Efficient utilization of available resources is a promising research direction. In-depth studies can provide a unique platform for reducing fuel consumption while simultaneously reducing pollution, thereby avoiding environmental pollution and health hazards for this purpose various fuel addictive are being used now. A laser additive for liquid and gaseous fuel is yet to be developed. In this context, we successfully used the 2-6 mid-infrared spectrum as a fuel additive. To generate mid-infrared we invented a hand-lit pocket-size mid-infrared generating automizer (MIRGA). The trial fuels were irradiated with this spectral range, which caused chemical changes in the fuels.
## I. INTRODUCTION
Now, the automobile industry's urgent need is that internal combustion engines should consume less fuel produce more power, and also emit less pollutants (Krishania et al., 2020). On the contrary, emerging volatile fuel prices, economic policies, and war increased the number of vehicles and roads, thereby increasing pollution. The primary sources of air pollution are motor vehicle emissions and fossil fuel combustion (Kalghatgi et al., 2016). Comparatively diesel engines emit massive quantities of pollution which causes serious health (Dizziness to lung cancer) and environmental (global warming and acid rain, smog, etc.) hazards (Abdellatif et al., 2021; Daud et al., 2022). In spite of stringent measures, automobile pollution is a big challenge to our new technical world (Zhang et al., 2020). To overcome the hazards fuel component alteration, especially varieties of additives are in use but are to be improved.
The most used liquid fuels include diesel, gasoline, and kerosene. In developing countries, the most important household fuel is kerosene (Lam et al., 2012), contributing to the 4.3 million deaths that occur due to household air pollution (HAP) (Collins, 2014). Like other fuels, liquefied petroleum gas (LPG), an alternative fuel, has dynamic price increases and supply associated with high demand (Grand View Research, 2016).
Our technology of employing mid-IR is one of the new ways to overcome the said problems. Infrared wavelength is essential for earthly molecules. Daily received $66\%$ of the sun's radiant energy is infrared (Aboud et al., 2019). In the infrared spectrum mid-infrared (mid-IR) is the safest range (Prasad, 2005; Pereira et al., 2011) which penetrates most obscurants and coincides with nearly all molecules of Earth (Waynant et al., 2001; Toor et al., 2018), cause chemical bond changes, hence target substance's (fuels) physicochemical property alteration (Waynant et al., 2001; Tsai et al., 2017). We have invented a mid-infrared generating atomizer (MIRGA). In field and laboratory conditions, the tanked liquid and gaseous fuels were subjected to MIRGA irradiation. Their favorable efficiency and results are compared with the control (non-irradiated) and detailed here. We have also subjected the irradiated and non-irradiated fuels to instrumentations such as GC-MS, NMR, and FTIR and compared. Herein, we show that the comparatively MIRGA platform is safe, cost-effective, easy to use, and eco-friendly. Review literature showed that this laser fuel additive technology is the first of its kind to generate significant results.
## II. MATERIALS AND METHODS
### a) Design of Mid-Infrared Generating Atomizer (MIRGA)
MIRGA (patent no. 401387) is a 20-ml capacity polypropylene plastic atomizer containing a water-based inorganic solution (molar mass 118.44 g/mol) (containing approximately two sextillion cations and three sextillion anions). The atomizer has dimensions of $86 \times 55 \times 11 \, \text{mm}$, an orifice diameter of $0.375 \, \text{mm}$, an ejection volume of $0.062 \pm 0.005 \, \text{ml}$, an ejection time of
0.2 s, an average pressure of 3900 pascals, and a cone liquid back pressure of 2000 N/m2 (Fig. 1). Design of the MIRGA and emission of 2-6μm mid-IR has been presented in detail by Umakanthan et al., 2022a; Umakanthan et al., 2022b; Umakanthan et al., 2023c; Umakanthan et al., 2023d. Every time spraying emits 0.06ml which contains approximately seven quintillion cations and eleven quintillion anions.
 Panel label: Container.
 Panel label: Plunger.
 Panel label: Cap.
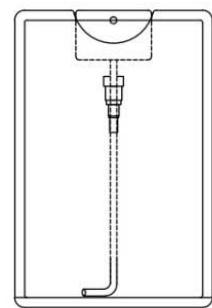
Container without cap
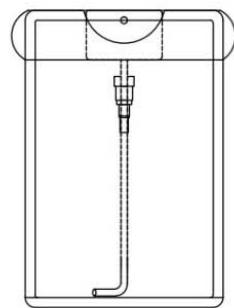
Container with cap Fig. 1: MIRGA's Design
The inorganic chemicals used in generation of mid-infrared are a perspective for biomedical applications (Tishkevich et al., 2019; Dukenbayev et al., 2019). This new method of synthesis the functional materials (mid-infrared) (Kozlovskiy et al., 2021; El-Shater et al., 2022). Different chemicals with excellent electronic properties leads to new composite material and has attracted great technological interest now (Kozlovskiy & Zdorovets, 2021; Almessiere et al., 2022).
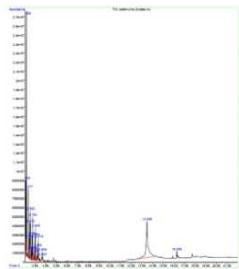
During spraying, approximately $1\mu \mathrm{g}$ of water as mist is lost, and the non-volatile material in the sprayed liquid is $153\mathrm{mg / ml}$. Depending on the pressure (varies with the user) applied to the plunger, every spray is designed to generate 2-6 $\mu \mathrm{m}$ mid-IR (Fig. 2) (Umakanthan et al., 2022a). Each spray emits $0.06\mathrm{ml}$ of solution, which contains approximately seven quintillion cations and eleven quintillion anions.
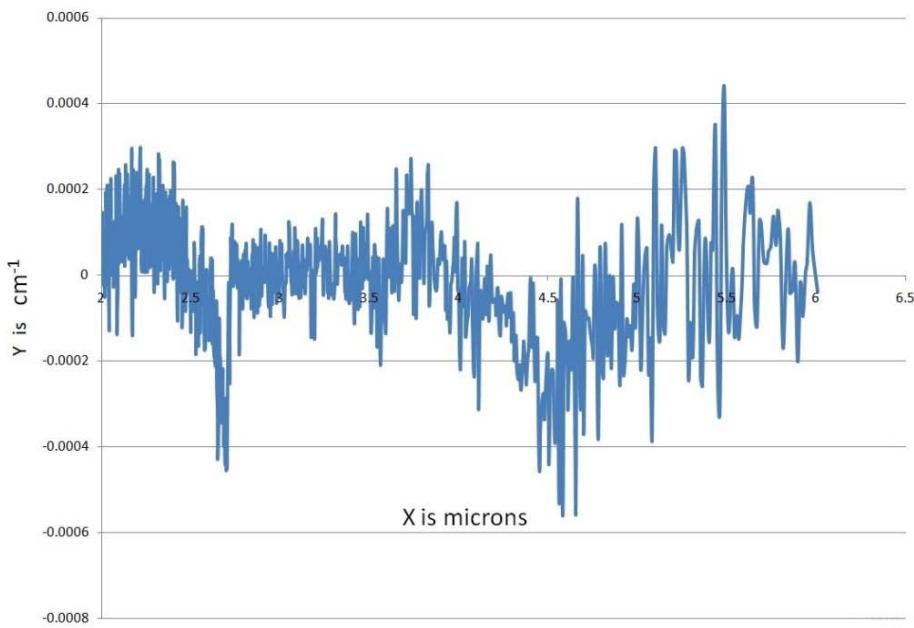 Fig. 2: Estimation of
$2 - 6\mu \mathrm{m}$ Mid-Infrared While Spraying MIRGA Atomizer
### b) Method of Mirga Spraying
The spraying should be done from the fuel tank mouth towards the fuel. This distance is essential for the MIRGA-sprayed solution to form ion clouds, to and fro oscillations, and generate mid-IR. The generated mid-IR can penetrate the intervening material—In an LPG iron cylinder—and act on the fuel contents inside (Fig. 3a, Fig. 3b) (Method of MIRGA spraying presented in Supplementary video V1).
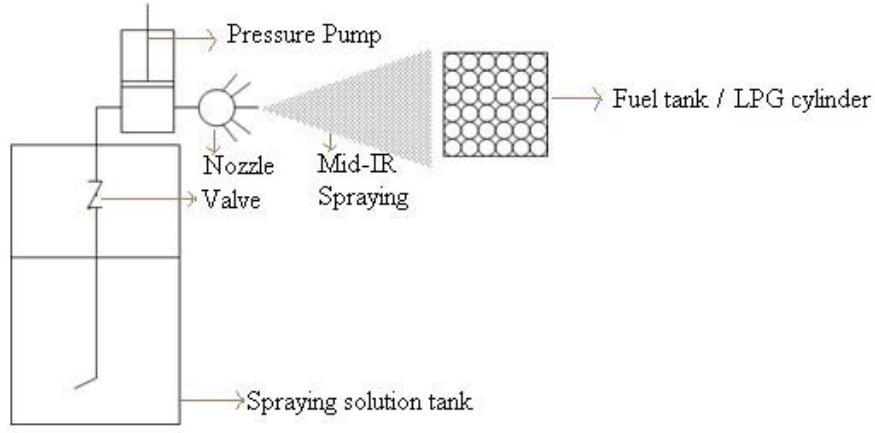
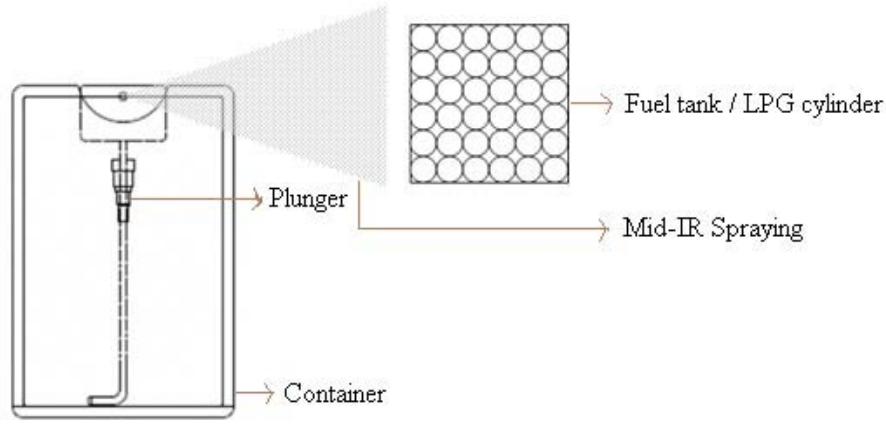 Fig. 3a: Schematic Apparatus of MIRGA Fig. 3b: Schematic Apparatus of MIRGA
### c) Vehicles Employed in the Study
Two, three, and four-wheeled vehicles, as well as multi-axle vehicles, of different brands, models, cylinders, horsepower, and manufacturing years, were employed. Nearly 500 such vehicles that have been operating on the road for more than a decade were tested with commercially available liquid fuels.
Kerosene-based equipment, viz., power generators, old model engines, and traditional lamps, was also filled with commercially available kerosene and tested. Commercial gasoline power generators and domestic LPG cylinders (14.2 kg) with stove burners were employed. The expert panel was comprised of 65 housewives $(n = 65)$. LPG experts from refineries also contributed to their outside opinion.
Diesel, gasoline, and kerosene samples were all taken from the same brand and batch, and different brands and batches were never mixed.
### d) Instrumentations Employed in the Study
Response variables and instruments included: Chemical compound transformation - Gas chromatography-mass spectrometry (GC-MS); Chemical bond changes - Fourier-transform infrared spectroscopy (FTIR); and Nuclear resonances - Proton nuclear magnetic resonance (1H-NMR).
GC-MS: Agilent technologies, 7820 GC system, 5977E MSD, Colomn DB-5, Over temperature $100 - 270^{\circ} \mathrm{C}$, Detector MS, Flow rate of 1.2, Carrier gas used was Helium.
FTIR: IR AFFINITY I - FTIR Spectrophotometer, FTIR 7600, Shimadzu
1H-NMR: The 1 H NMR spectra of the compounds were performed on a 500 MHz Bruker AVANCE III spectrometer operating at 500.13 MHz, using a 5-mm broad band (BBO) probe equipped with a z-gradient coil
(Bruker-Biospin, Switzerland). The samples were dissolved in CDCl 3. The chemical shifts $(\delta)$ were calibrated concerning TMS. All 1D spectra were acquired with 32K data points. Typical acquisition parameters for the 1 H NMR experiments were as follows: acquisition time 1.58 s, spectral width $10330\mathrm{Hz}$, pulse width $3.5\mu s$ (flip angle $\approx 30^{\circ}$ ), relaxation delay 1s, and number of scans 32.
## III. TRAILS CONDUCTED
### a) Diesel and Gasoline Trial
## i. Method I
Control - Each vehicle's fuel tank was filled with a specific brand and quantity of fuel and tested on different loads and road conditions. The specific fuel consumption (SFC), exhaust smoke, and other emissions were all recorded.
Trials - The protocol was the same as that of control, including the same vehicle. However, after filling with fuel before capping, MIRGA was sprayed into the tank via its mouth (then the tank was capped). The number of sprayings corresponding to the fuel was based on previous trial and error. For two and three-wheelers of below 20 liters of fuel - 1 spray for every 4 liters; for cars and SUVs of below 100 liters - 1 spray for every 10 liters; for heavy vehicles of above 100 liters - 1 spray for every 14 liters. The number of sprayings also depends on the engine model; usually, the estimated number may vary by one or two sprayings.
## ii. Method II
The same protocol as in Method I was followed in 35 and 40 table-mounted various brands of diesel and gasoline engines at laboratories and academic institutions, respectively.
### b) Kerosene Trial
## i. Method I
Each equipment's kerosene tank was filled with a specific brand and quantity of kerosene, and then it ran until the kerosene was exhausted and the running time was recorded (control group). For trials, after filling the same tank with the same brand and quantity of kerosene, MIRGA was sprayed into the tank via its mouth, and the same methods as the control were followed. The running times of control and trial were compared. The number of sprayings is as follows:
4-5 litres - 2 sprays
5-7 litres - 3 sprays
7-10 litres - 4 sprays
## ii. Method II
The same method was used in 12 table-mounted kerosene engines in labs and academic institutions.
### c) Electricity Trial
Control: The power generator was connected to a bottle containing $100 \mathrm{ml}$ of gasoline and ran until it shut down automatically.
Trials: The same power generator was connected to the same bottle containing $100 \mathrm{ml}$ of 1 MIRGA-sprayed gasoline and ran until it automatically stopped (first trial). Like this, in the second trial, 2 sprayings of $100 \mathrm{ml}$ of gasoline in the same bottle ran until they automatically stopped. Then, in the third trial, 3 sprayings of $100 \mathrm{ml}$ of gasoline in the same bottle were run until it automatically stopped.
In control and trials, time of running, power output, watt-hour (Wh), and kilowatt-hours (kWh) were calculated.
Though we used a variety of branded thermal (gasoline) power generators, the one that generated $28\%$ more electricity (model Z 36Z RO; model name EP1000; type RD) is discussed here. A 200-watt bulb was the load given to this generator. The marketed gasoline (petrol) was used as a thermal power source. For each control and trial study, the same brand and source of gasoline were used, i.e., for every trial (1 control and 3 trials), 5 liters of gasoline were kept as the source.
### d) LPG Trial
## i. Method I - Field trial
This method was tested for almost 5 years using nearly 800 LPG domestic cylinders in houses, hostels, hotels, and mass kitchens.
Control: A new domestic LPG cylinder was connected to a stove, the regulator knob was kept in "ON" mode, gas was lit, and then the burning flame color, density, height, and calorific value were all measured. It was then left for the consumer's routine use.
Trial: A domestic LPG cylinder was connected to a stove, and the same parameters as the control were measured. While the flame was burning, MIRGA was sprayed continuously 6 times around the cylinder from a distance of $0.25 - 0.50 \mathrm{~m}$. Then, burning flame color, density, height, and calorific value were measured, and it was then left for consumers' routine use. The control and trial cylinders' performance parameters were recorded and compared.
During our study, we increased the spraying number incrementally from 1 to 20. The trails were repeated several times, and 6 sprayings were found to be optimal for $14.2\mathrm{kg}$ and 9 sprayings for $19.5\mathrm{kg}$ LPG capacity cylinders.
## i. Method II - Laboratory trial
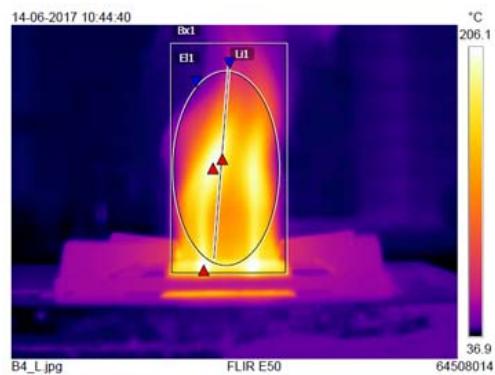
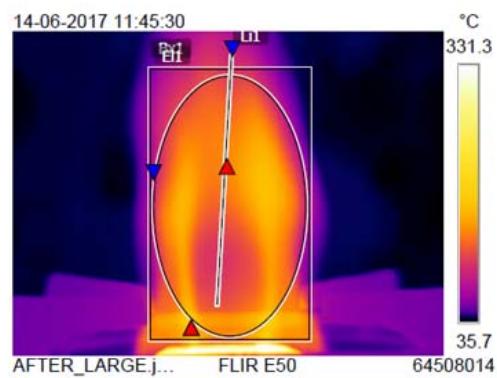
A non-sprayed (control) and 6 time-sprayed LPG cylinders (trials of same brand and weight) were simultaneously lit, and the regulator knobs were kept in ON mode and let to continuously burn until gas exhausted and flames were lost. During burning, the flames' parameters were recorded. This was repeated 6 times with 12 cylinders from the same batch. The temperatures of small and large flames before and after spraying were also measured and compared.
### e) Instrumentation Sampling Technique
To identify the chemical changes happening for every MIRGA spray, various instrumentations were performed. For this purpose, 4 samples of diesel and gasoline each $100\mathrm{ml}$ were taken. One formed a nonsprayed control; the other 3 trial samples correspondingly received 1, 2, and 3 sprayings. For kerosene, 5 samples were taken: one non-sprayed control and the other 4 trial samples correspondingly received 1, 2, 3, and 4 sprayings.
## IV. RESULTS
### a) Diesel and Gasoline
Table 1 and 2 respectively shows that the MIRGA irradiated diesel and gasoline has resulted in significantly reduced consumption and exhaust emissions besides reducing engine noise and smooth running within 5 minutes of on the road.
Table 1: Consumption and Emission Data - Diesel
<table><tr><td>Sl. No.</td><td>Exhaust</td><td>Result</td></tr><tr><td>1</td><td>Consumption</td><td>30-50% reduced</td></tr><tr><td>2</td><td>CO</td><td>20-61% reduced</td></tr><tr><td>3</td><td>CO2</td><td>1-29% reduced, in some vehicles increased</td></tr><tr><td>4</td><td>NOx</td><td>15-60% reduced</td></tr><tr><td>5</td><td>Oxygen</td><td>0.5-62% increase. In some vehicles, both CO2and O2emissions were found to increase.</td></tr><tr><td>6</td><td>HC</td><td>2-59% reduced, but in some vehicles increased</td></tr></table>
Table 2: Consumption and Emission Data - Gasoline
<table><tr><td>Sl. No.</td><td>Exhaust</td><td>Result</td></tr><tr><td>1</td><td>Consumption</td><td>12-58% reduced</td></tr><tr><td>2</td><td>CO</td><td>12-68% reduced</td></tr><tr><td>3</td><td>CO2</td><td>1-29% reduced</td></tr><tr><td>4</td><td>NOx</td><td>2-23% reduced</td></tr><tr><td>5</td><td>Oxygen</td><td>2-52% increased</td></tr><tr><td>6</td><td>HC</td><td>5-65% reduced, but some engines showed a slight increase</td></tr><tr><td>8</td><td>RPM</td><td>16% increased, some engines showed a slight decrease</td></tr></table>
### b) Kerosene
Depending on the instrument model, $35 - 80\%$ consumption is reduced.
### c) Electricity
Table 3: Comparison of Power Generation using Control and MIRGA Treated Gasoline
<table><tr><td>Before spraying (Control)Time of Running: 17.22 minFuel consumed: 100 ml</td><td>After 1 spraying (Trial)Time of Running: 22.08 minFuel consumed: 100 ml</td></tr><tr><td>Power output:P = V * I (Power = voltage * current)P = 200 watts, V = 230, I = 0.87 A200 W = 230V * 0.87 AWh = P * H (Watt hours = Power * Hours)P = 200 watts, H = 0.287 Hrs (17.22 min) = 200 * 0.287Wh = 57.4/100mlFor 1 Litre = 574 Wh(i.e) 0.574 kWh for one liter of petrol.</td><td>Power output:P = V * I (Power = voltage * current)P = 200 watts, V = 230, I = 0.87 A200 W = 230 V * 0.87 AWh = P * H (Watt hours = Power * Hours)P = 200 watts, H = 0.287 Hrs (22.08 min) = 200 * 0.368Wh = 73.6/100mlFor 1 Litre = 736 Wh(i.e) 0.736 kWh for one liter of Petrol.</td></tr></table>
Table 3 illustrates that the 1 sprayed gasoline produced $28\%$ more electricity compared to the control. The 2 and 3 sprayed samples generated less than $28\%$ electricity.
Tables 1, 2, and 3 demonstrated the benefits of $2 - 6\mu \mathrm{m}$ mid-IR on liquid fuels.
### d) For LPG
In trial cylinders after 6 sprayings, between 7 and 60 seconds the flame became dense, rose in height, and turned completely yellow (indication of MIRGA's action on LPG). This burning phenomenon was found to be not soot radiation emission because this occurred only when spraying was done on the trialed cylinders (some control and trial cylinders during burning showed very mild occasional soot radiation emission). After use, when cylinders are exhausted the duration of burning is calculated and compared between trial and control. In the trailed cylinders $28 - 35\%$ reduction in LPG fuel consumption was recorded (i.e. approximately a $30\%$ utility time increase) with no apparent pollution.
Six MIRGA sprayings given once were enough until a cylinder was exhausted and effects were found to have retained in LPG for 30-34 months (depending on the brand).
 Panel label: Before spray.
 Panel label: After spray.
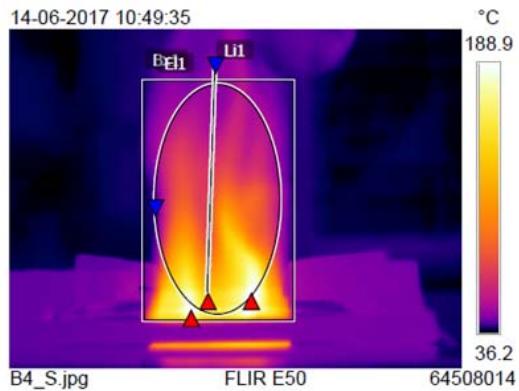
Fig. 4a: LPG Trial with Large-Sized Burner Before spray
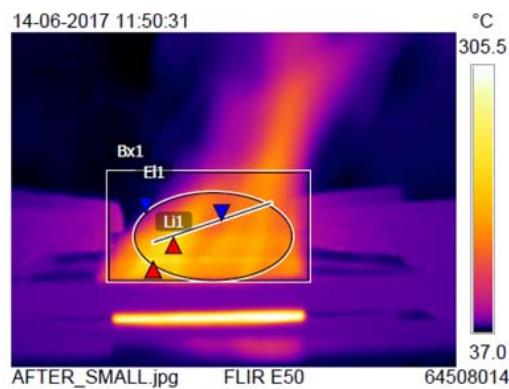
After spray Fig. 4b: LPG Trial with Small-Sized Burner
Table 4: Burner Trial Result Comparison
<table><tr><td rowspan="2">Burner type</td><td colspan="4">B x 1°C</td><td colspan="4">EL 1 (Elliptical flame) °C</td><td colspan="4">Li 1 (Linear flame) °C</td><td colspan="4">Whole flame temperature °C</td></tr><tr><td>Bs</td><td>As</td><td>Df</td><td>Imp%</td><td>Bs</td><td>As</td><td>Df</td><td>Imp%</td><td>Bs</td><td>As</td><td>Df</td><td>Imp%</td><td>Bs</td><td>As</td><td>Df</td><td>Imp%</td></tr><tr><td>Large sized burner</td><td>219</td><td>220</td><td>1</td><td>0.45</td><td>144</td><td>168</td><td>24</td><td>16</td><td>158</td><td>155</td><td>-3</td><td>-2</td><td>206</td><td>331</td><td>125</td><td>60</td></tr><tr><td>Small sized burner</td><td>202</td><td>234</td><td>32</td><td>15</td><td>99</td><td>171</td><td>72</td><td>73</td><td>84</td><td>177</td><td>93</td><td>110</td><td>188</td><td>305</td><td>117</td><td>62</td></tr></table>
From Table 4, compared to the non-sprayed cylinder, the sprayed LPG cylinder's large-sized burner flame temperature was found to be increased viz., elliptical flame $16\%$ and whole flame $60\%$, and linear flame $-2\%$. (Fig. 4a).
Compared to the non-sprayed cylinder, the sprayed LPG cylinder's small-sized burner flame temperature was found to be increased viz., elliptical flame $73\%$, linear flame $110\%$, and whole flame $62\%$. (Fig. 4b).
For the LPG field trial, please view: https://drive.google.com/file/d/1r-no10foxaOD_W7fvuscJ5Yj-aGXP_n/view
 Panel label: Control.
 Panel label: I sprayed.
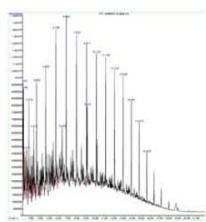
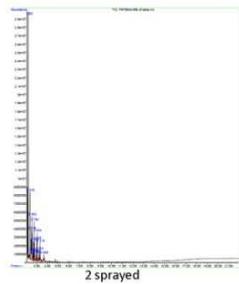
(a) GCMS - Diesel

 Panel label: Control.

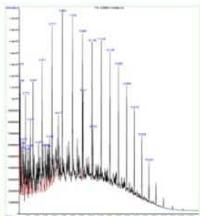
1 sprayed

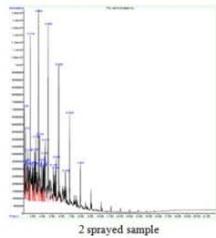
(b) GCMS - Gasoline

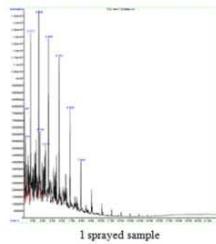

(c) GCMS - Kerosene
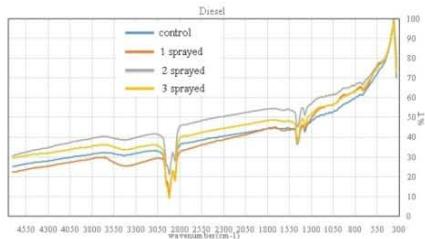
Fig. 5: GCMS Spectra (a) Diesel, (b) Gasoline, (c) Kerosene (a) FTIR - Diesel
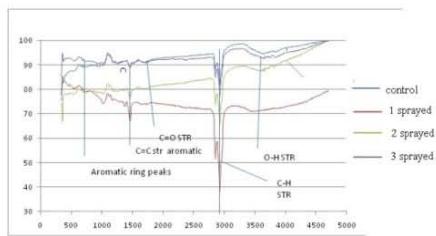
(b) FTIR - Gasoline
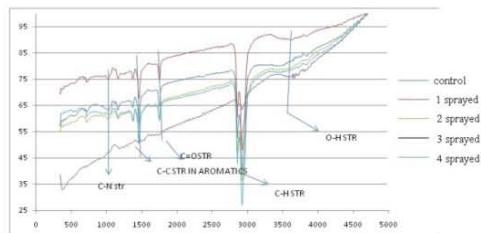
(c) FTIR - Kerosene
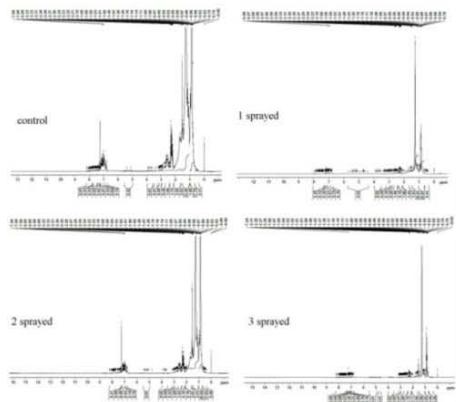
Fig. 6: FTIR Spectra (a) Diesel, (b) Gasoline, (c) Kerosene (a) NMR - Diesel
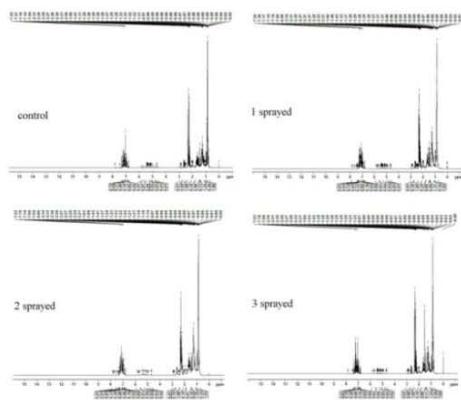
(b) NMR - Gasoline
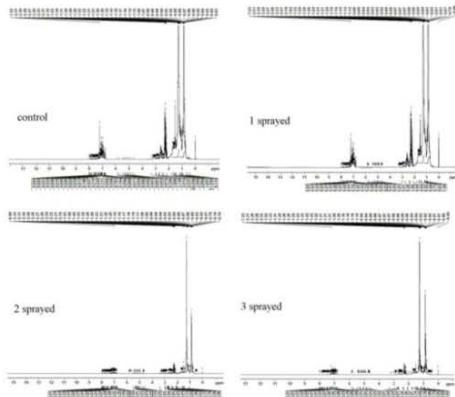
(c) NMR - Kerosene Fig. 7: 1H-NMR Spectra (a) Diesel, (b) Gasoline, (c) Kerosene
## V. INSTRUMENTATION RESULTS
(Raw data files of instrumentations for Diesel, Gasoline and Kerosene presented in Supplementary data D1).
### a) GC-MS
## i. GCMS - diesel
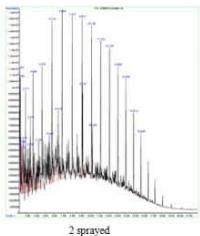
The control sample contained typical hydrocarbon components like Decane, Undecane, Tridecane, Tetradecane, Pentadecane, Hexadecane, Heptadecane, Octadecane, Nonadecane, Eicosane, Heneicosane, and Tetracosane. These peaks (with comparatively low content) were also presented in all the sprayed samples suggesting that the diesel samples have not changed their principle components after spraying. However, each spraying has generated a new unique peak in each sample and is responsible for corresponding changes. One sprayed sample has shown a unique peak of Tridecane, 6-cyclohexyl, while 2 sprayed samples have shown Pentacosane as a unique peak, and 1-H-Indene, 2,3-dihydro-4,7-dimethyl was the unique peak for 3 sprayed samples. (Fig. 5a)
## ii. GC-MS - gasoline
The control sample contains components like Benzene, 1-ethyl-2-methyl, Benzene, 1, 2, 3-trimethyl, Indane, o-cymene, and Oleic acid as major products. The peak of Benzene, 1, 2, 3-trimethyl (high in content) was also presented in all the sprayed samples suggesting that the petrol samples have undergone considerable changes its components after spraying. Additionally, spraying has generated several new unique peaks Naphthalene, 1-methyl, Indane, 1-methyl, etc. in a respective sample and is responsible for corresponding changes. The 1 sprayed sample has shown an increase in the peak of O-Cymene, Indane, etc., while 2 sprayed samples showed Naphthalene as a unique peak and Naphthalene, 1-methyl, and Indane, 1-methyl were unique peaks for 3 sprayed samples. (Fig. 5b)
## iii. GC-MS - Kerosene
The kerosene control sample contained typical components like Decane derivative, Undecane, Dodecane, Nanone derivative, Triodecane, Tetradecane, Pentdecane, Hexdecane, etc. All these peaks (with comparatively low and high content) were also presented in all the sprayed samples suggesting that the kerosene samples have undergone considerable changes in their components after spraying. Additionally, all the sprayings have generated several new unique peaks like Decane, 3-methyl, Tridecane, 7-methyl, 1-hexadecanol, and 1-hexadecanolin a respective sample and could be responsible for corresponding changes. The 1 sprayed sample showed unique of Dodecane and Tridecane, 7-methyl-while 2 sprayed sample showed a higher peak of Decane, 2-methyl and Undecane, 2,6-dimethyl than control and
Decane, 3,6-dimethyl was a unique peak in 3 sprayed sample. (Fig. 5c)
### b) FTIR
## i. FTIR - Diesel
The main bands of the spectra originated from saturated, aliphatic compounds as they represent most of the molecules present in the sample. These bands (the ones between 3000-2800 $\mathrm{cm}^{-1}$, and the ones between 1450-1350 $\mathrm{cm}^{-1}$ ) show very similar transmittance values in control, 1, and 3 sprayed samples, pointing to comparable concentrations. In 2 sprayed samples, those bands show a significantly higher transmittance (lower absorption), indicative of a lower concentration of the molecules contributing to them. Regarding the transmittance of the baseline, behind which some bands coming from minor components are present, the absorption (concentration) decreases following this order: 1 sprayed $>$ control $>$ 3 sprayed $>$ 2 sprayed. This observation indicates that 1 spraying causes an augmentation in the concentration of some components of the sample. However, upon successive sprayings a reduction of the concentration takes place (with 2 spraying) and, somehow, concentration is partially recovered (with 3 sprayings). (Fig. 6a)
Since diesel is a mixture of many different hydrocarbons, changes observed in the properties of the samples are related to variations taking place in the ratio of those hydrocarbons. For the same kind of hydrocarbon, a higher number of carbon atoms leads to a higher heating value. The effect of mid-IR spraying favors the loss of the more volatile compounds (this is, those with lower molecular mass and therefore lower number of carbons). So, as the sample is more and more sprayed, the concentration of hydrocarbons with a higher number of carbons increases, and the heating value of the sample rises leading to a lower consumption. For the same reason, these changes in composition could improve combustion and thus reduce the pollutants produced as suggested before.
## ii. FTIR - Gasoline
A broad peak due to O-H stretching at 3400-3600 $\mathrm{cm}^{-1}$ is observed. This indicates the presence of the phenolic group. C-H stretching at $2924~\mathrm{cm}^{-1}$ due to $-\mathrm{CH}_2$, $\mathrm{CH}_3$ of saturated hydrocarbon. The peak at 1700 $\mathrm{cm}^{-1}$ is due to $\mathbf{C} = \mathbf{O}$ stretching which overlaps in the control sample and 3 sprayed samples. The peak at $1465~\mathrm{cm}^{-1}$ is due to $\mathbf{C} = \mathbf{C}$ str in the aromatic ring. The peak at $748~\mathrm{cm}^{-1}$ is due to aromatic rings which are more intense in 1 and 2 sprayed samples than control. The increased intensity of the $\mathbf{C} = \mathbf{C}$ stretching at 1465 $\mathrm{cm}^{-1}$ in sample 1 sprayed and 2 sprayed samples, and increased intensity of $-\mathrm{C - H}$ stretching in all the sprayed samples. Compared to the control indicates that photochemical transformation is happening and polycyclic aromatic hydrocarbons are formed from benzene derivatives. The higher intensity of polycyclic aromatic hydrocarbon makes the sprayed sample more homogeneous and better quality compared to the control. (Fig. 6b)
## iii. FTIR - Kerosene
There is a broad peak due to O-H stretching at $3400 - 3600\mathrm{cm}^{-1}$ which indicates the presence of the phenolic group. The peak intensity due to O-H str is decreased in 2, 3, and 4 sprayed samples in comparison to control. However, the peak intensity is higher in 1 sprayed sample. The peak at $2854.64\mathrm{cm}^{-1}$, $2924\mathrm{cm}^{-1}$, and $2954\mathrm{cm}^{-1}$ is due to C-H stretching of $-\mathrm{CH}_2$, $\mathrm{CH}_3$ of saturated hydrocarbons. The intensity of this peak is higher in 4 sprayed samples and is decreased in 3 sprayed samples. The peak at $1751\mathrm{cm}^{-1}$ is due to C=O stretching which disappears in 3 sprayed samples. The peak at $1465\mathrm{cm}^{-1}$ is due to C-C str in the aromatic ring which is not present in 3 sprayed samples. The peak at $1188\mathrm{cm}^{-1}$ is due to C-O str being found in all except 3 sprayed samples. This peak overlaps in 2 and 4 sprayed samples. The peak at $748\mathrm{cm}^{-1}$ is removed in 3 sprayed samples which indicates the amount of unsaturation is decreased after 3 sprayings. (Fig. 6c)
The increased intensity of $-\mathrm{C - H}$ stretching in all samples. Compared to control indicates that photochemical transformation[17] is happening and mono-substituted and para-substituted benzene molecules are converted to polycyclic aromatic hydrocarbons.
### c) Proton NMR Spectra
## i. Proton NMR - Diesel
Significant variations in the integral values of some regions are observed, pointing to changes in the concentration of some chemical species. If the most volatile compounds are reduced upon MIRGA spraying, the signals originated by them in the NMR spectra will have a lower integral value. The most volatile compounds are expected to be aliphatic molecules with a low number of carbons and thus their signals will be located between 0.3 and 2.1 ppm. Unfortunately, it is difficult to observe a clear diminution of the integral value, because of the high overlapping. The high number of present species causes that in every region signals of very diverse molecules are present (Fig. 7a). For example, in the aliphatic region not only the signals from simple aliphatic molecules present but also aliphatic moieties from more complex hydrocarbons are also there. For this reason, it is a complex task to drag a clear correlation between changes observed in sample properties and variations in the integration of NMR signals. However, those changes are directly related to changes in the concentration of the present chemical species and undoubtedly this has an impact on the proportion between diesel components that ultimately affects its properties.
## ii. Proton NMR - Gasoline
The 1H NMR spectra reveal the presence of a three-proton singlet at $\delta 2.2$ for a $\mathrm{CH}_3$ group on an aromatic ring, two peaks each of three-proton intensity at $\delta 0.8 - 0.9$ for $\mathrm{CH}_3$. It also shows a group at $\delta 1.2$. The $\mathrm{CH}_3$ group resonances are attributed to the different $\mathrm{CH}_3$ groups. To distinguish between the 3 subsamples, the peak integral of each sample was normalized. The number of $\mathrm{CH}_3$ aliphatic groups is the same in all samples. However, there is a reduction in the number of $\mathrm{CH}_3$ aromatic upon MRGA spraying i.e. $50\%$ reduction from 4 in the Control to 2 in all the sprayed samples (Fig. 7b). This suggests changes in the aromatic component which could be responsible for the reduced pollutant in gasoline.
## iii. Proton NMR - Kerosene
The 1H NMR spectra reveal the presence of a three-proton singlet for the $\mathrm{CH}_3$ group in aromatic rings, and the peak of three-proton intensity at $\delta 0.9$ for $\mathrm{CH}_3$. It also shows the $\mathrm{CH}_2$ group at $\delta 1.2$. The $\mathrm{CH}_3$ group resonances are attributed to the different $\mathrm{CH}_3$ groups. In order to distinguish between the 3 sub-samples, the peak integral of each sample was normalized. The number of $\mathrm{CH}_3$ aliphatic groups is the same in all samples. However, there is a clear reduction in the number of $\mathrm{CH}_3$ aromatic upon MIRGA spraying (reduced significantly from 8 in the Control sample to 1 in all the sprayed samples) (Fig. 7c). This suggests changes in the aromatic component which could be responsible for the reduced pollutant in the kerosene.
Compared with control data, all the instrumentation data suggested that MIRGA spraying has altered chemical bonding, chemical composition, configuration, and compound transformation leading to alteration in molecular characteristics.
## VI. BENEFITS AND FUTURE PROSPECTS OF MIRGA
1. An average of $30\%$ of the natural resource has been demonstrated to be saved, and associated pollution is reduced.
2. Clear restoration of a cleaner environment and health issues reduction.
3. Efficient engine functioning and found to operate smoothly.
4. Old motor engines performed nearly as well as recent models in fuel consumption and toxic emission reduction.
5. One spraying series is enough for an entire fuel tank/LPG cylinder until exhausted.
6. Increased electricity generation, enhancing economic efficiency.
7. More utility days of LPG hence economy.
## VII. DISCUSSION
### a) Action of MIRGA emitted 2-6 $\mu$ m mid-IR on Fuels
MIRGA was designed to generate $2 - 6\mu \mathrm{m}$ mid-IR and alter targets chemical bond parameters thereby to produce more beneficial effects Umakanthan et al., 2022a; Umakanthan et al., 2022b, Umakanthan T, Mathi M, 2023c) (detailed discussion presented in Supplementary Text T1). In this research, we observed that MIRGA spraying in diesel has caused chemical bond changes, and increased hydrocarbon concentration, thereby improving combustion, and hence reducing pollution. In gasoline, MIRGA spraying has caused O-H, C=C, C=O, and C-H chemical bond stretches, photodissociation, photochemical transformation, and formation of polycyclic aromatic hydrocarbons from benzene derivatives, more homogenation leading to better quality compared to the non-sprayed control. With just a little experience, a driver can identify his vehicle's spraying number requirement concerning fuel quantity.
The composition/ properties of the hydrocarbons determine the performance and emission of the internal combustion engine. Fuel additives influence the properties of the fuels hence additive research dynamic. Gaseous, liquid, and solid (Metal and carbon-based) additives are now in use. Using these additives in diesel and gasoline engines various studies were done as cited by (Abdellatif et al., 2021; Daud et al., 2022). They used B20, diesel, biodiesel, diesel ethanol, diesel methanol, etc. in diesel engines; and bio ethanol, prenol, furan mixture, dimate (isohexane), isooctene (di-isobutylene) in gasoline engine. And full load with different RPM, constant speed, and different blends with various load were employed. They studied the performance viz power, BTE, BSFC, and torque. The net emission result was inconsistent with their limitations. Among all additives tried to date Graphene nanoplatelets additive is found to be promising but this research is still insufficient (Daud et al., 2022) engine hybridization (Schifter et al., 2020) biofuel, electric vehicle (Pattanaik et al., 2017 and Cano et al., 2018) studies also showed inconsistent result and limitations. Turbocharging is a better technology but it has increased the demands on the detonation resistance of fuel (Alabas et al., 2020).
Comparing these studies, MIRGA techniques also seem to be favorable hence may be placed as one of the fuel additive. It also seems that except for MIRGA technology no literature or techniques are available to improve the electricity generation and LPG utility days. MIRGA sprayer is user-friendly and economical. A MIRGA sprayer that emits 300 sprayings approximately costs USD 0.3.
## VIII. CONCLUSION
In summary, we have shown that applying 2-6 $\mu$ m wavelength range mid-infrared rays to liquid and gaseous fuels. The mid-IR caused photode gradation of the fuels. There by considerably lower their overall consumption and simultaneously associated pollution at affordable cost. An average of $30\%$ of the natural resource has been demonstrated to be saved. Furthermore, irradiated gasoline generated more (28%) electricity. This technology is demonstrated to be safe and economical for practical use, as well as beneficial to the environment and reduces human health risks. In the future unique features of MIRGA technology and research on similar resources may shed more light on potential avenues for manipulating fuels more desirable.
### Conflict of Interest
In accordance with the journal's policy and our ethical obligation as researchers, we submit that the authors Dr. Umakanthan and Dr. Madhu Mathi are the inventors and patentee of Indian patent for MIRGA (under-patent no.: 401387) which is a major material employed in this study.
#### Funding
This study received no specific funding.
Supplementary data D1: Raw data files of instrumentations for Diesel, Gasoline and Kerosene https://drive.google.com/open?id=1335uu4v87jy7LE2J v3VxkoqbIFQ0V6Qt.
Supplementary video V1: Method of MIRGA spraying https://drive.google.com/open?id=1QoRwTESKfSdoJTf D--xIG9YpTDnVonGW.
Supplementary Text T1: Detailed Discussion
#### 1. Detailed Discussion [1]
#### 1.1 Invention Background
The four observable states of matter (solid, liquid, gas, and plasma) are composed of intermolecular and intramolecular bonds. The inherent characteristics of neutrons, protons and electrons are unique, however, differences in their numbers are what constitute different atoms, and how these atoms bind together develops into different molecules with unique characteristics. In the electromagnetic wave (EMW) spectrum, the mid-IR region is vital and interesting for many applications since this region coincides with the internal vibration of most molecules [2]. Almost all thermal radiation on the surface of the Earth lies in the mid-IR region, indeed, $66\%$ of the Sun's energy we receive is infrared [3] and is absorbed and radiated by all particles on the Earth. At the molecular level, the interaction of mid-IR wavelength energy elicits rotational and vibrational modes (from about 4500–500 cm $^{-1}$, roughly 2.2 to 20 microns) through a change in the dipole movement, leading to chemical bond alterations [4].
During our research we have observed: (A) In all objects, even though atoms always remain as atoms, their chemical bond parameters are continuously prone to alteration by cosmic and physical energies (e.g.: EMW, heat, pressure, and humidity) causing the bonds to compress/stretch/bend [5-8], break [9,10], or new bonds to be formed [11]. These alterations ultimately lead to changes in the physicochemical characteristics of the objects. (B) The dynamic, constant, and mutual influences of EMW among the Earth and the celestial and living bodies are continuously causing alterations in the inherent physiochemical characters of earthly objects, for instance, enhancement due to an optimum dose of energy or decrease/destruction due to a high dose of energy (detailed below). Thus, based on these concepts, MIRGA was developed to alter the bond parameters, thereby potentiating the natural characteristics of products.
#### 1.2 MIRGA Definition
We define MIRGA as 'a harmless, economical atomizer containing an imbalanced ratio of ions suspended in water, which influence the natural potency of target substances by generating mid-IR while spraying'.
#### 1.3 Technique of Mid-IR Generation from MIRGA
We designed MIRGA as to accommodate an imbalanced ratio of ions suspended in water in their fundamental state, which can move as free particles. The solution exhibits very little detectable background frequency, below even that of cosmic events. By comparison humans emit more radioactivity (around 10 microns) [12, 13]. We designed MIRGA to generate energy based on various processes such as: (A) spraying leads to ionization (electrons getting separated from atoms) and many pathways for electron reabsorption; due to these two oscillatory processes, energy is generated; (B) while spraying, a water-based ionic solution gets excited/charged, which in turn leads to oscillation among the imbalanced ions [14] in their excited state, resulting in the emission of photons [15,16]; (C) although a low electromagnetic field exists between the charged particles of the MIRGA's ionic solution, during spraying the induced oscillation between these charged particles produces energy [17-21]; and $(D)$ in the natural rainfall process, more energy is required to break the water bonds for creating smaller water droplets [22]. Therefore, these droplets should have more stored energy, which then travels down at velocity from a specific distance, thus gaining kinetic energy. When the rain hits the Earth's surface, it forms a very thin film of mid-IR (nearly 6 micron), hence there is a net heat gain [22, 23]. We simulated this rainfall's energy-gaining process in MIRGA (i.e., when imbalanced ions in liquid media are atomized, the ejected smaller droplets should have higher internal energy as well as acquired kinetic energy, and the energy emitted by breaking the surface tension). From trial and error, we calibrated the ejection pressure to obtain a desired fine mist, and minimized the evaporation rate by altering the pH and density of the solution. Moreover, the accelerated ions in the sprayed ionic clouds collide among themselves and generate energy [24], thus, we incorporated these phenomena in our atomizer and designed it in such a way as to emit energy in the $2 - 6\mu \mathrm{m}$ mid-IR depending on the given plunger pressure.
Yousif et al. [25] described this process as a photo dissociation of molecules caused by the absorption of photons from sunlight, including those of infrared radiation, visible light, and ultraviolet light, leading to changes in the molecular structure.
#### 1.4 Safety of MIRGA-Sprayed Products
In our nearly two-decades of research, we have observed that MIRGA-induced bond-altered target substances do not show any adverse reaction upon consumption/use. In nature, (A) Stereochemical configuration has great influence on taste [26](e.g., varieties of mango, grapes, rice, etc.), (B) Cooking and digestive enzymes break chemical bonds, thereby softening foods. This indicates that alterations in chemical bonds occur naturally and do not represent a risk to human health. As an example, boiled rice, puffed rice, flat rice, and rice flour have a unique aroma, taste, texture, and shelf-life but conserving the same molecular formula $(\mathsf{C}_6\mathsf{H}_{10}\mathsf{O}_5)$. (C) In the food industry, sensory attributes and shelf-life are enhanced by altering the food's chemical bonds using various irradiation processes like radappertization, radicidation, and radurization [27]. (D) Upon heating, water changes from ice to liquid to steam, which are manifestations of changes in the hydrogen bonds [28] but the chemical composition $(\mathsf{H}_2\mathsf{O})$ remains the same [29].
#### 1.5 MIRGA's Primeval and Future Scope
The water-based MIRGA could be the first novel potentiating technology. This type of atomizer technology also seems to be present with the extraterrestrials for their therapeutic use during visitations [30].
In various products, we have achieved a range from $30\%$ to $173\%$ potentiation. Even the smaller improvement resulted in $30\%$ monetary and resource savings as well as health benefits. However, there is a knowledge gap between potentiation from $30\%$ to at least $100\%$ for all products, which can be filled-up by refining MIRGA's ionic solution, concentration, atomizer pressure, and other parameters and even formulating a better solution.
Various mid-IR emitters are now available (e.g., silicon photonic devices [31], cascade lasers quantum and interband [32], non-cascade-based lasers, chalcogenide fiber-based photonic devices [33], and suspended-core tellurium-based chalcogenide fiber photonic devices [34]). These emitters are not as cost-effective as MIRGA and are useful only in astronomy, military, medicine, industry, and research applications. These emitters are too complex for domestic application by the average user.
Because of MIRGA's wide range of applications, we believe that this technique will resonate in many scientific fields including biophotonics, therapeutics, health, ecology, and others. We are currently conducting research on MIRGA and its applications, namely MIRGA salt, MIRGA vapor and MIRGA plasma.
Generating HTML Viewer...
References
64 Cites in Article
Narendra Krishania,Upendra Rajak,Tikendra Nath Verma,Anil Kumar Birru,Arivalagan Pugazhendhi (2020). Effect of microalgae, tyre pyrolysis oil and Jatropha biodiesel enriched with diesel fuel on performance and emission characteristics of CI engine.
Gautam Kalghatgi,C Gosling,M Wier (2016). The outlook for fuels for internal combustion engines.
Tamer Abdellatief,Mikhail Ershov,Vladimir Kapustin,Mohammad Ali Abdelkareem,Mohammed Kamil,A Olabi (2021). Recent trends for introducing promising fuel components to enhance the anti-knock quality of gasoline: A systematic review.
Sarbani Daud,Mohd Hamidi,Rizalman Mamat (2022). A review of fuel additives' effects and predictions on internal combustion engine performance and emissions.
W Zhang,X Ma,S Shuai,K Wu,J Macias,Y Shen (2020). Effect of gasoline aromatic compositions coupled with single and double injection strategy on GDI engine combustion and emissions.
Nicholas Lam,Kirk Smith,Alison Gauthier,Michael Bates (2012). Kerosene: A Review of Household Uses and their Hazards in Low- and Middle-Income Countries.
K Collins (2014). Kerosene Lamps are an important Target for reducing Indoor Air pollution and climate Emissions.
LPG Market Size Projected To Reach.
Billion By (2016). Unknown Title.
Salam Aboud,Ammar Altemimi,Asaad R. S. Al-Hiiphy,Lee Yi-Chen,Francesco Cacciola (2019). A Comprehensive Review on Infrared Heating Applications in Food Processing.
Narasimha Prasad (2005). Optical communications in the mid-wave IR spectral band.
M F Pereira,O Shulika (2011). Terahertz and Mid Infrared Radiation: Generation, Detection and Applications.
R Waynant,I Ilev,I Gannot (2001). Mid-infrared laser applications in medicine and biology.
Fatima Toor,Stuart Jackson,Xiaoming Shang,Shamsul Arafin,Haining Yang (2018). Mid-infrared Lasers for Medical Applications: introduction to the feature issue.
Shang-Ru Tsai,Michael Hamblin (2017). Biological effects and medical applications of infrared radiation.
Umakanthan,Madhu Mathi (2022). Decaffeination and improvement of taste, flavor and health safety of coffee and tea using mid-infrared wavelength rays.
T Umakanthan,M Mathi (2022). Quantitative reduction of heavy metals and caffeine in cocoa using mid-infrared spectrum irradiation.
Thangaraju Umakanthan,Madhu Mathi (2023). Increasing saltiness of salts (<scp>NaCl</scp>) using mid‐infrared radiation to reduce the health hazards.
Madhu Umakanthan,Mathi (2023). Potentiation of Siddha medicine using Muppu (Universal Potentiator).
D Tishkevich,I Korolkov,A Kozlovskiy,M Anisovich,D Vinnik,A Ermekova,A Vorobjova,E Shumskaya,T Zubar,S Trukhanov,M Zdorovets,A Trukhanov (2019). RETRACTED: Immobilization of boron-rich compound on Fe3O4 nanoparticles: Stability and cytotoxicity.
Kanat Dukenbayev,Ilya Korolkov,Daria Tishkevich,Artem Kozlovskiy,Sergey Trukhanov,Yevgeniy Gorin,Elena Shumskaya,Egor Kaniukov,Denis Vinnik,Maxim Zdorovets,Marina Anisovich,Alex Trukhanov,Daniele Tosi,Carlo Molardi (2019). RETRACTED: Fe3O4 Nanoparticles for Complex Targeted Delivery and Boron Neutron Capture Therapy.
A Kozlovskiy,M Zdorovets (2021). Effect of doping of Ce4+/3+ on optical, strength and shielding properties of (0.5-x)TeO2-0.25MoO-0.25Bi2O3-xCeO2 glasses.
A Kozlovskiy,M Zdorovets (2021). Effect of doping of Ce4+/3+ on optical, strength and shielding properties of (0.5-x)TeO2-0.25MoO-0.25Bi2O3-xCeO2 glasses.
Artem Kozlovskiy,Alua Alina,Maxim Zdorovets (2021). Study of the effect of ion irradiation on increasing the photocatalytic activity of WO3 microparticles.
Reda El-Shater,Hassan El Shimy,Samia Saafan,Moustafa Darwish,Di Zhou,Alex Trukhanov,Sergei Trukhanov,Fatma Fakhry (2022). Synthesis, characterization, and magnetic properties of Mn nanoferrites.
M Almessiere,N Algarou,Y Slimani,A Sadaqat,A Baykal,A Manikandan,S Trukhanov,A Trukhanov,I Ercan (2022). Investigation of exchange coupling and microwave properties of hard/soft (SrNi0.02Zr0.01Fe11.96O19)/(CoFe2O4)x nanocomposites.
I Schifter,L Díaz,G Sánchez-Reyna,C González-Macías,U González,R Rodríguez (2020). Influence of gasoline olefin and aromatic content on exhaust emissions of 15% ethanol blends.
B Pattanaik,J Jena,R Misra (2017). The effect of oxygen content in soapnut biodiesel-diesel blends on performance of a diesel engine.
Zachary Cano,Dustin Banham,Siyu Ye,Andreas Hintennach,Jun Lu,Michael Fowler,Zhongwei Chen (2018). Batteries and fuel cells for emerging electric vehicle markets.
B Alabas¸,G Tunç,M Tas¸tan,˙i Yilmaz (2020). Experimental investigation of the oxygen enrichment in synthetic gases flames.
Umakanthan,Madhu Mathi (2022). Decaffeination and improvement of taste, flavor and health safety of coffee and tea using mid-infrared wavelength rays.
Majid Ebrahim-Zadeh (2019). Compact, High-Power, Fiber-Laser-Based Coherent Sources Tunable in the Mid-Infrared and THz Spectrum.
A Salam,A Ammar,L Asaad,C Yi-Chen,Francesco (2019). Molecules A Comprehensive Review on Infrared Heating Applications in Food Processing.
J Girard (2014). Contaminant Behavior in the Environment: Basic Principles.
James Girard (2014). Principles of Environmental Chemistry.
Ravi Dwivedi,Shankar (2017). Remote Sensing of Soils.
Jag Mohan (2004). Book Reviews William A. Vogely, ed.; Economics of the Mineral Industries, 4th edition. New York: American Institute of Mining, Metallurgical and Petroleum Engincers, 1985. vi + 660 pages. No index.Susan Owens, Energy, Planning ond Urban Form. London: Pion Ltd., 1986, 116 pages. (In the US. and Canada from Methuen Inc., New York.)Mohan Munasinghe, Rural Electrification for Development-Policy Analysis and Applicalions. Boulder, Colorado: Westview Press, 1987Khong, Cho Oon, The Politics of Oil in Indonesia: Foreign Company-Host Government Relations. Cambridge, England: LSE Monographs in International Studies, Cambridge University Press, 1986, 253 pages.Jeffrey A. Dubin, Consumer Durable Choice and the Demand for Electricity. Amsterdam, New York and Oxford: North Holland, 1985. Distributed by Elsevier Science, New York. 265 pages.William A. Donnelly, The Econometrics of Energy Demand: A Survey of Applications. New York, Westport and London: Praeger. 1987. 328 pages.Jacqueline Lang Weaver, Unitization of Oil and Gas Fields in Texas, A Study of Legislative, Administrative, and Judicial Policies. Washington, D.C.: Resources for the Future, 1986. 555 pp.Joseph P. Kalt and Frank C. Schuller, eds. Drawing the Line on Natural Gas Regulation: The Harvard Study on the Future of Natural Gas. Westport, Conn.: Quorum Books, 1987. 267 pages.Richard P. Mattione, OPEC's Investments and the International Financial System. Washington, D.C.: The Brookings Institution, 1985..
Carolyn Mcmakin (2011). Frequency specific Microcurrent in pain management E-book.
David Moss (2011). Contents.
Peter Raven,Linda Berg,David Hassenzahl (2012). Environment.
Frances Ashcroft (2000). Life at the Extremes: The Science of Survival.
Robert Sanders (2014). Revealing the Heart of the Galaxy.
Frank Verheest (2000). Waves in Dusty Space Plasmas.
Sun Keping,Yu Gefei (2004). Preface.
Pierre Fauchais,Joachim Heberlein,I Maher,Boulos (2014). Thermal Spray Fundamentals from Powder to Part.
Manfred Wendish,Jean-Louis Brenguier (2019). Airborne Measurements for environmental Research: Methods and Instruments.
Chandramani Kongbam,Singh (2009). Basic Physics.
Mathura Prasad (2017). Soul, God and Buddha in Language of Science.
Stephen Pople (1999). Complete Physics.
Roger Barry,Richard Chorley (1998). Atmosphere, Weather and Climate.
Eniday (2019). DIN EN ISO 11547:2019-02, Kleine Wasserfahrzeuge_- Schutz vor Start unter Last (ISO_11547:1994); Deutsche Fassung EN_ISO_11547:2018.
T Krishnakumar (2019). Application Of Microwave Heating In Food Industry.
Emad Yousif,Raghad Haddad (2013). Photodegradation and photostabilization of polymers, especially polystyrene: review.
L Kenneth,Katherine Williamson,Masters (2011). Macroscale and Microscale Organic Experiments.
B Sivasankar (2014). Food Processing and preservation, PHI Learning Private Limited.
Trevor Day (1999). Ecosystems: Oceans.
Kenneth Raymond (2010). General Organic and Biological Chemistry.
(1996). Contracts for field projects and supporting research on enhanced oil recovery. Quarterly technical progress report, October 1994--December 1994. Progress review No. 81.
(2012). Wochenrückblick: Google Books, Content ID, Verwaiste Werke.
D Jung,S Bank,M Lee,D Wasserman (2017). Next-generation mid-infrared sources.
A Sincore,Justin Cook,Felix & El Tan,A El Halawany,A Riggins,S Mcdaniel,G Cook,D Martyshkin,V Fedorov,S Mirov,L Shah,A Abouraddy,M Richardson,Kenneth Schepler (2018). High power single-mode delivery of mid-infrared sources through chalcogenide fiber.
Bo Wu,Zheming Zhao,Xunsi Wang,Youmei Tian,Nan Mi,Peng Chen,Zugang Xue,Zijun Liu,Peiqing Zhang,Xiang Shen,Qiuhua Nie,Shixun Dai,Rongping Wang (2018). Mid-infrared supercontinuum generation in a suspended-core tellurium-based chalcogenide fiber.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Umakanthan T. 2026. \u201cTechnology of fuel consumption and emission reduction, and enhanced electricity generation using mid-infrared rays – A Laser Additive\u201d. Global Journal of Science Frontier Research - I: Interdisciplinary GJSFR-I Volume 24 (GJSFR Volume 24 Issue I1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Efficient utilization of available resources is a promising research direction. In-depth studies can provide a unique platform for reducing fuel consumption while simultaneously reducing pollution, thereby avoiding environmental pollution and health hazards for this purpose various fuel addictive are being used now. A laser additive for liquid and gaseous fuel is yet to be developed. In this context, we successfully used the 2-6 mid-infrared spectrum as a fuel additive. To generate mid-infrared we invented a hand-lit pocket-size mid-infrared generating automizer (MIRGA). The trial fuels were irradiated with this spectral range, which caused chemical changes in the fuels.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.