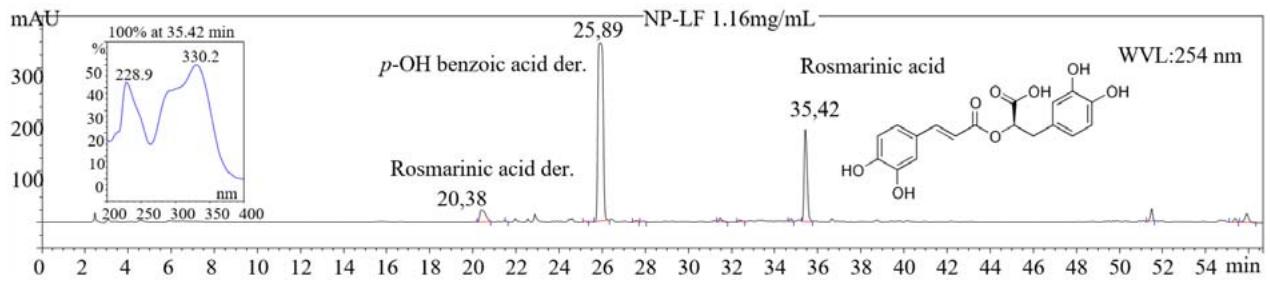 Figure 1: HPLC-DAD chromatogram of the extract from leaf and flower parts of Nepeta pilinux
Protocatechuic, chlorogenic, caffeic, $\rho$ -coumaric, chicoric acids and luteolin-7-glycoside were also determined in the extracts by comparing the retention time and UV spectrum of the peaks with those of the standard phenolics. There was extremely large peak among others in the chromatograms at 254 nm which was eluted at around 25.9 min and had overlaying two compounds with 243 and 257 nm maximum wavelengths. These peaks were identified as $p$ -OH benzoic acid der. and quantified as its equivalent (Fig. 1 and Table 2). The peak eluting at 21.4 min. and having max. absorbance at $293~\mathrm{nm}$ was identified as $\rho$ -OH benzaldehyde (4-hydroxybenzaldehyde) since its elution order and UV spectrum were consistent with this phenolic aldehyde (53). The regression equation of vanillin which is the 4-hydroxy-3-methoxybenzaldehyde was used to quantified $\rho$ -OH-benzaldehyde due to their structural and spectral similarity. Four tangeretin derivatives were identified tentatively by comparing UV spectrum of these peaks with that of tangeretin. Since tangeretin has 5 metoxy groups without hydroxyl group, it is eluting in the last part of the chromatogram from
Table 2: The amount and spectral details of phenolic compounds determined in the extracts by HPLC-DAD
<table><tr><td rowspan="2"></td><td colspan="2">RT</td><td colspan="2">Maximum Wavelength</td><td colspan="2">mg/100g extract</td></tr><tr><td>NP-LF</td><td>NP-S</td><td>A max</td><td>Compounds</td><td>NP-LF</td><td>NP-S</td></tr><tr><td>1</td><td>ND</td><td>8.6</td><td>259-292</td><td>protocatechuic acid</td><td>ND</td><td>5.5</td></tr><tr><td>2</td><td>15.8</td><td>15.3</td><td>232-284</td><td>catechin der.</td><td>83.8</td><td>57.7</td></tr><tr><td>3</td><td>20.4</td><td>20.3</td><td>330-300sh-232</td><td>rosmarinic acid der.</td><td>1055.9</td><td>164.9</td></tr><tr><td>4</td><td>21.4</td><td>21.2</td><td>293</td><td>p-OH benzaldehyde</td><td>2.8</td><td>16.2</td></tr><tr><td>6</td><td>22.0</td><td>21.7</td><td>326-295sh-237</td><td>chlorogenic acid</td><td>86.9</td><td>6.7</td></tr><tr><td>7</td><td>22.9</td><td>ND</td><td>328-295-242</td><td>caffeic acid</td><td>75.9</td><td>ND</td></tr><tr><td>8</td><td>ND</td><td>22.9</td><td>315-230</td><td>p-coumaric acid der.</td><td>ND</td><td>14.2</td></tr><tr><td>9</td><td>23.7</td><td>ND</td><td>333-243</td><td>chicoric acid der.</td><td>2.8</td><td>ND</td></tr><tr><td>10</td><td>ND</td><td>23.9</td><td>281-229</td><td>syringic acid der.</td><td>ND</td><td>5.4</td></tr><tr><td>11</td><td>24.5</td><td>24.3</td><td>329-302sh-249</td><td>rosmarinic acid der.</td><td>84.3</td><td>31.9</td></tr><tr><td>12</td><td>25.2</td><td>25.0</td><td>330-300sh-243</td><td>rosmarinic acid der.</td><td>29.0</td><td>6.7</td></tr><tr><td>13</td><td>25.9</td><td>25.7</td><td>243 and 257 mix</td><td>p-OH benzoic acid equivalent</td><td>2204.7</td><td>1605.3</td></tr><tr><td>14</td><td>26.8</td><td>26.6</td><td>229-310</td><td>p-coumaric acid</td><td>3.7</td><td>2.4</td></tr><tr><td>15</td><td>27.1</td><td>26.8</td><td>352-255</td><td>luteolin-7-glycoside der.</td><td>12.7</td><td>5.7</td></tr><tr><td>16</td><td>28.9</td><td>ND</td><td>312-295-228</td><td>p-coumaric acid der.</td><td>11.6</td><td>ND</td></tr><tr><td>17</td><td>29.2</td><td>29.0</td><td>329-305sh-243</td><td>chicoric acid</td><td>3.7</td><td>2.1</td></tr><tr><td>18</td><td>ND</td><td>30.8</td><td>329-233</td><td>rosmarinic acid der.</td><td>ND</td><td>3.7</td></tr><tr><td>19</td><td>31.5</td><td>31.2</td><td>338-253-225</td><td>apigenin-glycoside der.</td><td>54.4</td><td>3.9</td></tr><tr><td>20</td><td>ND</td><td>31.4</td><td>342-246-226</td><td>luteolin-glycoside der.</td><td>ND</td><td>2.5</td></tr><tr><td>21</td><td>32.4</td><td>ND</td><td>327-228 and 347-270</td><td>ferulic acid der. and luteolin-7-glycoside</td><td>mix</td><td>ND</td></tr><tr><td>22</td><td>ND</td><td>32.4</td><td>320-244</td><td>caffeic acid der.</td><td>ND</td><td>1.2</td></tr><tr><td>23</td><td>33.2</td><td>33.0</td><td>228-278</td><td>syringic acid der.</td><td>31.4</td><td>20.1</td></tr><tr><td>24</td><td>33.4</td><td>33.2</td><td>228-278</td><td>syringic acid der.</td><td>17.8</td><td>17.3</td></tr><tr><td>25</td><td>ND</td><td>33.4</td><td>331-275</td><td>apigenin-glycoside der.</td><td>ND</td><td>3.2</td></tr><tr><td>26</td><td>ND</td><td>34.1</td><td>338-275</td><td>apigenin-glycoside der.</td><td>ND</td><td>5.6</td></tr><tr><td>27</td><td>35.4</td><td>35.3</td><td>330-290sh-229</td><td>rosmarinic acid</td><td>3099.7</td><td>59.1</td></tr><tr><td>28</td><td>ND</td><td>35.7</td><td>314-230</td><td>p-coumaric acid der.</td><td>ND</td><td>2.7</td></tr><tr><td>29</td><td>36.6</td><td>ND</td><td>328-300sh-234</td><td>caffeic acid der.</td><td>21.6</td><td>ND</td></tr><tr><td>30</td><td>ND</td><td>38.3</td><td>319-230-295sh</td><td>ferulic acid der.</td><td>ND</td><td>6.8</td></tr><tr><td>31</td><td>38.7</td><td>38.6</td><td>328-277-226</td><td>tangeretine der.</td><td>10.7</td><td>43.6</td></tr><tr><td>32</td><td>39.4</td><td>39.2</td><td>329-277</td><td>chicoric acid der.</td><td>6.3</td><td>1.6</td></tr><tr><td>33</td><td>ND</td><td>39.4</td><td>338-276</td><td>apigenin der.</td><td>ND</td><td>5.6</td></tr><tr><td>34</td><td>39.8</td><td>ND</td><td>327-300sh-239</td><td>chlorogenic acid der.</td><td>8.4</td><td>ND</td></tr><tr><td>35</td><td>ND</td><td>41.6</td><td>267-340</td><td>apigenin der.</td><td>ND</td><td>6.4</td></tr><tr><td>36</td><td>ND</td><td>42.0</td><td>324-270</td><td>tangeretin der.</td><td>ND</td><td>1.6</td></tr><tr><td>38</td><td>ND</td><td>45.1</td><td>328-277-214</td><td>tangeretin der.</td><td>ND</td><td>1.4</td></tr><tr><td>39</td><td>ND</td><td>49.8</td><td>327-275-223</td><td>tangeretin der.</td><td>ND</td><td>7.4</td></tr><tr><td>40</td><td>ND</td><td>50.3</td><td>316</td><td>p-coumaric acid der.</td><td>ND</td><td>16.2</td></tr><tr><td>41</td><td>50.4</td><td>50.6</td><td>315-214</td><td>p-coumaric acid der.</td><td>11.5</td><td>0.9</td></tr><tr><td></td><td></td><td></td><td></td><td>total</td><td>6919.7</td><td>2135.2</td></tr><tr><td></td><td></td><td></td><td></td><td>total rosmarinic acids</td><td>4269.0</td><td>266.3</td></tr></table>
Since Nepeta pilinux is an endemic species and newly recorded in Sanliurfa flora, there is not any scientific report for its phenoreverse phase chromatography (Table 2). Tangeretin derivatives may differ from each other in the number of hydroxyl and methoxyl groups and the presence of sugar or other functional groups. Thymusin which is the $5,6,4^{\prime}$ -Trihydroxy-7,8-dimethoxyflavone $(5,6,4^{\prime})$ demethyltangeretin) and isothymusin were determined in Nepeta asterotricha Rech. (29). Therefore, two of the tangeretin der. could be thymusin and isothymusin. Apigenin and luteolin derivatives, determined in the extracts based on their UV spectra, were identified and quantified as apigenin, apigenin glycoside der. or luteolin glycoside der. based on their elution orders.
lic composition. On the other hand, there are some reports for chemical composition of the other species of Nepeta genus to compare with. For instance, epicatechin, caffeic acids, chlorogenic acid, quercetin and ellagic acid, well-known as antioxidant compounds were found in the ethanol extract of Nepeta cadmea Boiss. by HPLC-DAD (27). They couldn't have determined RA because it was not in their standard compound list. Unlike their results, epicatechin, quercetin and ellagic acid were not detected in Nepeta pilinux extracts (Table 2). Observation of similar but different phenolic compounds in species shows that differences between species also cause differences in metabolic product synthesis. In addition, different derivatives of phenolic compounds can be observed in samples of the same species grown in different geographical conditions. RA, apigenin, and quercetin were determined as major compounds in the ethanol extract of Nepeta nuda subsp. albiflora (28). Isolation of some iridoid glycosides such as nepetamoside, nepetaracemoside B, nepetonic acid and some polyphenol and flavonoid components such as RA and its methyle ester, thymusin, luteolin and apigenin from Nepeta asterotricha Rech. were performed by Goldansaz et al. (2019) (29). Methanolic extracts from Nepeta rtanjensis Diklic & Milojevic which is an endemic perennial plant, in a very limited area in Southeast Serbia were investigated in a scientific study (30). The presence of high levels of chlorogenic acid, RA and rutin in these extracts was thought to be the reason for their antigenotoxicity.
RA, which is the main component in these extracts, is synthesized by the phenylpropanoid pathway starting with L-phenylalanine and L-tyrosine. From L-phenylalanine, t-cinnamic acid, p-coumaric acid and p-coumaroyl-CoA are successively produced, while p-hydroxyphenylpyruvic acid, p-hydroxyphenyllactic acid are successively produced from L-tyrosine. Then, the production of p-coumaroyl-p-hydroxy-phenyllactic acid by condensation of p-coumaroyl-CoA and p-hydroxyphenyllactic acid is followed by RA synthesis (54). Chlorogenic acid is synthesized by the condensation of p-coumaroyl-CoA with quinic acid (55). These metabolites which were found in the Nepeta pilinux extracts, support the survival of plants against harsh environmental conditions, therefore they have been seen by humans as a remedy for various diseases for many years. It has been reported that RA, chlorogenic acid and its metabolite caffeic acid have a neuroprotective effect due to their antioxidant capacity (56, 57).
### c) Total Antioxidant Characterization of the Methanol Extracts
Total phenolic content (TPC) and radical scavenging activities (RSA) against DPPH and ABTS radical were evaluated to characterize the antioxidant capacity of the extracts and results were presented in Table 3. TPC of the NP-LF and NP-S extract from Nepeta pilinux was 50.81 and $13.37\mathrm{mg}$ GAE/g respectively. Consistent with the TPC of NP-LF which was found around fourfold of NP-S, RSA of NP-LF were higher (433.18 and $82.29\mu \mathrm{g} / \mathrm{mL}$ for DPPH and ABTS respectively) than NP-S as expected (Table 3). The antioxidant capacity of this species was found to be quite consistent comparing with other species in the literature reports. Antioxidant activity of methanolic extracts from flower and leaf parts of Nepeta rtanjensis were reported by Bosnjak-Neumuller et al. (2017). They have found that the leaf extract had higher antioxidant capacity with TPC of $62.73\mathrm{mg}$ of GAE/g and $\mathrm{IC}_{50}$ value of RSA against DPPH as $112.59\mu \mathrm{g / mL}$ than those of flower extract. The antioxidant activities of ethanol, methanol, acetone, and water extracts from Nepeta cadmea were presented by Kaska et al. (2018). They have found that the water extract had the highest RSA $(\mathrm{IC}_{50}$ value of DPPH as $25.54\mu \mathrm{g / mL}$ and ABTS, 14.51 $\mu \mathrm{g / mL})$ in these four extracts. Highest TPC with 79.84 mg GAE/g, was found in the methanol extract while the highest total flavonoids with $77.09\mathrm{mgQE / g}$ was in the acetone extract. It was reported by Teber and Bursal (2020) that ethanol and water extracts of Nepeta nuda subsp. albiflora had strong antioxidant effects with $\mathrm{IC}_{50}$ values of DPPH as 54.4 and $113.0~\mu \mathrm{g / mL}$, respectively. The TPC of flowers, leaves and roots methanol extracts of Nepeta humulis were found as 123.18, 66.20 and 54.77 mg GAE/g extract, respectively. Flower extract which had the highest TPC displayed best RCA with $\mathrm{IC}_{50}$ of 1290 and $350~\mu \mathrm{g.mL}^{-1}$ against DPPH and ABTS respectively (58).
Table 3: Total phenolic content, radical scavenging activity and enzyme inhibition of the extracts
<table><tr><td rowspan="2">Samples and standards</td><td colspan="2">TPCa</td><td colspan="2">RSAb</td><td colspan="2">Enzyme inhibition</td></tr><tr><td>mgGAE/g</td><td>mgQE/g</td><td>DPPH SC50</td><td>ABTS SC50</td><td>urease IC50</td><td>XO IC50</td></tr><tr><td>NP-LF</td><td>50.81±1.50</td><td>35.40±1.06</td><td>433.18±12.74</td><td>82.29±0.98</td><td>62.47±0.10</td><td>48.48±0.10</td></tr><tr><td>NP-S</td><td>13.37±0.34</td><td>9.03±0.24</td><td>523.49±5.82</td><td>381.58±5.15</td><td>230.59±0.23</td><td>222.67±0.13</td></tr><tr><td>Gallic Acid</td><td></td><td></td><td>1.52±0.06</td><td>3.29±0.09</td><td></td><td></td></tr><tr><td>Quercetin</td><td></td><td></td><td>5.87±0.13</td><td>8.52±0.16</td><td></td><td></td></tr><tr><td>Acetohydroxamic acid</td><td></td><td></td><td></td><td></td><td>24.56±0.29</td><td></td></tr><tr><td>Allopurinol</td><td></td><td></td><td></td><td></td><td></td><td>0.54±0.04</td></tr></table>
GAE, Gallic acid equivalent; QE, quercetin equivalent; $\mathrm{SC}_{50}$, value of the concentration of extract required to scavenge $50\%$ of DPPH and ABTS radicals ( $\mu$ g extract per mL methanol); $\mathrm{IC}_{50}$, value of the concentration of extract required to inhibit $50\%$ of Jack bean urease and bovine milk xanthine oxidase enzymes ( $\mu$ g extract per mL methanol). aTotal phenolic contents are expressed in mg GAE/g extract and mg QE/g extract. Radical Scavenging Activity
### d) Urease and Xanthine oxidase inhibitions of the methanol extracts
The urease enzyme inhibition of the NP-LF with $\mathrm{IC}_{50}$ value of $62.47\mu \mathrm{g.mL}^{-1}$ was only three times lower than the inhibition of acetohydroxamic acid (24.56 $\mu \mathrm{g.mL}^{-1}$ ) which is standard medicine (Table 3). NP-S had low inhibition against this enzyme with $\mathrm{IC}_{50}$ value of $230.59\mu \mathrm{g.mL}^{-1}$. The xanthine oxidase inhibition of the NP-LF and NP-S extracts, 48.48 and $222.67\mu \mathrm{g.mL}^{-1}$ respectively, were quite lower comparing to the inhibition of allopurinol $(0.54\mu \mathrm{g.mL}^{-1})$ which is standard medicine reducing the production of uric acid in the body caused by certain cancer medications and kidney stones. Akdeniz et al. (2020) were screened urease inhibition effect of the essential oils and ethanolic extracts of Nepeta heliotropifolia and Nepeta congesta subsp. cryptantha comparing with the standard thiourea. They reported that none of them exhibited urease inhibitory activity (47). In another study, ethyl acetate sub fraction of Nepeta praetervisa showed significant urease inhibitory activity (68%) (59). The structure-activity relationship revealed that the planar flavones and flavonols with a 7-hydroxyl group such as chrysin, luteolin, kaempferol, quercetin, myricetin, and isorhamnetin inhibited xanthine oxidase activity at low concentrations, while the nonplanar flavonoids, isoflavones and anthocyanidins were less inhibitory (35). Although the inhibitory effects of the extracts cannot compete with acetohydroxamic acid and allopurinol, the fact that they are a natural herbal inhibitor source shows that these extracts are more suitable for use.
## IV. CONCLUSION
This was the first investigation on the chemical composition and bioactivities of the Nepeta pilinux. The polar and apolar extracts of aerial parts had high amount of phenolic and essential oil compounds and demonstrated the potential antioxidant capacities. The polar extract with $3.1\%$ rosmarinic acid had urease and xanthine oxidase inhibition as well. This comprehensive evaluation of Nepeta pilinux revealed that this endemic plant could be the source of valuable therapeutic compounds. Besides, this report would be the incentive for further works on this plant's metabolites.
### ACKNOWLEDGMENTS
This study was supported by the scientific research projects unit of Recep Tayyip Erdogan University with the project numbered FDK-2017-777.
#### Disclosure Statement
No potential conflict of interest was reported by the authors
[^7]: mM of ABTS solution and 2.4 mM of potassium persulfate solution were mixed in equal quantities and allowing to oxidation reaction of ABTS by $\mathrm{K}_2\mathrm{S}_2\mathrm{O}_8$ for $18\mathrm{~h}$ at room temperature in the dark to form _(p.3)_
Generating HTML Viewer...
References
59 Cites in Article
K Baser,N Kirimer,M Kurkcuoglu,B Demirci (2000). Essential Oils of Nepeta Species Growing in Turkey.
A Kaya,T Dirmenci (2008). Nutlet surface micromorphology of the genus Nepeta L. (Lamiaceae) in Turkey.
I Hedge,I,J Lamond (1982). Flora of Turkey and the East Aegean Islands.
A Guner,N Ozhatay,T Ekim,K Baser (2000). Flora of Turkey and the East Aegean Islands.
F Celep,T Dirmenci (2017). Systematic and biogeographic overview of Lamiaceae in Turkey.
Carmen Formisano,Daniela Rigano,Felice Senatore (2011). Chemical Constituents and Biological Activities of <i>Nepeta</i> Species.
Benjamin Lichman,Grant Godden,John Hamilton,Lira Palmer,Mohamed Kamileen,Dongyan Zhao,Brieanne Vaillancourt,Joshua Wood,Miao Sun,Taliesin Kinser,Laura Henry,Carlos Rodriguez-Lopez,Natalia Dudareva,Douglas Soltis,Pamela Soltis,C Buell,Sarah O’connor (2020). The evolutionary origins of the cat attractant nepetalactone in catnip.
Ulrich Bernier,Kay Furman,Daniel Kline,Sandra Allan,Donald Barnard (2005). Comparison of Contact and Spatial Repellency of Catnip Oil and<i>N</i>,<i>N</i>-Diethyl-3-methylbenzamide (Deet) Against Mosquitoes.
Gretchen Schultz,Erica Simbro,Jason Belden,Junwei Zhu,Joel Coats (2004). Catnip, <I>Nepeta cataria</I> (Lamiales: Lamiaceae)—A Closer Look: Seasonal Occurrence of Nepetalactone Isomers and Comparative Repellency of Three Terpenoids to Insects.
W Reichert,J Ejercito,T Guda,X Dong,Q Wu,A Ray,J Simon (2019). Repellency Assessment of Nepeta cataria essential oils and ısolated nepetalactones on Aedes aegypti.
D Rigano,N Arnold,F Conforti,F Menichini,C Formisano,F Piozzi,F Senatore (2011). Characterisation of the essential oil of Nepeta glomerata Montbret et Aucher ex Bentham from Lebanon and its biological activities.
K Baser,T Özek (1994). Composition of the Essential Oil of<i>Nepeta caesarea</i>Boiss. from Turkey.
K Baser,T Özek,G Tümen (1995). Composition of the Essential Oil of<i>Nepeta viscida</i>Boiss. from Turkey.
G Kokdil,S Kurucu,G Topcu (1996). Composition of the essential oil of Nepeta nuda L. ssp. albiflora (Boiss.) Gams.
Gamze Kökdil,Semra Kurucu,Gülaçti Topçu (1997). Chemical Constituents of the Essential Oils of Nepeta italica L. and Nepeta sulfuriflora P. H. Davis.
G Kokdil,M Tanker,S Kurucu,G Topçu (1997). Essential oil analysis of Nepeta cilicia Boiss.
K Baser,B Demircakmak,A Altintas,H Duman (1998). Composition of the essential oils of Nepeta cadmea Boiss.
G Tümen,K Baser,M Kürkçüoglu,B Demirci,B Yildiz (1999). Composition of the Essential Oil of<i>Nepeta trachonitica</i>Post from Turkey.
M Dabiri,F Sefidkon (2003). Chemical composition of the essential oil of <i>Nepeta racemosa</i> Lam. from Iran.
O Kilic,S Hayta,E Bagci (2011). Chemical composition of essential oil of Nepeta nuda L. subsp. nuda (Lamiaceae) from Turkey.
A Adiguzel,H Ozer,M Sokmen,M Gulluce,A Sokmen,H Kilic,O Baris (2009). Antimicrobial and antioxidant activity of the essential oil and methanol extract of Nepeta cataria.
Arzu Gormez,Sedat Bozari,Derya Yanmis,Medine Gulluce,Guleray Agar,Fikrettin Sahin (2013). Antibacterial activity and chemical composition of essential oil obtained from<i>Nepeta nuda</i>against phytopathogenic bacteria.
N Hasimi,S Kizil,V Tolan (2015). Essential oil components, microelement contents and antioxidant effects of Nepeta italica L. and Achillea filipendulina Lam.
Fuat Bozok,Menderes Cenet,Gökhan Sezer,Zeynep Ulukanli (2017). Essential Oil and Bioherbicidal Potential of the Aerial Parts of<i>Nepeta nuda</i>subsp.<i>albiflora</i>(Lamiaceae).
Cengiz Sarikurkcu,Olcay Ceylan,Said Targan,Sanja Ćavar Zeljković (2018). Chemical composition and biological activities of the essential oils of two endemic Nepeta species.
A Gilani,A Shah,A Zubair,S Khalid,J Kiani,A Ahmed,V Ahmad (2009). Chemical composition and mechanisms underlying the spasmolytic and bronchodilatory properties of the essential oil of Nepeta cataria L.
Arzu Kaska,Nahide Deniz,Mehmet Çiçek,Ramazan Mammadov (2018). Evaluation of Antioxidant Properties, Phenolic Compounds, Anthelmintic, and Cytotoxic Activities of Various Extracts Isolated from <i>Nepeta cadmea</i> : An Endemic Plant for Turkey.
İbrahim Teber,Ercan Bursal (2020). Phenolic Compounds and Antioxidant Activity of <i>Nepeta nuda </i>subsp.<i> Albiflora</i>.
S Goldansaz,C Festa,E Pagano,S De Marino,C Finamore,O Parisi,F Borrelli,A Sonboli,M & D'auria (2019). Phytochemical and Biological Studies of Nepeta asterotricha Rech. f. (Lamiaceae): Isolation of Nepetamoside.
J Bošnjak-Neumüller,Radakovi,M Djelić,N Vuković-Gačić,B Stevanović,Z Kolarević,S Mišić,D Stankovi Ć M, Knežević -Vukčević,J Spremo-Potparević,B Stanimirović,Z (2017). Nepeta rtanjensis (Lamiaceae), a plant endemic to the Balkans: Phenolic composition, antioxidant activity, and in vitro antigenotoxic effects in triiodothyronineinduced DNA damage in human lymphocytes.
A Balasubramanian,K Ponnuraj (2010). Crystal structure of the first plant urease from jack bean: 83 years of journey from its first crystal to molecular structure.
Da-Hua Shi,Zhong-Lu You,Chen Xu,Qiang Zhang,Hai-Liang Zhu (2007). Synthesis, crystal structure and urease inhibitory activities of Schiff base metal complexes.
Toshihisa Ohta,Hideyuki Shibata,Toshihiko Kawamori,Masaki Iimuro,Takashi Sugimura,Keiji Wakabayashi (2001). Marked Reduction of Helicobacter pylori-Induced Gastritis by Urease Inhibitors, Acetohydroxamic Acid and Flurofamide, in Mongolian Gerbils.
Toshimitsu Hayashi,Kazuko Sawa,Masaru Kawasaki,Munehisa Arisawa,Minen Shimizu,Naokata Morita (1988). Inhibition of Cow's Milk Xanthine Oxidase by Flavonoids.
A Nagao,M Seki,H Kobayashi (1999). Inhibition of Xanthine Oxidase by Flavonoids.
S Lin,G Zhang,Y Liao,J Pan,D Gong (2015). Dietary Flavonoids as Xanthine Oxidase Inhibitors: Structure-Affinity and Structure-Activity Relationships.
D Van Hoorn,R Nijveldt,P Van Leeuwen,Z Hofman,L M'rabet,D De Bont,K Van Norren (2002). Accurate prediction of xanthine oxidase inhibition based on the structure of flavonoids.
F Abak (2018). Şanlıurfa ili Lamiaceae (Ballıbabagiller) Familyasının Florası, Bazı Taksonların Fitokimyasal ve Etnobotanik Özellikleri.
F Mclafferty,D Stauffer (1989). The Wiley/NBS Registry of Mass Spectral Data.
R Adams (2017). Identification of Essential Oil Components by Gas chromatography/Mass Spectrometry.
George Hochmuth,Robert Hochmuth,Stephen Olson (2008). Polyethylene Mulching for Early Vegetable Production in North Florida.
J Curvers,J Rijks,C Cramers,K Knauss,P Larson (1985). Temperature programmed retention indices: Calculation from isothermal data. Part 1: Theory.
V Singleton (1985). Citation Classic -Colorimetry of Total Phenolics with Phosphomolybdic-Phosphotungstic Acid Reagents.
W Brand-Williams,M Cuvelier,C Berset (1995). Use of a free radical method to evaluate antioxidant activity.
Marino Arnao,Antonio Cano,Manuel Acosta (2001). The hydrophilic and lipophilic contribution to total antioxidant activity.
M Weatherburn (1967). Phenol-hypochlorite reaction for determination of ammonia.
M Akdeniz,A Ertas,I Yemer,M Firat,U Kolak (2020). Phytochemical and biological investigations on two Nepetaspecies: Nepeta heliotropifolia and N. congesta subsp. Cryptantha.
K Başer,Gülmira Özek,Temel Özek,Ahmet Duran,Hayri Duman (2006). Composition of the essential oils ofRhabdosciadium oligocarpum (Post ex Boiss.) Hedge et Lamond andRhabdosciadium microcalycinum Hand.-Mazz..
A Ali,N Tabanca,B Demirci,E Blythe,K Baser,I Khan (2016). Chemical composition and biological activity of essential oils from four Nepeta species and hybrids against Aedes aegypti (L.) (Diptera: Culicidae).
E Sezik,E Kocakulak,K Baser,T Ozek (2005). Composition of the Essential Oils of Juniperus oxycedrus subsp. Macrocarpa from Turkey.
V Babushok,P Linstrom,I Zenkevich (2011). Retention indices for frequently reported compounds of plant essential oils.
N Zeynep Tunalıer,K Kırımer,Başer (2004). A Potential New Source of Cedarwood Oil: Juniperusfoetidissima Willd.
M Margraf (2015). Comparison of Compounds in Bourbon Vanilla Extract and Vanilla Flavour.
M Petersen (2013). Rosmarinic acid: new aspects.
G Bartley,R Avena-Bustillos,W.-X Du,M Hidalgo,B Cain,A Breksa,Iii (2016). Transcriptional regulation of chlorogenic acid biosynthesis in carrot root slices exposed to UV-B light.
Faten Taram,Aimee Winter,Daniel Linseman (2016). Neuroprotection comparison of chlorogenic acid and its metabolites against mechanistically distinct cell death-inducing agents in cultured cerebellar granule neurons.
Faten Taram,Elizabeth Ignowski,Nathan Duval,Daniel Linseman (2018). Neuroprotection Comparison of Rosmarinic Acid and Carnosic Acid in Primary Cultures of Cerebellar Granule Neurons.
Ghulam Fareed,Nighat Afza,Ali Versiani,Nazia Fareed,Rasheed Mughal,Ali Kalhoro,Lubna Iqbal,Mehreen Lateef (2013). Synthesis, spectroscopic characterization and pharmacological evaluation of oxazolone derivatives.
A Gökbulut,G Yılmaz (2020). Reithrodontomys humulis.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Fatma Seker. 2026. \u201cScreening of Therapeutic Potential and Compounds of Endemic Nepeta pilinux P.H. Davis in Kew Bull. from Şanlıurfa\u201d. Global Journal of Science Frontier Research - B: Chemistry GJSFR-B Volume 24 (GJSFR Volume 24 Issue B1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
and their composition was investigated using GC-FID and GC/MS. Determination of antioxidant capacity, and urease and xanthine oxidase inhibitions of the methanolic extracts were performed with HPLC-DAD and spectrophotometry. 34 compounds were identified constituting 89.2% of the total essential oil compounds. The major components were determined as T-cadinol (31.1%), γmuurolene (14.4%) and 14-nor-cadin-5-en-4-one isomer A (11.0%) in the oil. Mainly rosmarinic acid, chlorogenic acid, and caffeic acid derivatives were quantified together with apigenin, luteolin and tangeretin derivatives in the extracts by HPLC-DAD. The total phenolics of the extract from leaf and flower parts, 50.81 mg GAE.g-1, was higher than the extract from stem part, and the radical scavenging activity of this extract was also stronger. While, the leaf and flower extract had significant urease and xanthine oxidase inhibitory activities (62.47 and 48.48 µg.mL-1), stem extract had low inhibition on both enzymes.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.