## I. INTRODUCTION
In economically developed countries, an increase in the life span and a decrease in the birthrate leads to population aging, which is accompanied by an increase in neurodegenerative, cerebrovascular diseases, and dementia. In $60 - 80\%$ of cases, dementia develops as a result of Alzheimer's disease (AD), which is the most common neurodegenerative disorder. The number of people suffering from this disease is constantly increasing. In 2021, in the United States alone, 6.2 million people aged 65 and over have AD. By 2060, this number can increase to 13.8 million [1].
To date, the etiology and pathogenesis of AD have not been fully understood. The process of AD development is a complex and multicomponent one [2-4]. In recent years, an increasing number of studies have appeared underlying the important role of cerebral angioarchitectonics and microcirculation disorders in AD development [5-13]. These disorders have their own particularities, specific only for this disease.
In AD, vascular lesions show themselves as cerebral small vessel disease (CSVD), which is manifested in Dyscirculatory Angiopathy of Alzheimer's type (DAAT). This angiopathy leads to structural changes in blood supply and microcirculation specific for this disease, which in turn contributes to the development of metabolic disorders of amyloid- $\beta$ (A $\beta$ ) and tau protein [6-12, 14-18].
Vascular and microcirculatory changes during AD begin to develop many years before the visual symptoms of the disease and are observed in the offspring of patients with AD even at an early age [1,10,19]. These changes in the microvasculature might be hereditary [20].
Reduction of cerebral capillaries play the main role in the development of this process [6-12, 15-17]. In the hippocampus and temporal areas, and, then, in the frontoparietal regions, capillaries become thinner, their branching and their number decrease, which leads to the formation of hypovascular zones [6, 8-12]. These changes result in reduced arterial flow to cerebral tissues and lead to AD-specific hemodynamic changes
[9, 15-17]. Since the blood flowing through the arterial branches cannot pass through the reduced capillary bed, increased "tortuosity" of the intracerebral arterial branches develops [6, 10]. At the same time, there open large arteriovenous shunts, through which arterial blood is discharged into the venous bed [6, 10, 20]. In the temporal and frontoparietal areas, the venous bed overflows, causing venous stasis and impaired venous outflow [10, 20]. The arteriovenous shunts and impaired venous outflow contribute to the deterioration of cerebral hemodynamics even more [9, 10] Patients suffering from other neurodegenerative and cerebrovascular diseases do not have the combination of these pathological changes [10, 17, 20].
Changes in the cerebral arterial, microcirculatory and venous beds, as well as the developed hypoxia, cause death of mitochondria in the cells of the smooth endoplasmic reticulum and the Golgi apparatus, loss of synapses, degeneration, and death of neurons [9,10,12,15-17,21-26]. The combination of these changes result in a complex lesion of the neurovascular unit (NVU) [7-9, 26]. At the same time, these changes cause blood-brain barrier disorders (BBB) [14, 27, 28].
In turn, hypoprefusion and hypoxia characteristic of AD affect the exchange of amyloid- $\beta$, decreasing its excretion and increasing its accumulation. These metabolic disorders lead to the deposition of amyloid- $\beta$ in the cerebral tissue and vascular wall [7, 9, 12, 27]. These deposits cause a decrease in microvascular elasticity, they narrow the capillary lumen, reduce intracerebral blood flow and lead to an even greater increase in hypoxia and neurodegeneration development [13, 22-26, 29]. The growth of these changes advances AD development and progression [20, 23, 24].
While developing new AD treatment methods, it is necessary to keep in mind improvement of capillary blood supply and stimulation of the development of regenerative processes in the cerebral tissue. To achieve these, it is advisable to apply complex methods of action.
A big step in the development of such new complex AD treatments is the use of laser with low output power of the red or near-infrared (NIR) spectrum (600-1100 nm), which is called laser Photobiomodulation Therapy (PBMT) [30].
As a result of experimental and clinical studies on the treatment of AD, it has been found out that lasers with low output power stimulate metabolic processes in the cerebral tissue, restore the exchange of ATP in the mitochondria of neurons, stimulate physiological angiogenesis causing collateral and capillary cerebral revascularization, stimulate neurogenesis and restoration of cerebral tissues. This complex effect leads to a decrease in the level of dementia, restoration of cognitive functions and improved daily living [31-38].
According to the methods of laser energy delivery, PBMT is subdivided into transcranial, [31, 32, 35] intranasal, often combined with transcranial, [34, 37] and transcatheter intracerebral methods [39].
Previous experimental studies have allowed us to move to the clinical application of transcatheter intracerebral laser PBMT [29, 40-44].
This work is dedicated to the clinical application of transcatheter intracerebral laser PBMT for stimulating angiogenesis and neurogenesis in various AD stages.
## II. METHODS
All examinations, conservative treatment methods and intracerebral transcatheter laser interventions in this research were carried out with the approval of the Ethical Review Board (ERB) (Protocol No. 3 of 01-12-2003, Protocol No. 12 of 04-30-2014), as well as with the written consent of patients and their relatives.
The selection of patients for the test and control groups includes:
- Consent of patients, as well as their relatives, for examination and treatment;
- Absence of serious concomitant diseases that could interfere with examination and treatment;
- The somatic state of the patients allowing their examination and treatment;
- Involutive changes in the temporal and frontal parts of the brain corresponding to AD;
- Signs of dementia and cognitive disorders corresponding to AD.
Of 210 people who had been diagnosed with AD in its various stages, we selected 97 patients aged 34 to 80 (average age 67.6 years), 34 (35.05%) men and 63 (64.95%) women. The patients were divided into the test group - 48 (49.48%) patients - and the control group - 49 (50.52%) patients.
All patients of the test and control groups had similar somatic state and severity of dementia and cognitive impairment, in accordance with their AD stages.
### Examination plan of patients
The examinations of the patients were carried out according to the following scheme:
- Assessment of the clinical severity of dementia was carried out using The Clinical Dementia Rating scale (CDR) [45];
- Assessment of the severity of cognitive impairment was carried out using the Mini-Mental State Examination (MMSE) [46];
- Assessment of cerebral blood flow and microcirculation was made by means of cerebral scintigraphy (SG) in static and dynamic modes by means of a gamma camera of the "Ohio Nuclear" company, USA, using TC 99M pertechnetate 555;
- Assessment of cerebral perfusion blood filling was carried out using rheoencephalography (REG) on the apparatus "Reospectrum-8", "Neurosoft", Russia; (All of the abovementioned examinations were carried out at patients' admission, then at their discharge, and then in a more distant period with an interval of 6-12 months).
- Laboratory examination was carried out in accordance with the criteria of interventional neuroangiology;
- Assessment of cerebral structural changes was carried out by means of CT and MRI on Somatom (Siemens), Hi Speed (GE), Tomoscan (Philips), Apetro Eterna (Hitachi) using digital program of image processing ATAA (Advance Tomo Area Analysis). The digital processing allows, in percentage terms, to reveal a decrease in the tissue volume of temporal lobes in comparison with their normal volume, thereby showing the severity of involutive changes [6, 10, 41].
- The digital scale of Tomography Dementia Rating scale (TDR) was also used, with the help of which the stage of dementia in AD was morphometrically determined in accordance with the severity of atrophic changes in the temporal lobes detected during CT and MRI [40-42, 47]. Thus, patients were
divided into groups: TDR-0 - preclinical AD stage (atrophy of temporal lobes of $4 - 8\%$ ), TDR-1 - mild AD stage (atrophy of temporal lobes $9 - 18\%$ ), TDR-2 - moderately severe AD stage (atrophy of temporal lobes of $19 - 32\%$ ), TDR-3 - severe AD stage (atrophy of temporal lobes of $33 - 62\%$ ) [47]. The examinations were carried out upon the admission of the patient, then, to determine the dynamics of cerebral changes and dementia, at intervals of 6-12 months (the examinations were carried out in independent laboratories);
Assessment of the intracerebral vascular and capillary bed was carried out using cerebral multigated angiography (MUGA) carried out according to the classical technique by transfemoral access using Advantx (GE) devices. The intensity of capillary blood flow was recorded using the digital program of image processing "Angio Vision", which shows changes in the density of capillary blood flow [6, 10, 23, 39, 42]. The primary examination was carried out upon admission of the patient to the clinic and repeated immediately after the PBMT (for the patients of the test group). Further studies were carried out with an interval of 2 to 7 years. In some cases, MSCT angiography (MSCTA) or MR angiography (MRA) was used.
Table 1: Results of examination of patients in the test and control groups
<table><tr><td>CHARACTERISTIC OF IDENTIFIED CHANGES</td><td>Test Group
N - 48</td><td>Control Group
N - 49</td></tr><tr><td>Clinical Dementia Determination</td><td></td><td></td></tr><tr><td>CDR - 1</td><td>16</td><td>12</td></tr><tr><td>CDR - 2</td><td>21</td><td>21</td></tr><tr><td>CDR - 3</td><td>7</td><td>10</td></tr><tr><td>Cognitive Disorders</td><td></td><td></td></tr><tr><td>Decrease to 26-28 MMSE points</td><td>4</td><td>6</td></tr><tr><td>Decrease to 20-25 MMSE points</td><td>16</td><td>12</td></tr><tr><td>Decrease to 12-19 MMSE points</td><td>21</td><td>15</td></tr><tr><td>Decrease to 7-11 MMSE points</td><td>7</td><td>10</td></tr><tr><td>Morphometric determination of dementia stages on TDR scale according to CT and MRI</td><td></td><td></td></tr><tr><td>TDR - 0 (4-8% temporal lobes atrophy)</td><td>4</td><td>6</td></tr><tr><td>TDR - 1 (9-18% temporal lobes atrophy)</td><td>16</td><td>12</td></tr><tr><td>TDR - 2 (19-32% temporal lobes atrophy)</td><td>21</td><td>21</td></tr><tr><td>TDR - 3 (33-62% temporal lobes atrophy)</td><td>7</td><td>10</td></tr><tr><td>Assessment of cerebral blood flow according to SG</td><td></td><td></td></tr><tr><td>Decreased blood flow in cerebral hemispheres</td><td>48</td><td>49</td></tr><tr><td>Assessment of cerebral perfusion blood filling according to REG</td><td></td><td></td></tr><tr><td>Decreased volumetric pulse blood supply</td><td>48</td><td>49</td></tr><tr><td>Dyscirculatory angiopathy of Alzheimer's type (DAAT) according to MUGA</td><td></td><td></td></tr><tr><td>Reduction of capillaries in the temporal areas</td><td>48</td><td>49</td></tr><tr><td>Development of hypovascular zones in temporal areas</td><td>48</td><td>49</td></tr><tr><td>Decreased arterial flow in the temporal areas</td><td>48</td><td>49</td></tr><tr><td>Development of arteriovenous shunts in the temporal areas</td><td>48</td><td>49</td></tr><tr><td>Development of venous stasis and impaired venous outflow</td><td>44</td><td>43</td></tr><tr><td>Development of increased tortuosity of intracerebral arteries</td><td>38</td><td>38</td></tr></table>
Treatment methods
The Test Group: 48 (51.61%) patients, 17 (35.42%) men and 31 (54.58%) women, received Transcatheter Intracerebral Laser Photobiomodulation Therapy (PBMT).
Patients with the preclinical stage of the disease (TDR-0) underwent the intervention against the background of growing memory impairment. Patients with clinical stages of the disease (TDR-1, TDR-2, TDR-3) received their interventional treatment in the period from 1 to 12 years after the diagnosis of AD.
Transcatheter Intracerebral Laser Photobiomodulation Therapy (PBMT) In catheterization laboratories, under local anesthesia and fluoroscopic control, guiding catheters are used to probe intracerebral arterial branches approaching hypovascular zones [10,23]. Coaxially through these catheters, a flexible laser fiber optic light guide instrument, 25-100 micrometers in diameter and connected to a laser machine, is passed. Laser PBMT is performed using a helium-neon laser ULF-01 (Russia) [39,43,44] The parameters of intracerebral transcatheter laser exposure: wavelength 632.8 nanometers; laser output power 25-45 mW; fiber output power 24-44 mW; treatment session duration 1200-2400 seconds; beam spot diameter in the vessel 1-2 mm; average dose during treatment 29-106 J [40]. With the transcatheter intracerebral method of delivery, the depth of laser energy penetration into the cerebral tissues is 20-40 mm. [40]. Consequently, power density and energy density are variable [39,43,44].
PBMT is performed on both the right and left hemispheres. If necessary, a solution of a radiopaque substance (Omnipak 350) is injected in small doses for fluoroscopic control.
After PBMT, repeated cerebral MUGA is performed using the digital program of image processing "Angio Vision", which makes it possible to determine changes in the density of arterial and capillary blood flow. The results are used to assess the severity of intracerebral angiogenesis, the degree of collateral revascularization and restoration of microcirculation [23,39,40,43,44]. This program automatically registers the obtained changes in the vascular image in real time, in the corresponding contrast phase.
After trascatheter intracerebral PBMT, according to the generally accepted schemes, the patients underwent disaggregant, anticoagulant, antioxidant, vasodilating and nootropic therapy. The patients received: Aspirin, depending on the parameters of the blood coagulation system, Heparin, indirect anticoagulants, Pentoxifylline $100\mathrm{mg}$, Complamin 150 mg, Inosin $200\mathrm{mg}$, Nootropil (Piracetam) $1200\mathrm{mg}$ (or Gliatilin $1000\mathrm{mg}$ ) intravenously, with a drop counter, No. 10-15, followed by pills. In the subsequent period, the courses of pills were repeated twice a year. The patients did not receive any specific therapy aimed at treating AD.
The Control Group: 49 (50.52%) patients, 16 (32.65%) men, 33 (67.35%) women, received conservative treatment.
The conservative treatment was carried out according to generally accepted schemes [49, 50]. Patients from TDR-0 group received: Nootropil (Piracetam) $2400\mathrm{mg}$ per day (courses of 3-4 months) or Gliatilin $1200\mathrm{mg}$ per day (courses of 4-6 months). Patients from groups TDR-1, TDR-2, TDR-3 received Memantine $5 - 20\mathrm{mg}$ per day or Rivastigmine $3 - 12\mathrm{mg}$ per day. At the same time, like patients from the Test Group, all patients from the Control Group received vasoactive drugs Pentoxifylline $800\mathrm{mg}$ per day in courses of 3 months and Complamin $450\mathrm{mg}$ per day in courses of 2-3 months, which were repeated twice a year.
Statistical analysis
The result data were processed statistically using the Statsoft Statistica 10 software (StatSoft Inc., USA). In the test and control groups, a contingency table analysis was made by means of the Chi-square test to compare the characteristics of Before/After treatment. Post-treatment indicators significantly differed from pre-treatment indicators in each group $(p < 0.05)$, and the statistical significance of the results was significantly higher in the test group $(p = 0.00130)$ than in the control group $(p = 0.01044)$.
## III. RESULTS
The Test Group
Direct results
After transcatheter intracerebral laser PBMT, according to digital MUGA, all 48 (100%) patients showed a direct positive result manifested in pronounced angiogenesis, collateral and capillary revascularization, reduction of arteriovenous shunts, as well as an improvement in venous outflow (Figures 1A, 1B, Figures 2A, 2B).
There were no complications associated with transcatheter intracerebral laser PBMT.
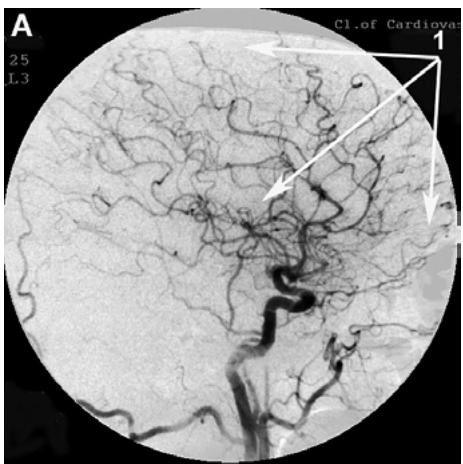
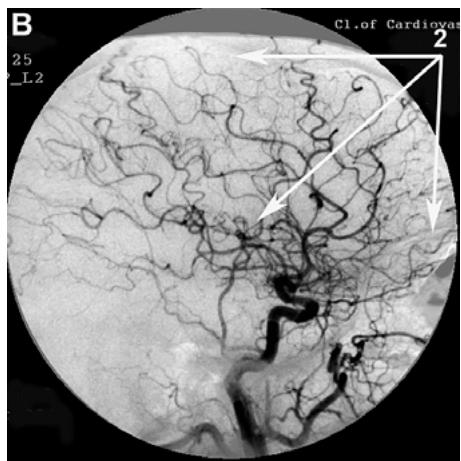 Figure 1: Patient S., 42 years old, male. Medical history of AD - 2 years, TDR-1.
A. Left internal carotid artery angiogram, arterial phase, before transcatheter Intracerebral laser PBMT: 1. hypovascular areas in temporal and frontoparietal regions. B. Left internal carotid artery angiogram, arterial phase, after transcatheter intracerebral laser PBMT: 2. stimulation of angiogenesis, restoration of collateral and capillary blood supply in the temporal and frontoparietal region.
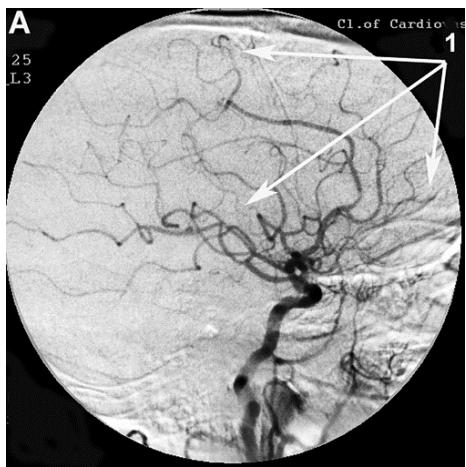
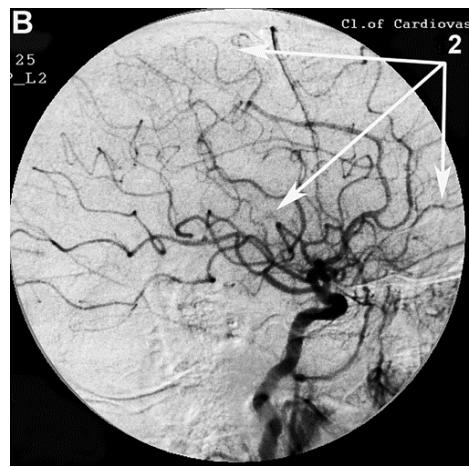 Figure 2: Patient P., 75 years old, female. Medical history of AD - 12 years, TDR-3.
A. Left internal carotid artery angiogram, arterial phase, before transcatheter Intracerebral laser PBMT: 1. hypovascular areas in temporal and frontoparietal regions; B. Left internal carotid artery angiogram, arterial phase, after transcatheter Intracerebral laser PBMT: 2. stimulation of angiogenesis, restoration of collateral and capillary blood supply in the temporal and frontoparietal regions.
Early period (1-6 months) after transcatheter intracerebral laser PBMT
Clinically, all 4 (100%) patients showed improvement of memory as well as restoration of cognitive functions to the level of 28-30 MMSE points.
According to CT and MRI data, all 4 (100%) patients had an increase in the volume of the temporal lobes, which was accompanied by narrowing of the Sylvian fissures and restoration of the subarachnoid space.
According to SG and REG data, all 4 (100%) patients demonstrated normalization of blood flow velocity and pulse blood filling in the cerebral hemispheres.
Patients in AD mild stage (TDR-1)
Clinically, all 16 (100%) patients showed a decrease in the level of dementia. Of these, 6 (37.50%) showed an improvement in cognitive functions to the level of 25-26 MMSE points, and 10 (62.50%) to the level of 27-28 MMSE points.
According to CT and MRI data, all 16 (100%) patients had a tendency to an increase in the volume of temporal lobes accompanied by narrowing of the Sylvian fissures and subarachnoid space reduction.
According to SG and REG data, all 16 (100%) patients demonstrated normalization of blood flow velocity and pulse blood filling in the cerebral hemispheres.
Clinically, all 21 (100%) patients demonstrated a decrease in the level of dementia. Of these, 12 (57.14%) showed an improvement in cognitive functions to the level of 19-20 MMSE points, and 9 (42.86%) to the level of 21-22 MMSE points.
According to CT and MRI data, all 21 (100%) patients had a tendency to an increase in the volume of temporal lobes accompanied by narrowing of the Sylvian fissures and subarachnoid space reduction.
According to SG and REG data, all 21 (100%) patients showed signs of restoration of blood flow velocity and pulse blood filling in the cerebral hemispheres.
Patients in severe AD stage (TDR-3)
Clinically, all 7 (100%) patients demonstrated signs of a decrease in the level of dementia, as well as an improvement in cognitive functions to the level of 11-12 MMSE points.
According to CT and MRI data, all 7 (100%) patients had a tendency to an increase in the volume of the cerebral temporal lobes, accompanied by narrowing of the Sylvian fissures and the subarachnoid space reduction.
According to SG and REG data, all 7 (100%) patients showed signs of restoration of blood flow velocity and pulse blood filling in the cerebral hemispheres.
Long-term period (1-7 years) after transcatheter intracerebral laser PBMT
Patients in preclinical AD stage (TDR-0)
One year after transcatheter intracerebral PBMT, all 4 (100%) patients demonstrated sustained recovery of memory and cognitive functions to the level of 28-30 MMSE points (Table 2).
According to the results of digital processing of CT and MRI images, all 4 (100%) patients had restoration of the volume of the cerebral temporal lobes to the age norm, along with narrowing of the Sylvian fissures and restoration of the subarachnoid space. As a result, all 4 (100%) patients were considered to be practically healthy people without dementia or cognitive impairment.
According to SG and REG data, all 4 (100%) patients showed normalization of blood flow velocity and pulse blood filling in the cerebral hemispheres.
In a longer period of over one year, all 4 (100%) patients demonstrated positive dynamics persisting throughout the observation period (Table 2).
Patients in mild AD stage (TDR-1)
One year after PBMT, all 16 (100%) patients showed no signs of dementia and demonstrated sustained recovery of cognitive functions to the level of 27-28 MMSE points (Table 2).
According to the results of digital processing of CT and MRI images one year after the treatment, all 16 (100%) patients had an 8-10% decrease in the temporal lobes atrophy (Figures 3A, 3B). After 2-4 years, 13 (81.25%) patients showed a further 4-5.5% decrease in the temporal lobes atrophy leading to an almost complete restoration of the temporal lobe volume to the age norm (Figure 3B, 3C). Narrowing of the Sylvian fissures and restoration of the subarachnoid space accompanied the process. In accordance with the abovementioned criteria, all 16 (100%) patients were transferred to Group TDR-0.
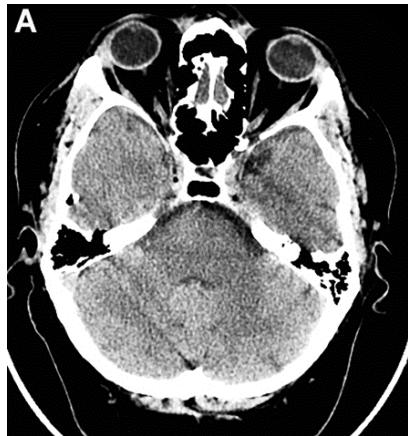
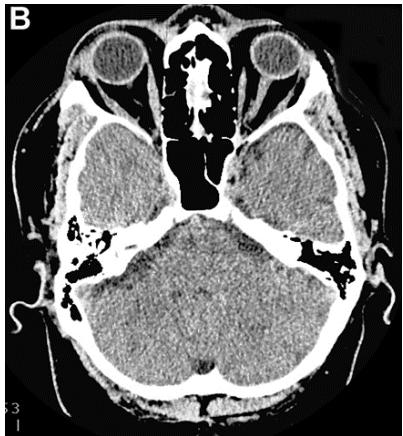
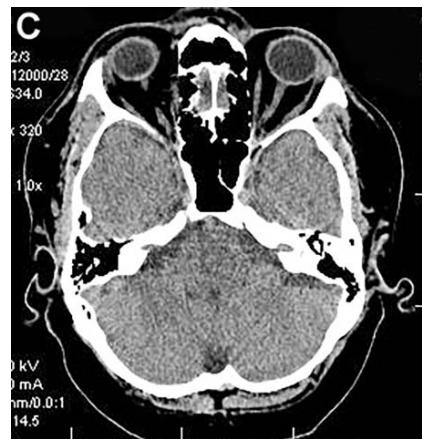 Figure 3: Same patient S., 42 years old, male.
A. Cerebral CT before transcatheter intracerebral laser PBMT: Total atrophy of the temporal lobes is $18\%$ of the total tissue volume (TDR-1). B. Cerebral CT in 12 months after transcatheter intracerebral laser PBMT: total atrophy of the temporal lobes decreased to $9\%$ of the total tissue volume (TDR-1). C. Cerebral CT in 4 years after transcatheter intracerebral laser PBMT: total atrophy of the temporal lobes decreased to $5.5\%$ of the total tissue volume.
The patient is transferred to AD group in TDR-0 stage.
According to SG and REG data, all 16 (100%) patients showed normalization of blood flow velocity and pulse blood filling in the cerebral hemispheres.
In a longer period of over one year, all 16 (100%) patients demonstrated that the obtained positive dynamics persisted throughout the observation period (Table 2).
### Patients in moderately severe AD stage (TDR-2)
One year after PBMT, all 21 (100%) patients showed a decrease in the level of dementia and an improvement in cognitive functions to the level of 21-22 MMSE points. After 2-3 years, 12 (57.14%) patients had a further decrease in the level of dementia and restoration of cognitive functions to the level of 23-25 MMSE points. 9 (42.86%) patients' cognitive functions remained at the same level equal to 21-22 MMSE points (Table. 2).
According to the results of digital processing of CT and MRI images one year after the treatment, all 21 (100%) patients had a 5-10% temporal lobe atrophy decrease. It was accompanied by narrowing of the Sylvian fissures and subarachnoid space. After 2-3 years, 12 (57.14%) patients had a decrease in the temporal lobe atrophy by another 4-5.5%. 9 (42.86%) patients showed no further pronounced decrease in involutive changes.
As a result, 16 (76.19%) patients were transferred to TDR-1 group, and 5 (23.81%) patients remained in TDR-2 group.
According to SG and REG data, all 21 (100%) patients demonstrated positive dynamics of blood flow velocity and pulse blood filling in the cerebral hemispheres.
4 years after the treatment, all 21 (100%) patients had a tendency to a gradual decrease in cognitive functions.
#### Patients in severe AD stage (TDR-3)
One year after PBMT, all 7 (100%) patients demonstrated a decrease in the level of dementia. 4 (57.14%) patients had an improvement of cognitive functions to the level of 11-14 MMSE points, and 3 (42.86%) patients to the level of 15-19 MMSE points (Table 2).
According to the results of digital processing of CT and MRI images one year after the treatment, all 7 (100%) patients had a decrease in the atrophy of the cerebral temporal lobes. It was accompanied by narrowing of the Sylvian fissures and subarachnoid space. Of these, 5 (71.43%) patients had a 10-12% decrease (Figures 4A, 4B), and 2 (28.57%) a 6-8% decrease.
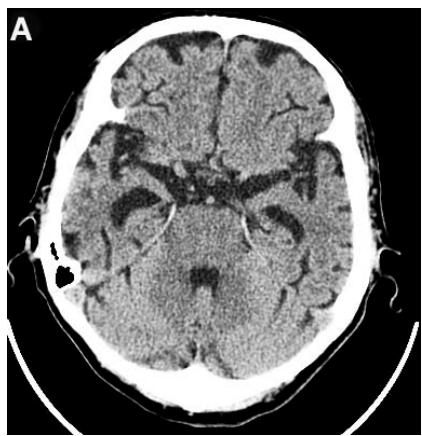
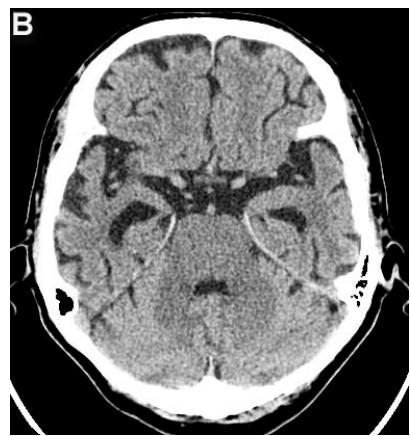 Figure 4: Same patient P., 75 years old, female TDR-3.
A. Cerebral CT before transcatheter intracerebral laser PBMT: Total atrophy of the temporal lobes is $40\%$ of the total tissue volume (TDR-3). B. Cerebral CT 12 months after transcatheter intracerebral laser PBMT: Reduction of the total atrophy of the temporal lobes by $12\%$. The patient is transferred to AD group in TDR-2 stage.
Accordingly, 5 (71.43%) patients were transferred to TDR-2 group, 2 (28.57%) patients remained in TDR-3 group (Table 2).
According to SG and REG data, all 7 (100%) patients showed preservation of the positive dynamics of blood flow velocity and pulse blood filling in the cerebral hemispheres.
After 2-2.5 years after the treatment, there was a tendency to an increase in dementia and a decrease in cognitive functions.
9 (21.74%) Test Group patients underwent repeated cerebral MUGA in the period from 2 to 6 years after transcatheter intracerebral PBMT. All 9 (100%) patients demonstrated preservation and further progression of angiogenesis, accompanied by collateral and capillary revascularization, was observed in.
Table 2: Long-term results of treating test and control groups patients according to the severity of dementia and cognitive impairment.
<table><tr><td colspan="2">Signs of dementia and cognitive impairment</td><td>Test Group before treatment n-48</td><td>Test Group after treatment n-48</td><td>p (chi-square)</td></tr><tr><td colspan="2">Practically healthy
(MMSE - 29-30 points)</td><td>0</td><td>4</td><td rowspan="5">p=0.00130</td></tr><tr><td>TDR-0</td><td>(MMSE - 26-28 points)</td><td>4</td><td>16</td></tr><tr><td>TDR-1</td><td>(MMSE - 20-25 points)</td><td>16</td><td>16</td></tr><tr><td>TDR-2</td><td>(MMSE - 12-19 points)</td><td>21</td><td>10</td></tr><tr><td>TDR-3</td><td>(MMSE - 7-11 points)</td><td>7</td><td>2</td></tr><tr><td></td><td></td><td>Control Group before treatment n-49</td><td>Control Group after treatment n-49</td><td></td></tr><tr><td colspan="2">Practically healthy
(MMSE - 29-30 points)</td><td>0</td><td>0</td><td rowspan="5">p=0.01044</td></tr><tr><td>TDR-0</td><td>(MMSE - 26-28 points)</td><td>6</td><td>3</td></tr><tr><td>TDR-1</td><td>(MMSE - 20-25 points)</td><td>12</td><td>5</td></tr><tr><td>TDR-2</td><td>(MMSE - 12-19 points)</td><td>21</td><td>10</td></tr><tr><td>TDR-3</td><td>(MMSE - 7-11 points)</td><td>10</td><td>31</td></tr></table>
The Control Group
### Direct results
Immediately after the first course of conservative treatment, all 6 (100%) patients in preclinical AD stage (TDR-0) and all 12 (100%) patients in mild AD stage (TDR-1) showed a trend towards an improvement in cognitive functions. Patients in moderately severe AD stage (TDR-2) and in severe AD stage (TDR-3) had no pronounced positive dynamics.
Early period (1-6 months) against the background of conservative treatment
Patients in preclinical AD stage (TDR-0)
Clinically, all 6 (100%) patients had a tendency to improve memory, as well as to restore cognitive functions to the level of 27-28 MMSE points.
According to CT and MRI data, all 6 (100%) patients had no structural cerebral changes.
According to SG and REG data, all 6 (100%) patients had an improvement in blood flow velocity and pulse blood filling in the hemispheres.
Clinically, all 12 (100%) patients had a tendency towards stabilization of their condition.
According to CT and MRI data, all 12 (100%) patients showed no structural cerebral changes.
According to SG and REG data, all 12 (100%) patients showed signs of improvement in blood flow velocity and pulse blood filling in the hemispheres.
Clinically, $15(71.43\%)$ patients showed a tendency towards a further increase in dementia and a decrease in cognitive functions. 6 $(28.57\%)$ patients showed no dynamics.
According to CT and MRI data, all 21 (100%) patients had no structural cerebral changes.
According to SG and REG data, all 21 (100%) patients demonstrated signs of improvement in blood flow velocity and pulse blood filling in the hemispheres.
Patients in severe AD stage (TDR-3)
Clinically, all 10 (100%) patients showed a trend towards a further increase in dementia and a decrease in cognitive functions.
According to CT and MRI data, all 10 (100%) patients had no structural cerebral changes.
According to SG and REG data, all 10 (100%) patients had signs of improvement in blood flow velocity and pulse blood filling in the hemispheres.
Long-term period (1-5 years) against the background of conservative treatment
Patients in preclinical AD stage (TDR-0)
Clinically, within 2 years after the start of the conservative treatment, all 6 (100%) patients had an improvement in memory and stabilization of cognitive functions within 27-29 MMSE points. After 3-5 years, 3 (50.00%) patients showed no obvious signs of dementia. 3 (50.00%) patients had early signs of dementia, as well as a decrease in cognitive functions to 24-25 MMSE points.
According to the results of digital processing of CT and MRI images after 2 years, 1 (16.67%) patient showed no signs of an increase in cerebral involutive changes. 3 (50.00%) patients showed signs of an increase in insignificant involutive changes. 2 (33.33%) patients demonstrated a pronounced increase in involutive changes in the temporal lobes and a 14-18% decrease in tissue volume.
As a result, 3 (50.00%) patients remained in TDR-0 group. 3 (50.00%) patients were transferred to TDR-1 group (Table 2).
According to SG and REG data, all 6 (100%) had persistent moderate positive dynamics of blood flow velocity and pulse blood filling in the brain.
#### Patients in mild AD stage (TDR-1)
Clinically, within 2-3 years after the start of the treatment, all 12 (100%) patients showed stabilization of the level of dementia and cognitive functions. In the period of more than 3 years, all 12 (100%) patients had an increase in signs of dementia and a decrease in cognitive functions. Of these, 2 (16.66%) patients showed a decrease in cognitive functions to the level of 20-21 MMSE points, and 10 (83.33%) patients to the level of 18-19 MMSE points.
According to the results of digital processing of CT and MRI images in 3 years, 2 (16.66%) patients did not have any intensification of cerebral involutive changes in the temporal lobes. 10 (83.33%) cases had growing involutive changes and a decrease in the volume of the temporal lobes to 19-24%. As a result, 2 (16.66%) patients remained in TDR-1 group, 10 (83.33%) patients were transferred to TDR-2 group (Table 2). Further on, all 12 (100%) cases showed signs of an increase in involutive changes.
According to SG and REG data, all 12 (100%) patients demonstrated weak positive dynamics of blood flow velocity and pulse blood filling.
## Patients in moderately severe AD stage (TDR-2)
Clinically, in the period of 1-2 years after the start of the treatment, all 21 (100%) patients showed an increase in dementia and cognitive impairment to 11-12 MMSE points. In the period of 3 years and more, all 21 (100%) patients had a further increase in dementia and a decrease in cognitive impairment to the level of 9-10 MMSE points.
According to the results of digital processing of CT and MRI images after 3 years, all 21 (100%) patients had an increase in involutive cerebral changes and a decrease in the volume of temporal lobes to $34 - 40\%$. As a result, all 21 (100%) patients were transferred to TDR-3 group (Table 2).
According to SG and REG, 8 (38.10%) patients had a tendency towards a decrease in cerebral blood flow velocity and pulse blood filling, 13 (61.90%) patients had a clear decrease in cerebral blood flow velocity and pulse blood filling.
Clinically, 1 year after the start of the conservative treatment, all 10 (100%) patients showed an increase in dementia and a decrease in cognitive functions to the level of 7-8 MMSE points.
According to the results of digital processing of CT and MRI images 1 year after the start of conservative treatment, all 10 (100%) patients showed an increase in cerebral involutive changes and a decrease in the volume of the temporal lobes to $40 - 45\%$.
According to SG and REG data, 7 (70.00%) patients had a tendency towards a decrease in the velocity of cerebral blood flow and pulse blood filling, and 3 (30.00%) patients showed a clear decrease in the parameters of cerebral blood flow and pulse blood filling.
## IV. DISCUSSION
### Test group
The progressive aging of the population and the associated cerebral pathological changes show the need to develop new effective methods of complex treatment of neurodegenerative and cerebrovascular diseases [24,38,39,40]. Transcatheter intracerebral laser PBMT has proved to be highly effective in the treatment of various cerebrovascular lesions, Binswanger's disease (BD), vascular parkinsonism (VP), [40,48] as well as Alzheimer's disease (AD).
During transcatheter intracerebral PBMT, laser energy at a wavelength of 632.8 nanometers with varying power densities passes through the blood, vascular wall, and cerebral tissues, which are essentially "turbid media." As we have noted, the depth of energy penetration is 20-40 mm [39,40,43,44]. As a result, rather large volumes of cerebral tissues are exposed to laser irradiation, which leads to a multicomponent action.
SG, REG and digital MUGA in the test group before, as well as in the immediate and remote periods after transcatheter intracerebral PBMT, shows that helium-neon laser stimulates angiogenesis making arterial and capillary collateral branches open. As a result, revascularization occurs in the hypovascular zones not only in the temporal and frontoparietal regions, but also in other parts of the brain. Improvement in arterial and capillary inflow leads to the closure of pathologically developed arteriovenous shunts and a decrease in venous stasis, which reduces hypoperfusion and restores cerebral hemodynamics.
The conducted research demonstrate the mechanism of action of the laser with low output power of the red spectrum on the vascular and microvascular system of the brain. A similar mechanism works during other methods of conducting PBMT: many authors describe improvement in cerebral blood supply and hemodynamics after transcranial or intranasal PBMT during AD and other neurodegenerative and ischemic lesions [30-38].
Digital processing of CT N MRI images performed before and at different times after transcatheter intracerebral PBMT showed that all 48 (100%) patients in the test group had a persistent decrease in cerebral involutive changes and an increase in the volume of the temporal and frontoparietal lobes. It should be noted that the cerebral tissue has a normal structure. The results obtained indicate restoration of cerebral metabolism, stimulation of neurogenesis and development of regenerative processes.
The data obtained are confirmed by experimental and clinical works by many authors. These studies have shown that in transcranial and intranasal PBMT, laser energy with low output power of the red or near-infrared spectrum not only improves blood supply, but also restores adenosine triphosphate (ATP) metabolism in the mitochondria of neurons, reduces neuronal death, reduces apoptosis, stimulates neurogenesis and regeneration of cerebral tissues. Clinically, this leads to a decrease in the level of dementia and an improvement in cognitive functions when tested by MMSE and ADAS-cog [31-38].
Thus, transcatheter intracerebral laser PBMT demonstrated a complex positive effect of the lasers with low output power of the red spectrum on the brain of the patients of the test group.
This exposure resulted in stimulated cerebral angiogenesis and neurogenesis for all 48 (100%) patients in various AD stages. As a result, there was an improvement in cerebral hemodynamics, a decrease in involutive changes and an increase in the volume of the temporal lobes, which led to a decrease in the level of dementia and an improvement in cognitive functions and quality of life in all 48 (100%) cases.
The clinical effect after transcatheter intracerebral PBMT, as well as its duration, depends on the disease stages, the initial volume of the affected cerebral tissue and the severity of the initial dementia. Patients in preclinical stage (TDR-0) and mild stage (TDR-1) showed no signs of dementia and had restoration of cognitive functions throughout the observation period. Patients in moderately severe AD stage (TDR-2) and severe AD stage (TDR-3) showed a decrease in dementia and an improvement in cognitive functions within 4-4.5 and 2-2.5 years respectively.
Control group
SG, REG and digital MUGA for patients of the control group before the treatment, as well as at various times after its beginning, shows that moderate positive dynamics of cerebral blood flow and pulse blood filling is observed mainly in the early period of the treatment. In the long term, there was a gradual decrease in the indicators.
Digital processing of CT and MRI images performed before and at different periods of the conservative treatment, not only did not show a decrease in cerebral involutive changes, but, on the contrary, revealed an increase in atrophic changes in cerebral tissues in all 49 (100%) patients.
Conservative treatment is not effective enough and does not reduce the level of dementia or improve cognitive functions. It allows stabilizing the condition of patients in the early stages of AD for a certain period of time. In the later stages of the disease, the drugs used are ineffective. Similar results have also been described by other authors who carried out similar conservative AD treatment [49, 50].
Pentoxifylline and Complamine allow improving cerebral microcirculation to some extent in cases of early, unpronounced disorders of the cerebral microcirculatory bed.
## V. CONCLUSION
Transcatheter intracerebral PBMT is an effective method for stimulating cerebral angiogenesis. After laser exposure, patients suffering from various stages of AD demonstrate arterial and capillary collateral revascularization leading to an improvement in cerebral hemodynamics and a decrease in hypoxia. At the same time, PBMT stimulates neurogenesis and induces regenerative processes in the brain. This complex effect results in declining cerebral involutive changes and growing volumes of the temporal and frontoparietal regions. The patients, show a decrease in the level of dementia, restoration of cognitive functions, an improvement in the quality of life and daily life activities.
The use of Memantine and Rivastigmine, as well as Pentoxifylline and Complamine in the conservative treatment of patients in various AD stages does not have a pronounced effect on the restoration of metabolic and regenerative processes and the improvement of microcirculation in the brain. With this therapy, the improvement of cerebral blood supply is observed only in the early stages of the disease. Clinically, conservative treatment gives a temporary positive effect, which is manifested in the stabilization of the initial condition of patients in early AD stages, while in the late AD stages these drugs are ineffective.
### Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
#### Funding
Generating HTML Viewer...
References
51 Cites in Article
(2021). 2021 Alzheimer's disease facts and figures.
E Burton,R Barber,E Mukaetova-Ladinska,J Robson,R Perry,E Jaros,R Kalaria,J O’brien (2009). Medial temporal lobe atrophy on MRI differentiates Alzheimer's disease from dementia with Lewy bodies and vascular cognitive impairment: a prospective study with pathological verification of diagnosis.
Willa Brenowitz,Rebecca Hubbard,C Keene,Stephen Hawes,W Longstreth,Randy Woltjer,Walter Kukull (2017). Mixed neuropathologies and estimated rates of clinical progression in a large autopsy sample.
W Weiner,D Veitch,P Aisen,L Beckett,N Cairns,J Cedarbaum (2014). Update of the Alzheimer's Disease Neuroimaging Initiative: A review of papers published since its inception.
Ferdinand Morel (1959). Petite contribution à l'étude d'une angiopathie apparemment dyshorique et topistique.
I Maksimovich (2008). Radiodiagnostics of Alzheimer's disease.
Berislav Zlokovic (2010). Neurodegeneration and the neurovascular unit.
Costantino Iadecola (2010). The overlap between neurodegenerative and vascular factors in the pathogenesis of dementia.
Berislav Zlokovic (2011). Neurovascular pathways to neurodegeneration in Alzheimer's disease and other disorders.
Ivan Maksimovich (2011). Dyscirculatory Angiopathy of Alzheimer's Type.
Stavros Baloyannis,Ioannis Baloyannis (2012). The vascular factor in Alzheimer's disease: A study in Golgi technique and electron microscopy.
Paula Grammas,Joseph Martinez,Alma Sanchez,Xiangling Yin,Jarred Riley,Dylan Gay,Katherine Desobry,Debjani Tripathy,Jinhua Luo,Marianne Evola,Alice Young (2014). A New Paradigm for the Treatment of Alzheimer's Disease: Targeting Vascular Activation.
Ian Kimbrough,Stefanie Robel,Erik Roberson,Harald Sontheimer (2015). Vascular amyloidosis impairs the gliovascular unit in a mouse model of Alzheimer’s disease.
B Zipser,C Johanson,L Gonzalez,T Berzin,R Tavares,C Hulette,M Vitek,V Hovanesian,E Stopa (2007). Microvascular injury and blood–brain barrier leakage in Alzheimer's disease.
J De La Torre,G Stefano (2000). Evidence that Alzheimer’s disease is a microvascular disorder: the role of constitutive nitric oxide.
Stavros Baloyannis (2015). Brain Capillaries in Alzheimer’s Disease.
Zhiyou Cai,Chuanling Wang,Wenbo He,Hanjun Tu,Zhengang Tang,Ming Xiao,Liang-Jun. Yan (2015). Cerebral small vessel disease and Alzheimer’s disease.
Kevin Richetin,Pascal Steullet,Mathieu Pachoud,Romain Perbet,Enea Parietti,Mathischan Maheswaran,Sabiha Eddarkaoui,Séverine Bégard,Catherine Pythoud,Maria Rey,Raphaëlle Caillierez,Kim Q Do,Sophie Halliez,Paola Bezzi,Luc Buée,Geneviève Leuba,Morvane Colin,Nicolas Toni,Nicole Déglon (2020). Tau accumulation in astrocytes of the dentate gyrus induces neuronal dysfunction and memory deficits in Alzheimer’s disease.
Ivan Maksimovich,Yuriy Polyaev (2010). P4‐073: The Importance of Early Diagnosis of Dyscircular Angiopathy of Alzheimer's Type in the Study of Heredity of Alzheimer's Disease.
Ivan Maksimovich (2012). Certain new aspects of etiology and pathogenesis of Alzheimer’s disease.
Raj Kalaria (2002). Small Vessel Disease and Alzheimer’s Dementia: Pathological Considerations.
W Brown,C Thore (2011). Review: Cerebral microvascular pathology in ageing and neurodegeneration.
Ivan Maksimovich (2015). Dementia and Cognitive Impairment Reduction after Laser Transcatheter Treatment of Alzheimer’s Disease.
Jack De La Torre (2016). Cerebral Perfusion Enhancing Interventions: A New Strategy for the Prevention of Alzheimer Dementia.
Seth Love,J Miners (2016). Cerebral Hypoperfusion and the Energy Deficit in <scp>A</scp>lzheimer's Disease.
Amy Nelson,Melanie Sweeney,Abhay Sagare,Berislav Zlokovic (2016). Neurovascular dysfunction and neurodegeneration in dementia and Alzheimer's disease.
Robert Bell,Berislav Zlokovic (2009). Neurovascular mechanisms and blood–brain barrier disorder in Alzheimer’s disease.
Axel Montagne,Samuel Barnes,Melanie Sweeney,Matthew Halliday,Abhay Sagare,Zhen Zhao,Arthur Toga,Russell Jacobs,Collin Liu,Lilyana Amezcua,Michael Harrington,Helena Chui,Meng Law,Berislav Zlokovic (2015). Blood-Brain Barrier Breakdown in the Aging Human Hippocampus.
I Maksimovich,M Lesnoy,V Zubov (1988). Transluminal laser angioplasty with low-intensity laser radiation.
Michael Hamblin,James Carroll,Lucas De Freitas,Ying-Ying Huang,Cleber Ferraresi (null). History of LLLT and Photobiomodulation.
R Michael,Ying-Ying Hamblin,Huang (2019). Photobiomodulation in the Brain.
Javad Hashmi,Ying‐ying Huang,Bushra Osmani,Sulbha Sharma,Margaret Naeser,Michael Hamblin (2010). Role of Low‐Level Laser Therapy in Neurorehabilitation.
Margaret Naeser,Michael Hamblin (2011). Potential for Transcranial Laser or LED Therapy to Treat Stroke, Traumatic Brain Injury, and Neurodegenerative Disease.
Michael Hamblin (2017). Mechanisms and Mitochondrial Redox Signaling in Photobiomodulation.
Anita Saltmarche,Margaret Naeser,Kai Ho,Michael Hamblin,Lew Lim (2017). Significant Improvement in Cognition in Mild to Moderately Severe Dementia Cases Treated with Transcranial Plus Intranasal Photobiomodulation: Case Series Report.
M Hamblin,M Hennessy (2017). Photobiomodulation and the brain: a new paradigm.
Michael Hamblin (2919). Photobiomodulation for Alzheimer’s Disease: Has the Light Dawned?.
Farzad Salehpour,Michael Hamblin,Joseph Diduro (2019). Rapid Reversal of Cognitive Decline, Olfactory Dysfunction, and Quality of Life Using Multi-Modality Photobiomodulation Therapy: Case Report.
Michael Hamblin (2019). Mechanisms of photobiomodulation in the brain.
Ivan Maksimovich (2019). Transcatheter intracerebral photobiomodulation in degenerative brain disorders: clinical studies (Part 1).
N Zagorodni,E Belyak,F Lazko,A Kubashev,A Prizov,M Lazko,I Grigoryev,T Skipenko,A Zakirova (2004). Experience of using subacromial balloon in combined treatment of patients with rupture of the rotator cuff.
I Maksimovich,L Gotman,S Masyuk (2006). Method of Determining Dimensions of Temporal Brain Lobes in Patients Suffering from Alzheimer's Disease.
I Maksimovich,L Gotman (2006). Short-term clinical outcomes for stages of NIA-AA preclinical Alzheimer disease.
I Maksimovich (2006). Method for Endovascular Treatment of Alzheimer's Disease.
Ivan Maksimovich (2006). P4‐117: Results of Alzheimer'S Disease Endovascular Treatment Method.
J Morris (1993). The Clinical Dementia Rating (CDR): Current Version and Scoring Rules.
Ivan Maksimovich (2012). The tomography dementia rating scale (TDR)—The rating scale of Alzheimer’s disease stages.
Ivan Maksimovich (2019). Intracerebral Transcatheter Laser Photobiomodulation Therapy in the Treatment of Binswanger's Disease and Vascular Parkinsonism: Research and Clinical Experience.
Shinji Matsunaga,Taro Kishi,Nakao Iwata (2015). Combination Therapy with Cholinesterase Inhibitors and Memantine for Alzheimer’s Disease: A Systematic Review and Meta-Analysis.
George Grossberg,Martin Farlow,Xiangyi Meng,Drew Velting (2015). Evaluating High-Dose Rivastigmine Patch in Severe Alzheimer’s Disease: Analyses with Concomitant Memantine Usage as a Factor.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Ivan V. Maksimovich. 2026. \u201cStimulation of Cerebral Angiogenesis and Neurogenesis by Transcatheter Intracerebral Laser Photobiomodulation Therapy in Alzheimers Disease\u201d. Global Journal of Medical Research - A: Neurology & Nervous System GJMR-A Volume 22 (GJMR Volume 22 Issue A2).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.