Facilitating successful early and sustained remission from substance use disorder remains an extraordinarily difficult puzzle for both clients and supporting persons to navigate. Ninety years since Alcoholics Anonymous developed its twelve step, spiritual approach to recovery from the devastation of alcoholism, initiating and sustaining abstinence from addictive substances remains a tremendous challenge. While the 12-step community continues to support addicted persons at no cost through meetings and relationships, psychology, counseling and medicine are continuing to develop approaches for people to achieve recovery from addiction. To date, there is minimal convergence in these approaches leaving affected individuals with often confusing choices in seeking necessary recovery support. The unfortunate result is the predominant view of substance use disorder as a chronic relapsing disorder whereby relapse is embedded as fundamental to its nature. Greater societal stressors and increasingly dangerous substance access is resulting in death by overdose becoming a major public health issue.
## I. INTRODUCTION
In 1939, Alcoholics Anonymous (AA) World Services Inc. published its text which, remarkably, is still in broad use today and shows no signs of losing popularity. Within the profession of caring for people suffering from addictions of all types in residential and ambulatory care sites, the Twelve Steps still remain a gold or, at minimum, a comparison standard. On the other hand, the field of addiction counselling is emerging in response to significantly growing population needs. Addiction Counsellors have developed professional certifications and licensing through embracing well developed therapeutic modalities (CCPA, 2025) and the scientific method. For the most part, the two approaches are believed to complement each other; it is rare that any current addictions counsellor would dissuade a client from attending a free support group.
The two predominant approaches, counselling and twelve step programs differ significantly: AA clearly advocates its approach through the twelves steps as spiritual in nature (Big Book, Appendix II, 2001). Counselling and psychotherapy are the skilled and principled use of relationship to facilitate self-knowledge, emotional acceptance and growth and the optimal development of personal resources (CCPA, 2025). Counseling modalities have embraced the scientific method for both developing and assessing the effectiveness of its various modalities (Fordham, B. et al, 2021). However, robust long term outcomes studies have not yet taken place which clearly identify evidence for best approaches for addressing addiction since Project Match (Project Match Research Group, 1998).
Project Match was a multi-centre study conducted from 1989 to 1997 which has received considerable criticism of its methods in the scientific field (Walters, G., 2002). Regardless, it failed to show any significant difference among any counselling methods and 12 Step programs in outcomes: most enrolled alcoholics showed minimal change in their drinking patterns at the one-year point. Studies since in the areas of treatment outcomes for addictions have been significantly smaller in terms of participants and time length. Our own commissioned work of 2014 (Ashton, C. and Duffie, D., 2014) exploring potential best practices in residential treatment for addictions revealed very few articles, and those centres that did publish reliable outcomes showed a success rate around $20\%$ overall at one year for clients.
In the absence of clear directions for counseling best practices and a multitude of potential options, the treatment industry has currently defaulted to a position which now assumes that each person finds their own path into recovery. Choice of types of therapy is mainly at the client's direction and consensus with the counsellor. There has, however, been a potentially paradigm-shifting body of knowledge which has emanated from advanced brain imaging techniques (PET and functional magnetic resonance imaging (fMRI)) and microbiology research technologies over the past 20 years. What can be collectively termed the neuroscience of addiction has provided incredible insights into relatively uniform and consistent pathways of addiction in the limbic system of the brain. That being said, we have yet seen little of the potential of neuroscience incorporated into present day programs (Verdejo-Garcia, A., 2019).
Our experience in treatment has strongly indicated that those persons who do achieve sustained recovery attribute their success to one predominant ideology that leaves the other far secondary in terms of perceived impact. This may be a suboptimal outcome in many cases. We have developed a novel and extraordinarily successful methodology to addiction counselling whereby the two philosophies merge through the neuroscience of addiction. Counseling and educating clients in the neuroscience behind Step One of AA is an approach that clients find fascinating and immensely gratifying through finally understanding the reasons for the complex array of compulsive and self-destructive thoughts, emotions and behaviours in addiction. Effective in facilitating sustained remission for $83\%$ of clients who complete the 30-day program, understanding the science of recovery through progressive, healthy neuroadaptations allows anticipation of challenges and accelerates the process toward healthy, neurotypical function.
The fields of psychology, addiction medicine and addiction counselling continue to make strides through research and practice. Counselling modalities such as cognitive behavioural therapy (CBT), dialectical behavioural therapy (DBT), motivational interviewing (MI) and acceptance commitment therapy (ACT) have become more refined, formalized and have a growing literature base. Their specific application at the varying stages of change (Prochaska and DiClemente, 1983) has become better articulated.
Drawing on the extraordinary work published in the field, our Step One method uses a robust neuroscientific evidence base to unify current counseling modalities with AA's first step to develop a common, highly effective framework. Coming to terms with the detailed organicism of addiction allows a tangible comprehension of the seeming incorporeal influence of substances over affected persons. At the same time, our program stimulates broader existential questions in contemplating the complexity and terrible perfection of this disorder, merging with the spiritual approach of AA.
### a) Introduction to Applied Behavioural Neuroscience
Our development of Step One Therapy began with a consideration of AA's Step 1:
"We admitted we were powerless over alcohol-- that our lives had become unmanageable."
A difficult admission for people faces even more resistance when the basis for recovery according to the Big Book is dependence on higher power and a spiritual solution. While it still gains favour among many, an abstract solution to excruciatingly painful, tangible human issues often gathers only minor commitment and unfortunately does not work for the vast majority of persons who try the program (Project Match Research Group, 1998). On the other hand, the counselling profession, as advanced as it is becoming, has yet to produce robust evidence of better efficacy with addictionthan twelve step methods since Project Match. Current systematic reviews (Balandeh et al, 2021) of CBT applied to addictions treatment still refer to it as a 'promising practice.' Since Project Match, a thorough review of the literature still shows a paucity of studies whereby adding the two therapies may prove summative.
Years of attending AA and Narcotics Anonymous and listening to people's narratives convinced us that the first half of Step 1, 'we admitted we were powerless,' was true. Since the AA text represented the only means of communicating to a potentially mass audience about this new solution for alcoholism in 1939, it was very carefully written. The Oxford English Dictionary of that era defines powerless as: 'without power or ability; devoid of power, helpless.' AA explains this as losing 'the power to choose whether he will drink or not.' The AA text expands on this state throughout the text speaking of the 'peculiar mental twist already acquired' whereby alcohol always wins out over willpower. Importantly, writing two years after AA formed, the text speaks to its members as 'were powerless,' indicating they had recovered (Alcoholics Anonymous, 1939).
The second part of Step 1, 'that our lives had become unmanageable,' is consistently given witness by the narratives of the varying terrible life circumstances that one comes into recovery with. Our notion in this regard was that this was attributable to more complexity than simply the cost of alcohol or behaviour when highly intoxicated on drugs or alcohol. Rather, when combined with the first half of Step 1, we surmised that the reason behind powerlessness and unmanageable life circumstances was fundamentally a broader disorder of decision making, preceding and progressive with the development of addiction.
Our framework builds on established models, in particular the iRISA (impaired Response Inhibition and Salience Attribution) (Goldstein, R. and Volkow, N., 2001; Kwako, L. et al, 2016; Ceceli, A. et al, 2023) modelwhere the neurobiology of salience of drug cues is postulated to overpower that of other reinforcers with a concomitant decrease in self-control. The iRISA model underpins the familiar cycle in addiction as shown in Figure 1 below:
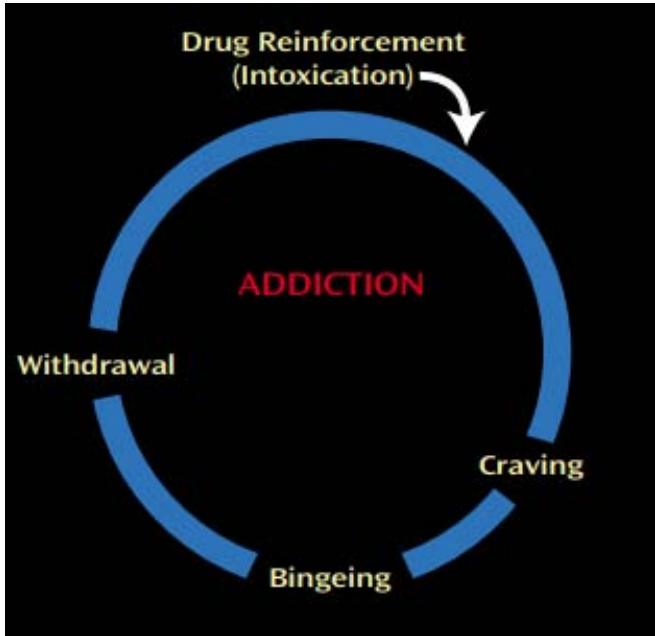
(Goldstein, R. and Volkow, N., 2001) Figure 1: Behavioral Manifestations of the I-RISA (Impaired Response Inhibition and Salience Attribution) Syndrome of Drug Addiction In further exploring the neurobiology or Step One, we additionally postulated that the severe dysphoria, often encountered in withdrawal, had a neurobiological basis which contributed to the overpowering of inhibitory circuitry demonstrated in the prefrontal cortex (Ceceli, A. et al, 2023). Contemplating the seemingly inexplicable relapses in persons in early
recovery just as positive life changes seemed to be occurring, our exploration in this regard was guided by previous work on the anti-reward system in addiction (Koob, G. and Le Moal, M., 2008). The anti-reward system refers to behavioural processes, such as aversion and avoidance, that are part of the normal homeostasis in reward function.
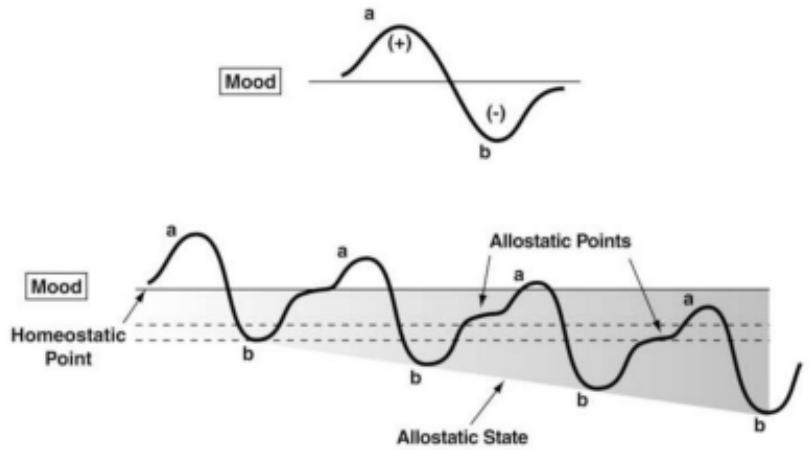
Figure 2: Progressive development of the anti-reward system from initial substance experience (top) to increasing dysphoria and allostatic load (internal physiologic stress) over time with repeated substance use. (Koob, G., 2015)
Just as progressive neuroadaptations develop over time, return to neurotypical functioning has been shown to fail to return to normal until months of abstinence have passed (Koob, G. and Mason, M., 2016). Time to return to normal function is multifactorial and includes the extent of anti-reward development and environmental stresses in recovery (Wemm, S. and Sinha, R., 2019). Portending precarious underlying neurobiology predisposing persons to relapse in the first months of abstinence, our framework expanded this through careful and often lengthy interviews with clients seeking greater insight surrounding prior relapse events.
Exploring the thought process prior to the decision over which people become 'powerless' in severe substance use disorder, that is taking the first drink or using a substance, revealed one commonality: while people could recall the circumstances surrounding the substance use, they most often had no recollection of their thoughts immediately preceding consumption. It became quite apparent that there was a momentary conscious lapse, that the decision was made automatically. It consistently appeared as if some other part of the brain was making the decision and conscious thought was overridden. Returning to the iRISA model (Goldstein, R. and Volkow, N., 2001), neurobiological changes in the brain over the course of addiction had pushed inhibition beyond 'impaired' to 'complete,' meaning no conscious resistance whatsoever in many cases. This marked our first direct indication validating the veracity of AA's beliefs with neurobiology.
We then postulated that a discrete neural circuitto drive the fateful decision and actions, existed in the brain which had become empowered unto itself beyond conscious control. Furthermore, looking at the downward trajectory of people's lives in addiction, we portended this was associated with fundamental decision-making processes that became increasingly self-destructive. Decisions made over the course of addiction became geared solely to creating the conditions for the circuit to activate. Thus, there was a broader impairment in decision making which made life truly 'unmanageable.' Addicted persons had maladaptive brain executive function to the point of being unable to have enough clarity of mind to stop substance use without external help, barring an act of God. Exploring the thought processes of addiction thus gave a working hypothesis for the biology of Step One, powerlessness and unmanageability being the result of specific brain disorders.
Continuing to expand this hypothesis through deductive reasoning, we were aware of the crucial interaction among emotions and decisions (George, S. and Zane, B., 2016). If the purpose of emotions is not fully to motivate decisions (consistent with the meaning of its Latin root, emovere), at minimum, emotions strongly influence the nature of decisions people make. To explain the reason behind the breadth and compulsive tendency of addicted persons to make decisions which inevitably became destructive, we expanded on research on the limbic system (Goldstein, R. and Volkow, N., 2001; Koob, G. and Mason, M., 2016; Ceceli, A. et al, 2023; Feltenstein, M. et al, 2021) of the brain that had become severely maladapted.
In developing a model for the neural circuit that activated the irresistible compulsion to drink or use substances in severe addiction (DSM V), we believed that decisions corresponded with electrical activity within the brain. As such, a neural circuit could be represented initially as an electrical circuit and explored through this perspective (Iles, J., 2005; MacGregor, R., 1987; Singh, J. and Kapur, G., 2019; Cho, S. et al, 2021; Harrison, R. 2008). An NMOS transistor (Chin, A. et al, 2019) amplifier provided a working model:
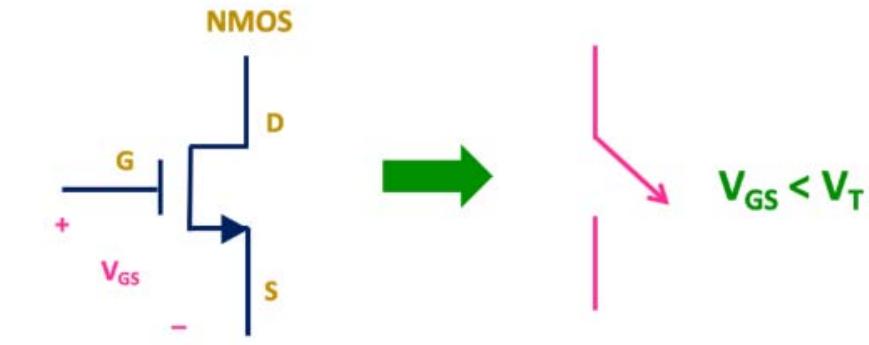

Figure 3: An NMOS transistor acting as a binary circuit: when the voltage at G,
$V_{GS}$ exceeds the threshold voltage for the transistor semiconductor, $V_{T}$, electrical current travels through D to S
Depending on the material nature of the semiconductor (Chin, A. et al, 2019) and the polarity and strengths of the input, a small current at the emitter (D) can be amplified significantly at the output collector (S). In similar fashion we theorized that a small impulse to use substances could pass through a neural network analogue and become amplified to overpower all circuitry in the executive part of the brain. This would model the impulse to use substances at D, overpowering the inhibitory circuitry of the prefrontal cortex at G and producing an amplified output (to consume substances) at S. In neurobiological terms, the result would be powerlessness over the decision to use substances.
Given that this transistor-like amplifier circuit seemed so far beyond reach in the brain (most persons with addictions have tried many times to abstain and sought professional help often to no avail), our therapeutic target became the inputs (the prefrontal cortex, modelled by $V_{\mathrm{GS}}$ above). Should neurotypical function of the inputs be established, the internal circuit could potentially be brought under control. We also hoped that the actual physical composition of the semiconductor-like circuit at G would resultantly return to healthy.
From such reasoning did the union of neuroscience, 12 Step programs and counselling emerge. A concrete understanding of the neurobiological truth of Alcoholics Anonymous' Step One with counselling addressed at the inputs (thoughts, emotions and behaviors) offered a novel, potentially dramatically effective method of helping people afflicted by addiction.
Additional logical assumptions underscored what became the Step One approach: since we were dealing with the brain, the principle of neuroplasticity applied. Neuroplasticity has only become well accepted this century and refers to the modifiability of the brain (Innocenti, G., 2022). Changes in both structure and, consequently, function of the brain are inherent properties that occur throughout life with greater propensity for this in the young developing brain. Nonetheless, neuroplasticity occurs throughout the entire life span. It is not necessarily exclusively adaptive to external stimuli but can also be the cause of neurological and psychiatric pathologies (Innocenti, 2022).
Further logical hypothesis expansions were made prior to the rigorous research to explore our notions: given that addiction seemed to develop over time to eventually dominate behavior, neuroadaptations were most likely to be created within previously healthy circuitry. Addiction was not a separate neural circuit from the rest of the brain, rather it was fully integrated with maleficent neuroadaptations in previous healthy networks. Without definitive healthy neuroplastic changes, the overall state of addiction would continue to exist. Of course, changes in structures and networks would be iterative and at times synergistic, leading to a non-linear progression. This fully matched our observations of persons over the course of recovery. The principles of neuroplasticity also offered the prospect of full recovery, as Alcoholics Anonymous had proclaimed in 1939 after only two years of its formation. A disorder could be reordered.
### b) Basic Science Framework
With experience, observation and access to rigorous scientific articles available for our methodology, we explored models from several disciplines to form a framework for research. The American Psychiatric Association defines addiction as a complex condition – a brain disorder that is manifested by compulsive substance use despite harmful consequences. The limbic system itself is markedly complex both in structures and interconnectedness (Catani, M. et al., 2013; Volkow, N., Michaelides, M. and Baler, M., 2019), not to mention introducing neuroplastic changes (see Figure 4).
We chose to frame our exploration of the behavioural neuroscience of addiction through the lens of a complex dynamic system (Close, C., Frederick, D., Newell, J., 2002). A system is any collection of interacting elements for which there are cause-and-effect relationships among the variables. While a reductionist approach (Mazzocchi, F., 2012) is often used in complex systems and was an initial phase of our exploration, the systemic approach insists on an understanding of the interaction among variables in modelling. The specific system model under investigation was the transistor amplifier circuit, seen as having multiple, likely interconnected inputs and a semiconductor element with variable material composition. The term complexity refers to both the number of variables (inputs) and the interactions among all.
The descriptor, dynamic, in complex systems acknowledges that the variables and relationships among them change over time (neuroplasticity). In our case, changes may or may not be entirely adaptive to external environmental stimuli but are dependent on all other system elements. Fortunately, overall, there is relative stability of the system over days to months to years despite individual elements functioning in millisecond timeframes. With most neural network functioning at generally low frequencies (<100 Hz), current diagnostic methods in micro and macrobiology reveal considerable not only about structure but most importantly, function.
To date, we have identified numerous areas and major nuclei in the limbic system involved in addiction. Each of these have distinct subregions and multiple functions. Additionally, there are a number of potential physiological neuroadaptations at the neuronal level for each neuron in the system. All areas and nuclei communicate with each other and information transmission integrity is subject to change although there is consistency in this aspect of neuroplasticity among all connections. Fortunately, the inter-element
communication is dominated by a number of discrete pathways simplifying understanding in this regard. This array of intercommunicating elements with a brief description of each is found in Tables 1., 2., and 3. and illustrated in Figures 4., 6., 7. and 9.
Table 1: Structures and Areas of the Limbic System Implicated in Addiction
<table><tr><td>Limbic Element</td><td>Brief Function(s) relevant to Addiction</td><td>Neuroadaptations in Addiction</td></tr><tr><td>Amygdala</td><td>Signals fear, anxiety, aggression, anger, male sexual impulse</td><td>Grows in size, hyperexcitable and hyperreactive</td></tr><tr><td>Hippocampus</td><td>Memory associated with emotion, also regulates cortisol in stress response</td><td>In active addiction, new information storage declines except regarding substances</td></tr><tr><td>Hypothalamus</td><td>Signals stress response from limbic system to the body</td><td>Maintains an elevated, less responsive baseline function</td></tr><tr><td>Lateral Habenula (LHb)</td><td>Important in aversion behavior, amplifies negative input and signals back to rest of system</td><td>Becomes increasingly sensitized to substance cessation</td></tr><tr><td>Mammillary Bodies</td><td>Involved in transmission of memory information, reward prediction</td><td>Function declines, especially with alcohol</td></tr><tr><td>Nucleus Accumbens (NAc)</td><td>Motivation for reward, reward prediction error, limbic-motor interface</td><td>Increasingly blunted DA response, greater influence over motor as PFC declines</td></tr><tr><td>Prefrontal Cortex (PFC)</td><td>Executive control over decision making, emotional regulation, working memory and calculation/planning, risk/benefit</td><td>Decline in all functions, appears (by deduction) to focus more on substances</td></tr><tr><td>Rostromedial Tegmentum (RMTg)</td><td>Reacts to negative DA in VTA, signals LHb, aversion</td><td>Responsive to substance cessation, temporarily relief if substances planned</td></tr><tr><td>Thalamus and Insula</td><td>Distributes external and interoceptive (Insula) stimuli to rest of system</td><td>Some literature points to preferential bias to transmit substance related information</td></tr><tr><td>Ventral Tangial Area (VTA)</td><td>Signals want or need to NAc through DA efferents</td><td>Increasing response to substance related cues</td></tr></table>
Table 2: Communication Pathways Involved in Addiction
<table><tr><td>Communication Pathway</td><td>Major Elements Connected (note pathways are bidirectional)</td><td>Function</td></tr><tr><td>Mesolimbic pathway</td><td>VTA to NAc</td><td>Physiological DA response</td></tr><tr><td>Amygdalofugal Pathway</td><td>Amygdala to Hippocampus</td><td>Automatic fear response</td></tr><tr><td>Hypothalamic-Pituitary-Adrenal Axis</td><td>Rest of limbic system signals Hypothalamus</td><td>Stress response</td></tr><tr><td>Medial Forebrain Bundle</td><td>PFC to all elements of limbic system</td><td>Emotional regulation, executive decision making</td></tr><tr><td>Mesocortical Pathway</td><td>PFC to NAc</td><td>Reward prediction & analysis, executive decision input</td></tr></table>
Table 3: Mechanisms of neuroplasticity
<table><tr><td colspan="2">Neuroadaptations at cellular level (neuroplasticity mechanisms)</td></tr><tr><td>1.</td><td>Mitosis (cell duplication in younger people)</td></tr><tr><td>2.</td><td>Myelination or demyelination (affects transmission among neurons)</td></tr><tr><td>3.</td><td>Synaptic growth or recession</td></tr><tr><td>4.</td><td>Dendritic branching (recession or growth)</td></tr><tr><td>5.</td><td>DA receptor up or down-regulation</td></tr><tr><td>6.</td><td>DA transporter up or down regulation</td></tr><tr><td>7.</td><td>DA reuptake up or down regulation</td></tr></table>
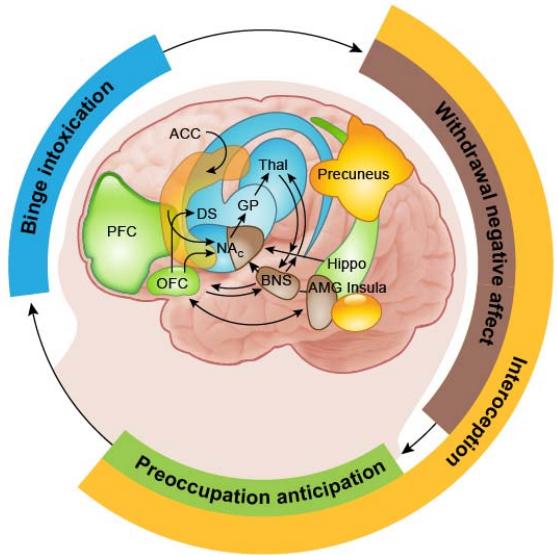
Figure 4: From Volkow, N., Michaelides, M. and Baler, M. (2019). The Neuroscience of Drug Reward and Addiction
Where: PFC represents Prefrontal Cortex, OFC is orbitofrontal cortex (within the PFC in our model), ACC is anterior cingulate gyrus, Thal is thalamus, Hippo is hippocampus, DS is dorsal striatum
What seems a near impossible task to consolidate and understand is made manageable through, first, a reductionist approach identifying anatomy and function of the specific elements. Fortunately, there is general uniformity of basic neuronal structures and networks, as well as commonalities of thinking, emotional and behavioural patterns of addicted people. Most of our clients, virtual or residential, are able to grasp an adequate understanding of this material and self-insight within 45 days to establish sustainable recovery.
### c) Specific Findings
We explored our model of an NMOS amplifier transistor as the decision circuit at the centre of addiction, in the context of consensus surrounding the neurobiology of addiction being dominated by 'the dopamine hypothesis.' This was put forward convincingly by a pioneer of addiction neuroscience, George Koob of The Scripps Research Institute (Koob, G., 1992). This hypothesis has been the focus of significant research to present day and remains the most popular theory underlying the neuroscience of addiction. Addictive substances are classified as such because they all create a dopamine rise in a part of the midbrain (assuming the triune brain anatomically) termed the nucleus accumbens (NAc).
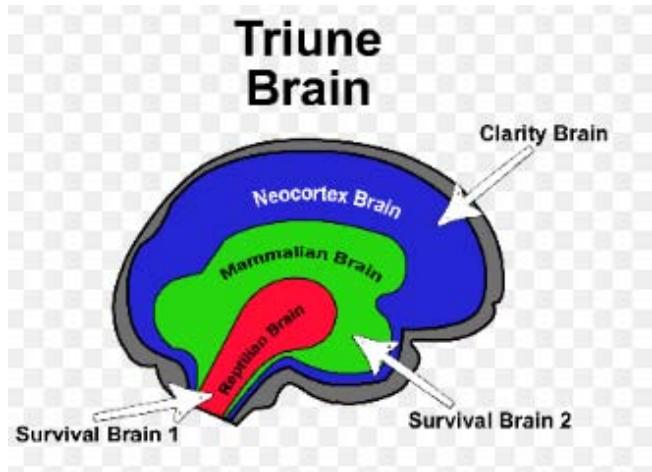
Figure 5: The triune brain schematic
A rise in dopamine is temporally associated with the high of addictive substances. The dopamine system projecting to synapses in the NAc is still considered by most to be the center of reward, the reward system.
This view is unfortunately incomplete when one explores further the complexity of addiction. We also believe that the prevalence of theories which emphasize an uncontrolled reward system as a fundamental issue that continues to maintain societal stigma of addiction. Through considering persons become so addicted to the reward of substances that they don't want to stop, implies continued choice on the part of addicted persons.
Through a reductionist framework, we deducted that it was an excess of dopaminergic neuron excitation, $V_{\mathrm{GS}}$, (compared to inhibitory neurons, $V_{\mathrm{t}}$ ) that ultimately activated the circuit to use substances as modelled by the NMOS transistor (Figure 3.).
An exploration of the dopamine (DA) neuron reveals much when considered in the experiential context of addiction. In brief, chemicals including addictive substances bind to receptors on dendrites (they look like Christmas trees) on the cell body and create electrical charge which then creates an electrical current which travels down the axon. Axons are insulated with myelin and the electrical charge, on reaching the pre-synaptic terminal, activates dopamine transporters (DATs) (see Figure 7.). DATs then bring vesicles of dopamine (like bubbles containing DA) to the membrane and release DA into the synaptic cleft. In this space, DA then binds to dopamine receptors (DARs) of the next neuron. In the binding process, $\mathrm{Ca + }$ molecules are released. This leads to accumulation of positive charge in the post synaptic terminal and an electrical current which then travels down the axon of the ensuing neuron. This simple model explains how substances which activate one neuron then turn on the next neuron in the neural network, which could be, say, the experience of reward.
Recall that we are considering the midbrain which is accepted to be essential for survival through maintaining physiological stability referred to as salience. Clearly, addictive substances, which dramatically elevate DA levels and transmission, threaten salience. In a response to 'control' the dopamine response and maintain salience, through mRNA transcribed through genes in the nucleus, the neuron creates proteins which, at the synapse (Sulzer, D., 2011):
- Down-regulate DATs
- Down-regulate DARs; and,
- Up-regulate DA reuptake transporters
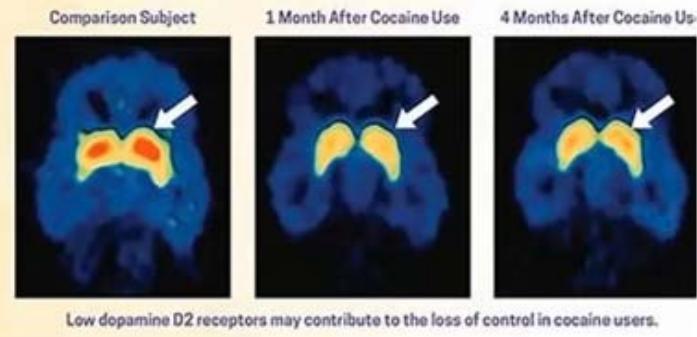
Figure 6: D2Rs (DA2 Receptors) in cocaine users. Harvard Medical School (2021). Understanding Addiction As a result, over time, more of the substance is required to achieve the same electrical energy as occurred with the first experience. Additional adaptations which the neuron can perform are duplicating itself (in developing brains, mitosis) and change dendritic configuration. Advanced substance
use can also affect the integrity of the myelin sheath, giving rise to conductive loss and crosstalk (Ashton, C. & Duffie, D., 2022). Finally, the neuron can create new synaptic connections when so stimulated, a process referred to as Hebbian plasticity (Turrigiano, G. and Nelson, S., 2000).
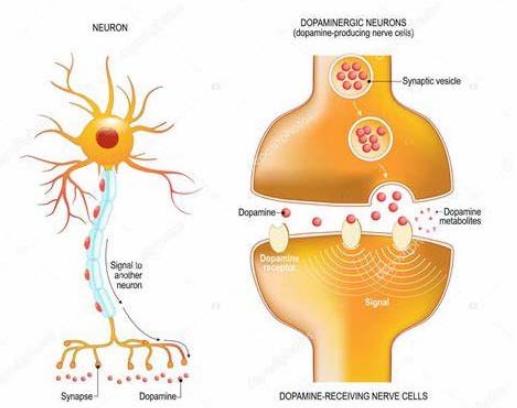
Figure 7: Schematic of the dopamine neuron and synapse
A few fundamental associations can be made through an understanding of neuroadaptations at the cellular level. Clearly, neurons can and do change through distinct mechanisms. Summarily, these help to demystify neuroplasticity in very simple terms. Second, a quick study of the neuron informs us about the science behind tolerance to substances which all addicted persons develop over time. Finally, this contributes to an understanding regarding the 'powerless' aspect of Step One and the inability to stop at one drink when driven to reach previous euphoria (trying to achieve the previous dopamine high in the face of resistant circuitry).
### d) The Drive Towards Substances: The Nucleus Accumbens And Ventral Segmental Area
Assuming the neural circuit to consume substances exists in the dopaminergic system within the nucleus accumbens, further proof of this mechanism as well as identification of inputs forms the next logical exploration. We were guided by much superb literature on the reward side of addiction (Volkow, N., Michaelides, M. and Baler, R., 2019), as well as partially conflicting literature surrounding the NAc in isolation (Floresco, S., 2015). There is good research confirming that the NAc is not simply a 'reward centre' responding proportionately to dopamine afferents. Rather, it is far more sophisticated and functionally aligned with the powerlessness aspect and narratives given and described as: 'there was always the curious mental phenomenon that parallel with our sound reasoning, there inevitably ran some insanely trivial excuse for taking the first drink (AA Basic Text, 1939).'
Returning to the triune brain model, the NAc is a deep structure within the survival-geared midbrain. Executive thought and consciousness are generally accepted to exist in the frontal cortex. Floresco (2015) in his broad review states that the current emphasis on the NAc being the reward center has veered from the more generalidea initially proposed by Mogenson (Mogenson et al., 1980) that this region serves as alimbic-motor interface. Emerging evidence further supports the NAc as having diffuse projections of axons to cortical motor areas (Salgado, S. and Kaplitt, M., 2015; Sawada, M. et al, 2015; Miyachi, S. et al, 2006). Additionally, while the focus is generally on the projections from the Ventral Tangmental Area (VTA), where the dopamine cell bodies reside, to the NAc, little is made of the bidirectionality of circuitry between the VTA and NAc.
Corkrum et al (2020) speak strongly to this as well as Floresco (2015) and Yang et al (2017). Logically, bidirectional communication better explains the actual functionality of reward driven behaviour as well as aversion (which will be explained later). While NAc dopamine synapse activation is seen in anticipation and reward experience, we suggest that the hedonic experience takes place through further projections to the basal ganglia and ventral pallidum (there are few known neurotransmitter sites typically associated with the pleasant feeling of reward within the NAcitself) (Root, D. et al, 2015; Castro, D. et al (2015). With the VTA accepted as signalling 'want or need,' it makes most sense that major inputs to create this emanate from the NAc as Corkrum et al (2020) suggest.
We suggest a primary role for the NAc as a dopamine regulator to maintain salience, and a motor interface (mediated of course by the PFC). Dopamine itself in the mesolimbic system (VTA and NAc) then also acts as a motivator of action for which there is much good evidence (Wise, R., 2004). Significant for our understanding of addiction is the NAc function of measuring reward prediction error (RPE), essential to learning and memory (Yang et al, 2017). This argument for the multiple functions for the NAc and VTA will make further sense as we consider the role of avoidance (we term 'the anti-reward system') as equally significant as the positive reward system in addiction.
We know that as addiction develops, there is progressive decrease in reactivity of DA synapses in the NAc. Termed tolerance, there is increasingly less response in the NAc for the same dopamine signal from the VTA as the mesolimbic system strives to maintain salience. Additionally, in advanced addiction, numerous authors (Corkrum et al, 2020; Febo, M. et al, 2018; Koob, G. and Mason, M., 2016) suggest a deficit in extracellular dopamine creating what is functionally a hypodopaminergic state in advanced addiction. Returning to Step 1, this is a major contributor to craving as experienced as part of the powerless over addiction phenomenon.
People with severe addictions will admit being unable to perform even the most minor activities of daily living, let alone function at all in a work environment without substances. This is unfortunately true physiologically: in severe addiction, the hypodopaminergic state resulting from resistance which has developed in the neural circuitry along with potentially low basal extracellular dopamine levels has a devastating effect on motivation. The simplest things such as showering and getting dressed seem like a monumental task without topping up on substances.
While this may appear as a willful lack of care of self or a manifestation of self-loathing on the part of severely addicted persons, they simply do not have the motivational energy through the NAc. This terrible phenomenon is in addition to the relentless emotional and thought dysphoria persons experience. In a complete hijack of healthy functioning, the limbic system hasnow equated substances with survival, as only substances will provide the necessary dopamine response to feel functional.
### e) Failure to Inhibit the Drive to Substances: The Prefrontal Cortex
The limbic system, and those elements specifically involved in addiction, is a complex dynamic system unto itself whose purpose is the maintenance of homeostasis. Homeostasis can be considered persistence of a living system and society in a state of specified and dynamic disequilibrium (Pross A., 2012). System wise, the interconnected limbic elements can be thought of as alternatively competing and cooperating with each other in active striving towards a persistent and specified orderliness (Turner, J., 2019). Viewed as an internal environment, the limbic system maintains homeostasis through adaptive interfaces (the various elements), which monitor external environments.
As a living, closed thermodynamic system unto itself, geared to maintaining the greater intracorporeal entity, energy is continuously required to survive in a dynamic environment. Knowledge and cognition are equally needed within the limbic system to construct cognitive representations of the environment in which it is embedded, with knowledge of what the contained environment should be, and continuously seek reward to maintain homeostasis in the face of ongoing perturbations to it (Feldman Barrett, L., 2006).
Implicit in this reward seeking function and processing external cues is the Nucleus Accumbens. As one of its critical functions, the NAc calculates reward prediction error (RPE) (Floresco, S., 2015) whereby positive RPE drives anticipation, motivation towards and memory of reward events. The primary neurotransmitter involved in RPE is, of course, dopamine. Resultant neurocircuitry of the midbrain in reward seeking and survival are motivation to activities, behaviours and external substances that result in dopamine increases in the mesolimbic system. Emotions are created through this process.
In simple terms, the elements of the limbic system react to internal and external stimuli, process it through intercommunication including the PFC to drive the mesolimbic system to drive decisions to maintain overall dopamine homeostasis, in an energy efficient manner as possible. Equally, the system creates negative RPE to direct diversion and avoidance. Proof of concept is highly supported by reviews of Deep Brain Stimulation (DBS) for addictions which is aimed at the Nucleus Accumbens (Chang et al, 2022). Electrical stimulation of the NAc through DBS acts as a system override of the rest of the contributions to the NAc of the limbic system.
Addictive substances and behaviours in vulnerable people all produce a positive rise in dopamine levels in the synapses of the NAc. This rise can be multiples of baseline levels and result in significantly elevated RPE initially on consumption. Regardless of input from the PFC, the midbrain interprets substances as contributing to positive valence
(Feldman Barrett, L., 2006). Despite societal beliefs influencing the PFC to inhibit substance use, the midbrain is susceptible to dominate the decision-making process and repeat the experience. In such a manner, addiction progresses to reach the widely accepted three-stage cycle of addiction as described by Koob (2001), Volkow (2018) and their associate researchers.
While acknowledging that participation and influence from other limbic structures is continuously present, focusing on the basics of the neuroscience involved in Step 1 does not detract from the veracity of this hypothesis. During the last decades, many studies have shown that addiction results in heterogeneous cognitive deficits (Pitel et al., 2023), including impairments of executive functions, memory, visuospatial abilities, difficulties in emotional processing, and theory of mind (ToM) abilities (Le Berre et al., 2019). ToM is defined as the capacity to infer mental states from others' social signals to predict their behaviors, desires, intentions, and beliefs. It is now well-known that attention, working memory, and executive functions rely notably on the prefrontal cortex (Maillard, A. et al., 2020).
Executive functions are cognitive abilities that control and regulate the emotional (survival) system to coordinate thoughts and actions toward a goal. Executive functions include mental flexibility, abstraction, planning, problem-solving, shifting of mental states, monitoring and updating of working memory representations, organization, rules deduction, reward delay discounting, and categorization (Maillard, A. et al, 2020).
There is a considerable and emerging literature concerning the role of the prefrontal cortex in emotional regulation, much of it focussing on the PFC and amygdala communication (Suzuki, Y. and Tanaka, S.,2021;Dixon, M. et al, 2017). Unfortunately, less research has been performed regarding the relationship between the PFC and the nucleus accumbens. The connection between the two is referred to as the mesocortical pathway. Within the mesocortical pathway, the anterior cingulate, prelimbic cortex, and orbital prefrontal cortex (OFC) have been accepted as being involved in the regulation of emotional responses, cognitive control, and executive function (Volkow et al. 2019).
Much of the literature points to neuroadaptations in the various aspects of the PFC as leading to persistent addictive behaviors, including the devaluation of natural rewards, diminished cognitive control, and hyper responsiveness to drug-associated stimuli (Koob, G.andLe Moal, M., 2001; Kalivas and Volkow, 2005; Carelli and West, 2014). Generally, the view continues that changes in the PFC contribute to escalation of substance use through mediating conditioned responses, drug craving, and loss of behavioral control (Feltenstein, M. et al, 2021) In light of our observations of clients with severe substance use disorder over the first 30 to 45 days of recovery (abstinence), neuroimaging studies(Goldstein, R. and Volkow, N., 2011) and additional studies focusing
on the effects of chronic stress and substances on the PFC (Koob, G. and Schulkin, J., 2019), we offer an alternative view which is best observed in recovery.
b Brain glucose metabolism
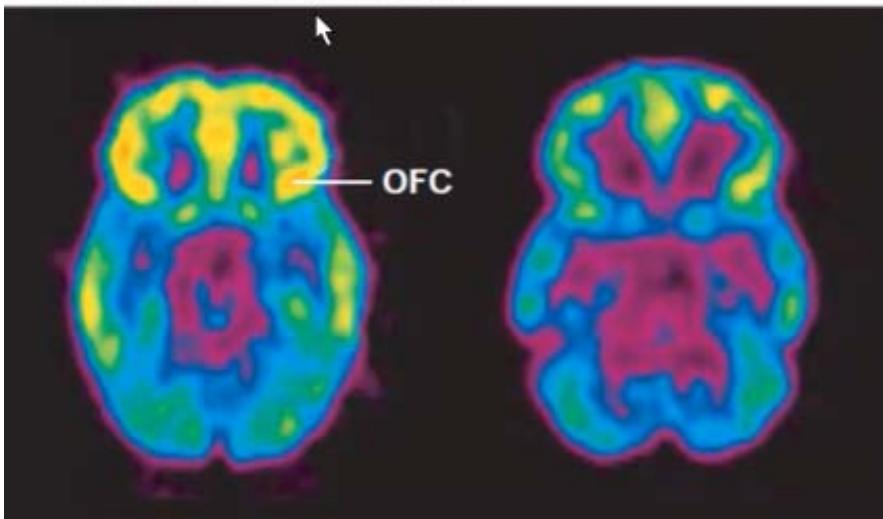
Figure 8: Harvard Medical School (2021). Understanding Addiction. OFC refers to orbitofrontal cortex
Healthy Control After 2 years cocaine abuse As shown in the scan above, neuroimaging studies reveal an emerging pattern of generalized prefrontal cortex (PFC) dysfunction in drug-addicted individuals. This worsening PFC hypofunction is coincident with more drug use, worse PFC-related task performance and greater likelihood of relapse. Widespread PFC activation is seen in drug-addicted individuals upon taking cocaine or other drugs and upon presentation of drug-related cues. We believe that this is compatible with dopamine surges from the NAc and VTA to the PFC, which then creates a transient focus on obtaining the drug of choice or otherwise. We additionally purport that a degree of PFC dysfunction precedes drug use, creating vulnerability for susceptible persons in developing substance use disorders.
Secondly, studies in neuropsychopharmacology demonstrate the molecular effect of stress hormones (Corticotropic Releasing Factor or CRF and cortisol) and substances together and alone in SUD (Wemm, S. and Sinha, R., 2019).
Accordingly, we assert that rather than PFC adaptations as exclusively pursuing substances (which is true to a small extent), the major issue is with the loss of PFC ability to regulate, and specifically inhibit, emotional and motivational drive from the midbrain. This is well observed as well as is partial recovery of the PFC in our clinical experience. Much of the behavioural impulsivity and extremes in emotions and compulsions seen in early recovery (ask anyone at AA about this phenomenon) is in large part due to loss of PFC inhibitory function.
In cognitive testing using the Creyos platformTM, a robust assessment tool developed through combining neuroimaging with neuropsychological studies, clients uniformly scored well below average in virtually all domains regardless of their backgrounds. Fortunately, at the 30 day mark, cognitive scores improved to normal in general. Additionally, scores using Muse 2TMneurofeedback meditation for 15 minutes daily improved substantially over the time in residence with us. This also supports PFC reactivation, a key goal of our program.
Interestingly and of tremendous clinical significance, Muse 2 scores declined greatly for all clients consistently over atwo-day period at our centerbetween 8 to 14 days from admission. Prior to Muse 2, the period from 8 to 14 days had been a time where clients would regularly insist on self-discharging, suffer severe anxiety at times and exhibit fully irrational thinking. We believe that this period refers to the time whereby the hypothalamus begins to resume normal activity (Ashton, C., 2014) and the resulting stress response is excessiveuntil the hypothalamus achieves a new baseline activity over 2 days.
Emotional management including impulsivity is a challenge for both staff and clients, particularly in the first two weeks in residence. Also frustrating to clients is loss of working memory and inability to remember educational material, as well as orientation to place from one day to the next. Again uniformly, working memory returns dramatically for clients (much to their relief) around 18 to 20 days in the program.
To a large extent, this severehypo functioning of the PFC explains powerlessness due to lack of inhibition over the NAc. Further research adds to understanding the magnitude of the drive to obtain substances and associated high risk behaviours. It is much more than a lack of inhibition and a notion which is slowly becoming more recognized in the scientific community (Ashton, GAB 2024). Recall the NMOS transistor amplifier model: in addition to acting as a binary circuit, the semiconductor is fully capable of amplifying the strength of the input signal. Strength of amplification depends on collateral inputs and the physical properties of the semiconductor.
Similarly, the decision to obtain and consume substances in persons with severe addiction often overwhelms any thought of consequences. This is the point where otherwise unconscionable acts are undertaken in substance use disorder, often resulting in severe, traumatic consequences as well as imbedding a seemingly hopeless level of shame.
In pursuit of understanding this third element creating powerless in severe addiction, we undertook to find the neurobiology which amplifies motivation to consume substances. Simple Pavlovian learned behavior is considered a fundamental process in the development of substance use disorder of which there is much literature (Day, J. and Carelli, R., 2007; Heinz, A. et al, 2019). On this basis, we explored the dopamine profile in the mesolimbic from steady state to being cued (small release of DA) to the full DA spike experience corresponding to reward.
It is well known in Pavlovian learning, this process becomes embedded such that reward is experienced in the mesolimbic system prior to the actual consumption and physiologic DA spike. This corresponds to narratives given by many clients that the anticipation of reward is often higher than the consumption experience itself. Additionally, if they do not experience the rewarding feeling anticipated, they become agitated and dysphoric. Complete omission of the reward following experiencing the high of anticipation results in further, often deep dysphoria. This phenomenon is articulated beautifully and termed reward prediction error (RPE) by Keiflin, R. and Janak, P. (2015).
RPE remains fundamental to the development of addiction. Assessment of a potential reward is predicted within the NAc through receiving information from other limbic areas and the cerebellum. This forms a prediction of the reward. What is physiologically the actual result of the behaviour or substance (i.e. the DA response) is measured as well by the NAc calculating reward prediction error. The magnitude of RPE (positive or negative) results in proportional glutamate projected to the hippocampus, as well as the VTA, facilitating encoding of memory. Simply put, the brain remembers things that are unexpectedly better than anticipated
$(+RPE)$ to be able to repeat again. Equally, the brain remembers things which resulted in an experience worse than predicted $(-RPE)$ to avoid repeating (avoidance learning).
Often termed the anti-reward system (Koob, G., 2015; Borsook, B. et al, 2016), this third element in powerlessness is as, if not more, influential as the reward seeking aspect in severe addiction. Negative DA states (Wise, R. and Robbie, M.; Keiflin, R. and Janak, P., 2015) are highly unpleasant and feel excessively fatiguing. There seems no healthy, fast way out of this state, barring external substances.
Reward omissions, as well as aversive outcomes, activate neurons in the lateral habenula (LHb) (Matsumoto, M. and Hikosaka, O., 2007), which indirectly inhibits DA neurons via activation of GABAergic neurons in the rostromedial tegmentum (RMTg). A role for involvement of the LHb in negative prediction errors has been established in animal studies (Keiflin, R. and Janak, P., 2015).
It is acknowledged that reduced activity in a large subset of dopaminergic projections to the nucleus accumbens is associated with aversion. Optogenetic inhibition of ventral tegmental area dopamine neurons produce avoidance of associated places and cues, an effect that depends on DA receptor signaling (Danjo et al., 2014). Inhibition of these neurons via DA receptor activation causes a conditioned place aversion and gives rise to negative affect (Liu et al. 2008). Aversive stimuli of various sensory modalities broadly inhibit dopamine release in the VTA and NAc (de Jong et al., 2018; Menegas et al., 2018).
This is considered a fundamental mechanism for aversion, a function necessary for survival. To put the neurobiology of addiction in perspective, recall our discussion on the mesolimbic pathway. This becomes so severely maladapted that substances are interpreted as necessary for survival. Following that logic, the severely addicted brain now sees absence of substances as a threat to survival and accordingly imposes its avoidance system to deter the person from stopping substance use.
At the prospect of abstinence, it is typical for addicted persons to respond with panic and often anger automatically as the avoidance defence manifests. Unexpected inability to obtain and consume substances is met with an often-prolonged state of severe dysphoria in severely addicted people. Superimposed on a hypodopaminergic state is sudden absence of substances following an anticipatory state, resulting in a negative dopamine spike (Keiflin, R. and Janak, P., 2015). This appears most relevant to the rostromedial tegmental nucleus (RMTg) aspect of the VTA, an area highly associated with registering and encoding aversion (Jhou, T. et al., 2009) which signals inhibitory control over DA neurons in the VTA.
The RMTg is located adjacent to the lateral habenula (LHb). There is a recently emerging literature surrounding both the RMTg and LHb which explains neurobiologically the terrible drive behind addiction (Suzuki, Y. and Tanaka, S., 2021). Simply put, in healthy persons, aversive stimuli activate lateral habenula neurons which, in turn, inhibit dopamine release in the VTA and mesolimbic pathway, thereby inhibiting positive action and creating aversion. As previously identified in this paper, two elements bear heavily on the science of powerless: reduced dopamine response (a chronic hypo dopaminergic state) and a faulty prefrontal cortex, unable to control impulses to consume substances.
The amplification of drive emanates from the LHband a $180^{\circ}$ hijack of the avoidance system. As addiction progresses, the dopaminergic mesolimbic system has been so fundamentally maladapted that it associates substances (natural rewards no longer are able to produce needed dopamine response) with survival; conversely cessation of substances is equated as a crisis to be avoided at all costs. This is particularly evidenced clinically in acute withdrawal, most noticeably in alcohol, opiate and benzodiazepine withdrawal. The pain, dysphoria and physical symptoms from activation of the anti-reward system become progressively severe to the point of becoming unbearable for persons with severe addiction.
With multiple episodes of full or partial acute withdrawal occurring over development of addiction, the anti-reward system becomes increasingly 'kindled' (Breese, G. et al., 2005), meaning increasingly sensitive and hyperreactive to negative stimuli. This is due in large part to the LHb becoming increasingly fierce in its response through progressive neuroadaptations due to stress and substances. While not completely understood (it is hypothesized that cell membrane changes are responsible (Authement, M. et al., 2018), progressive adaptations in the LHb create amplification of negative stimuli (Graziene, N. et al., 2018). This produces increasing distress with each attempt at stopping substances, an experience virtually all addicted people will attest to if they've had multiple attempts at quitting.
With responses to negative stimuli already magnified in other areas of the limbic system (the amygdala and hypothalamus particularly), this is then amplified by the LHb creating unbearable dysphoria. As well, with the kindling effect and dysphoria experienced with withdrawal episodes, this creates learned behavior to avoid abstinence in severely addicted people. Clinically, for persons in the contemplative stages for recovery, it is not uncommon for them to say they feel like they will die if they have to quit substances. For the caring professional, it is most helpful that this seeming complete irrationality is not driven by a compulsion to get high. Rather the anti-reward has created this level of thorough distortion neurobiologically: what appears as ambivalence regarding quitting, is truly an excruciatingly frightening prospect for the addicted person.
While not necessarily described by the DSM 5, it our firm belief that, over the course of development of substance use, the anti-reward system becomes dominant over reward and presents a daunting challenge for caregivers to support the client in overcoming this challenge. Typical for addiction, the level of discomfort anticipated by addicted people in quitting substances is of tenhigher than the experience of withdrawal it self. Given this shift to the major obstacle now being anti-reward and with concurrent diminishing of the reward system, clients will now generally admit to using substances to function, to try to feel and act normal, and to not get physically and mentally sick.
## II. DISCUSSION
Understanding severe addiction through a neurobiological lens is most helpful in informing caregivers of what is actually happening in the addicted persons world at that time. Given the advancement and failure of healthy decision-making processes, it is easy to become enrolled in trying to understand the chaotic situation addicted persons' lives have become, to make sense of this complexity. Understanding this neurobiology then gives evidence and a lens for the counselor to focus their best modalities to support the person out of ambivalence and make the decision to cease substances.
The first part of Step One of Alcoholics Anonymous, we admitted we were powerless over alcohol, now has a concrete neurobiological explanation and proof of veracity. Three separate main processes converge to not only making cessation seem impossible and unwanted, but also an overwhelming neural energy to consume:
1. Severe hypo functioning of the prefrontal cortex;
2. Resistance developed in the mesolimbic circuit to experiencing positive reward; and,
3. Amplification of negative valence by the anti-reward system.
The poorly functioning prefrontal cortex simply does not have the electrical inhibitory power to stop the consumption drive created by the reward and, later, the anti-reward system.
As most alcoholic and addicts will verify in substance use disorder, the immediate thought process prior to acquiring and consuming substances cannot be recalled in any detail. The midbrain has completely taken over conscious thought; most people with addictions when questioned in detail will speak of some unknown sort of force carrying them through the acquisition and consumption phase. At best, they will report some internal chatter (PFC) suggesting that it was not a good idea but did not have the force to overcome the strange drive.
Given the extent of neuroadaptations which have occurred, it then becomes easier to understand and validate the often-unbelievable narratives we hear in addictions counselling. As an example, I received a distressed call from a young professional man who had finished our program and returned home 3 weeks prior to the call. He told me that he was sitting at his kitchen table with an unopened bottle of spirits in front of him. He had no recollection of driving to the liquor store and making the purchase, but acknowledged that somehow, he must have. Recall that the NAc has connections to the motor cortex.
The man had found himself on returning home suddenly under extreme family stress which included involvement of the justice system. Stress hormones, particularly CRF and cortisol, impair prefrontal cortex functioning in similar manner to substances in severe addiction (Sequeira, M. and Gourley, S., 2021).; Wojdala, A., Molins, F. and Serrano, M., 2020). In this man's case, stress had overwhelmed the PFC to such an extent that the NAc was able to completely hijack his actions: he had travelled and purchased the bottle without conscious awareness of what he was doing, let alone the potentially tragic consequences.
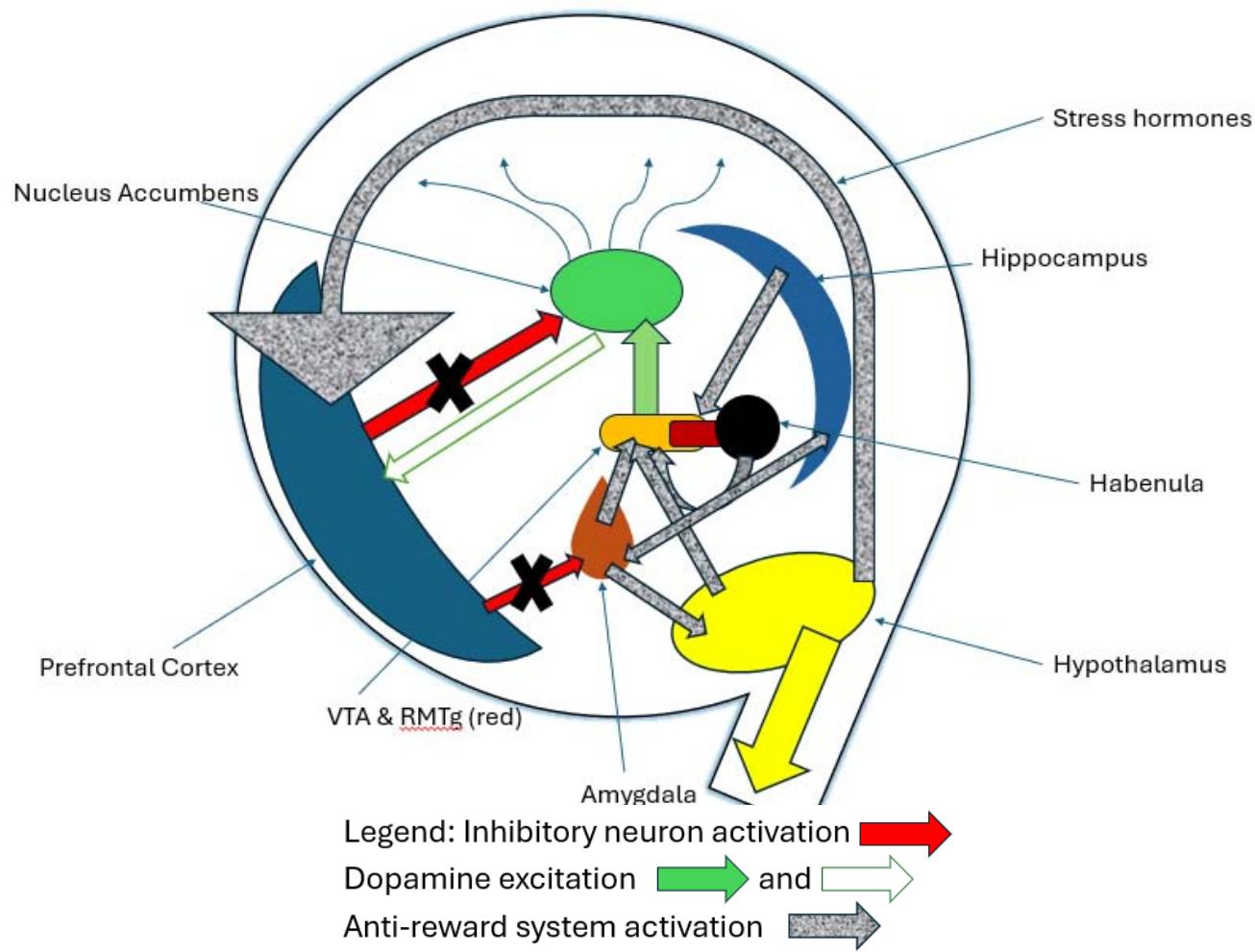
Figure 9: The Powerlessness Circuit. PFC: Prefrontal Cortex, showing disempowered inhibitory (X) influence over a. Mesolimbic (reward) pathway of VTA (ventral tegmental area), RMTg, (rostromedial tegmentum) projecting to Nucleus Accumbens,and b. Anti-reward system main parts being Amygdala, Hypothalamus, Hippocampus, and Lateral Habenula. Result is anti-reward and mesolimbic pathway in full control.
While a striking example of full hijack of the brain by the NAc, his experience is all too common in severe addiction and early recovery. Physiologic stress is always the cause underlying return to substances both in early and later recovery (Koob, G. and Schulkin, J., 2019) and a tremendousimpediment to recovery of neurotypical brain function. Counselling can be exceptionally effective at reducing allostatic load through facilitating new perspectives and solutions.
Unless one is very experienced in the field or has had lived experience with addiction, the irrational self-destruction seen is very difficult to comprehend or validate. Moreover, as substance use disorder has also affected most of the rest of the limbic system structures, thoughts, perceptions, explanations and actions become increasingly distorted. Emotions which are typically a challenge for everyone in life, now become neurobiologically distorted by the structures and system which creates them. For addicted persons, the false thought process becomes fully illogical in that what they firmly believe is keeping them alive is actually bringing on a premature and painful death.
Demystifying powerlessness in addiction is generally met with great relief by clients coming into recovery. It gives concrete answers to a seemingly incomprehensible constellation of symptoms, behaviors and actions that defy rational thought. Understanding the neurobiology of addiction can reduce the persistent self-shaming people in addiction and early recovery experience, as well as allowing clients to focus on the processes that definitively facilitate brain recovery.
Neurocounselling, Step One Therapy, is predominantly educational and focused on giving clients an understanding of what addiction truly is so that they can see its dominant influence on their life and reverse its course over time. Counselors trained in the behavioural neuroscience of addiction are able to effectively facilitate client self-insight into their past and lingering inability to make healthy decisions, knowing that addiction remains a potentially dominant force in early recovery. The educational process to facilitate the client arriving at a level of insight to accept the neurobiology of Step One as tangible, requires proficiency in near all the current counseling modalities. Even though insight is being developed, return to neurotypical brain function does take time and patient understanding is vital to maintaining the therapeutic relationship. Self-disclosure or examples are used often and cautiously to illustrate how the disorder functions. Alcoholics Anonymous employs self-disclosure as large part of its program to effectively illustrate common purpose and belonging among its members.
## III. CONCLUSIONS
The intent of this paper has been to unify two different schools of thought for treating addictions through the common denominator of behavioural neuroscience. This provides a common link between the counselling profession and self-help groups, demonstrating their compatibility and potential synergy. For the counselor, understanding the truth of the first part of AAs Step One, 'We admitted we were powerless over alcohol', gives further support to this community program and enriches the knowledge base for employing the various counselling modalities. Seen through the lens of behavioural neuroscience, counselors can interpret client issues in a precise fashion without being impeded by trying to interpret what is believable or not. What we term neurocounselling has definitive aims and processes:
1. Understand the robust neurobiology and associated behaviour patterns of Step One;
2. Through self-disclosure, examples or reflections of client experience, facilitate the client's understanding of this disorder and its outcomes within their own lives;
3. With addiction framed as a neurobiological disorder developed over a lifetime, facilitate client understanding of the actualeffect of circumstances and resultant behaviours over their lifetime to date (Dodge K. et al, 2009); and,
4. Support recovery of the prefrontal cortex and promote neuroadaptation of the reward system to healthy through positive life changes and experiences.
With the evidence presented herein, we are hopeful that this serves as evidence to unify approaches to care for persons with addiction through a new and more effective paradigm of neurocounselling. Our Step One Therapy approach encompasses a fundamental element of the Alcoholics Anonymous program, as well as its frequent use of self disclosure. Best practices in counseling modalities are fundamental to be able to maintain a positive therapeutic process and teach the underlying neuroscience. Understanding neuroplasticity gives tremendous hope to persons seeking recovery in that healthy functioning, unencumbered by addiction, can be the result over time. With reducing physiologic stress and allostatic load being a concurrent goal of therapy, counselors now have a far more focused approach than sifting through a constellation of issues.
Part 2 of this series will address the second part of AAs Step One: that our lives have become unmanageable. As written in 1939, this suggests that addiction is a progressive disorder to reach a point where life becomes unmanageable by the addicted person. Through combining neurobiology, genetics and environmental stresses we argue that addiction is a progressive disorder that occurs over a lifetime. Fundamentally, we see addiction as a progressive disorder of decision making. Seemingly rational choices made, often well before becoming addicted, set the life circumstances and internal conflictsnecessary to cause disorder progression. Decisions become increasingly unhealthy as do life circumstances to the point where addiction fully dominates brain function and affected persons are unable to see a path out of it.
While we admit that we have given a very brief summary of very robust neuroscience, exploration of the aggregate references herein supports proof of concept. Ultimately proof of concept exists for the vast majority of clients who have engaged in our program. Fortunately, in the five years of refining this method clinically, we are now able to fully expect that most people we see will experience sustained recovery and reclamation of lives that were once on the precipice of tragedy.
### ACKNOWLEDGEMENTS
We would like to thank the anonymous peer reviewers who provided many insights to improve the message and quality of this paper. We additionally thank Liam Carrie, MD, who assisted in research into the science and application of this approach, as well as reviewing this writing.
Generating HTML Viewer...
References
59 Cites in Article
Albert Chin,You-Da Chen (2019). Technologies Toward Three-Dimensional Brain-Mimicking IC Architecture.
C Close,D Frederick,J Newell (2002). Advanced organic chemistry Third edition), by G. W. Wheland. Pp. xi + 871. John Wiley & Sons Inc., New York; John Wiley & Sons Ltd, London. 1960. E7 net.
M Corkrum (2020). Dopamine-Evoked Synaptic Regulation in the Nucleus Accumbens Requires Astrocyte Activity.
Teruko Danjo,Kenji Yoshimi,Kazuo Funabiki,Satoshi Yawata,Shigetada Nakanishi (2014). Aversive behavior induced by optogenetic inactivation of ventral tegmental area dopamine neurons is mediated by dopamine D2 receptors in the nucleus accumbens.
Jeremy Day,Regina Carelli (2007). The Nucleus Accumbens and Pavlovian Reward Learning.
M Dixon (2017). Emotion and the prefrontal cortex: An integrative review.
K Dodge (2009). A dynamic cascade model of the development of substance-use onset.
M Febo (2017). Dopamine homeostasis: brain functional connectivity in reward deficiency syndrome.
L Feldman Barrett (2006). Valence is a basic building block of emotional life.
Matthew Feltenstein,Ronald See,Rita Fuchs (2021). Neural Substrates and Circuits of Drug Addiction.
S Floresco (2015). The Nucleus Accumbens: An Interface Between Cognition, Emotion, and Action.
Beth Fordham,Thavapriya Sugavanam,Katherine Edwards,Paul Stallard,Robert Howard,Roshan Das Nair,Bethan Copsey,Hopin Lee,Jeremy Howick,Karla Hemming,Sarah Lamb (2021). The evidence for cognitive behavioural therapy in any condition, population or context: a meta-review of systematic reviews and panoramic meta-analysis.
Jennifer George,Erik Dane (2016). Affect, emotion, and decision making.
Rita Goldstein,Nora Volkow (2011). Dysfunction of the prefrontal cortex in addiction: neuroimaging findings and clinical implications.
N Graziene (2018). A Focus on Reward Prediction and the Lateral Habenula: Functional Alterations and the Behavioral Outcomes Induced by Drugs of Abuse.
R Harrison (2008). The Design of Integrated Circuits to Observe Brain Activity.
Andreas Heinz,Anne Beck,Melissa Halil,Maximilian Pilhatsch,Michael Smolka,Shuyan Liu (2019). Addiction as Learned Behavior Patterns.
J Iles (2005). Simple models of stimulation of neurones in the brain by electric fields.
G Innocenti (2022). Defining neuroplasticity.
Thomas Jhou,Howard Fields,Mark Baxter,Clifford Saper,Peter Holland (2009). The Rostromedial Tegmental Nucleus (RMTg), a GABAergic Afferent to Midbrain Dopamine Neurons, Encodes Aversive Stimuli and Inhibits Motor Responses.
P Kalivas,N Volkow (2005). The Neural Basis of Addiction: A Pathology of Motivation and Choice.
Ronald Keiflin,Patricia Janak (2015). Dopamine Prediction Errors in Reward Learning and Addiction: From Theory to Neural Circuitry.
G Koob (1992). Dopamine, addiction and reward.
George Koob (2015). The dark side of emotion: The addiction perspective.
G Koob,M Le Moal (2001). Drug Addiction, Dysregulation of Reward, and Allostasis.
George Koob,Michel Le Moal (2008). Addiction and the Brain Antireward System.
G Koob,M Mason (2016). Existing and Future Drugs forthe Treatment of the Dark Sideof Addiction.
George Koob,Jay Schulkin (2019). Addiction and stress: An allostatic view.
George Koob,Jay Schulkin (2019). Addiction and stress: An allostatic view.
Laura Kwako,Reza Momenan,Raye Litten,George Koob,David Goldman (2016). Addictions Neuroclinical Assessment: A Neuroscience-Based Framework for Addictive Disorders.
Le Berre (2019). Emotional processing and social cognition in alcohol use disorder.
Liu (2008). Enhanced selective memory consolidation following post-learning pleasant and aversive arousal.
Ronald Macgregor (1987). Neural Networks.
Angéline Maillard,Nicolas Cabé,Fausto Viader,Anne Pitel (2020). Neuropsychological deficits in alcohol use disorder.
Masayuki Matsumoto,Okihide Hikosaka (2007). Lateral habenula as a source of negative reward signals in dopamine neurons.
F Mazzocchi (2012). Complexity and the reductionism-holism debate in systems biology.
Menegas (2018). Dopamine neurons projecting to the posterior striatum reinforce avoidance of threatening stimuli.
Shigehiro Miyachi,Xiaofeng Lu,Michiko Imanishi,Kaori Sawada,Atsushi Nambu,Masahiko Takada (2006). Somatotopically arranged inputs from putamen and subthalamic nucleus to primary motor cortex.
G Mogenson,D Jones,C Yim (1980). From motivation to action: Functional interface between the limbic system and the motor system.
A Pitel (2023). Impaired Global Precedence Effect in Severe Alcohol Use Disorder and Korsakoff's Syndrome: A Pilot Exploration through a Global/Local Visual Paradigm.
(1998). Matching patients with alcohol disorders to treatments: Clinical implications from Project MATCH.
A Pross (2012). What is life? How chemistry becomes biology.
David Root,Roberto Melendez,Laszlo Zaborszky,T Napier (2015). The ventral pallidum: Subregion-specific functional anatomy and roles in motivated behaviors.
Sanjay Salgado,Michael Kaplitt (2015). The Nucleus Accumbens: A Comprehensive Review.
M Sawada (2015). Function of the nucleus accumbens in motor control during recovery after spinal cord injury.
Michelle Sequeira,Shannon Gourley (2021). The stressed orbitofrontal cortex..
J Singh,G Kapur (2019). Study of Adaptive CMOS Circuits Extracting Feature of Neuroplasticity in Human Brain.
D Sulzer (2011). How Addictive Drugs Disrupt Presynaptic Dopamine Neurotransmission.
Yukihiro Suzuki,Saori Tanaka (2021). Functions of the ventromedial prefrontal cortex in emotion regulation under stress.
J Turner (2019). Homeostasis as a fundamental principle for a coherent theory of brains.
Gina Turrigiano,Sacha Nelson (2000). Hebb and homeostasis in neuronal plasticity.
Antonio Verdejo-Garcia,Valentina Lorenzetti,Victoria Manning,Hugh Piercy,Raimondo Bruno,Rob Hester,David Pennington,Serenella Tolomeo,Shalini Arunogiri,Marsha Bates,Henrietta Bowden-Jones,Salvatore Campanella,Stacey Daughters,Christos Kouimtsidis,Dan Lubman,Dieter Meyerhoff,Annaketurah Ralph,Tara Rezapour,Hosna Tavakoli,Mehran Zare-Bidoky,Anna Zilverstand,Douglas Steele,Scott Moeller,Martin Paulus,Alex Baldacchino,Hamed Ekhtiari (2019). A Roadmap for Integrating Neuroscience Into Addiction Treatment: A Consensus of the Neuroscience Interest Group of the International Society of Addiction Medicine.
Nora Volkow,Michael Michaelides,Ruben Baler (2019). The Neuroscience of Drug Reward and Addiction.
Glenn Walters (2002). Lessons Learned From Project MATCH.
Stephanie Wemm,Rajita Sinha (2019). Drug-induced stress responses and addiction risk and relapse.
R Wise (2004). Dopamine, learning and motivation.
Roy Wise,Mykel Robble (2020). Dopamine and Addiction.
Anna Wojdala,Francisco Molins,Miguel Serrano (2020). Stress and drug addiction: an up-to-date perspective from 2020.
H Yang (2018). Nucleus Accumbens Subnuclei Regulate Motivated Behaviour via Direct Inhibition and Disinhibition of VTA Dopamine Subpopulations.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Christopher Ashton. 2026. \u201cToward a Neurocounseling Paradigm: The Science Behind Step One in Addiction Recovery\u201d. Global Journal of Medical Research - A: Neurology & Nervous System GJMR-A Volume 25 (GJMR Volume 25 Issue A1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Facilitating successful early and sustained remission from substance use disorder remains an extraordinarily difficult puzzle for both clients and supporting persons to navigate. Ninety years since Alcoholics Anonymous developed its twelve step, spiritual approach to recovery from the devastation of alcoholism, initiating and sustaining abstinence from addictive substances remains a tremendous challenge. While the 12-step community continues to support addicted persons at no cost through meetings and relationships, psychology, counseling and medicine are continuing to develop approaches for people to achieve recovery from addiction. To date, there is minimal convergence in these approaches leaving affected individuals with often confusing choices in seeking necessary recovery support. The unfortunate result is the predominant view of substance use disorder as a chronic relapsing disorder whereby relapse is embedded as fundamental to its nature. Greater societal stressors and increasingly dangerous substance access is resulting in death by overdose becoming a major public health issue.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.