Blockage of PD-1 proteins by immune checkpoint inhibitors showed an accepted therapeutic effect in cancer. But; tumor microenvironment exerts its antitumor influence by various mechanisms. Malignant cells have the ability of PD-1/PD-L1 protein over synthesis, which can be a defense action against immune checkpoint inhibitors and immunotherapy. Binding nivolumab with platinum-containing STAT1 will be used to reduce PD-1 genetic level. Nivolumab has the option of endocytosis, while STAT1 is the transcription factor that binds to the DNA, specifically PD-1 gene. STAT1 is the activated protein in response to multiple cytokines stimulation of cancer cells, which are the same for increasing PD-1/ PD-L1 upregulation. The used STAT1 in our therapeutic strategy is the activated form and loaded with platinum particles for damaging DNA bases in PD-1 promoter regions upon translocation to the nucleus. STAT1platinum molecule is connected to nivolumab Fc region by solamargine polymer for selective cancer cell targeting.
## I. BACKGROUND
### a) PD-1/ PD-L1/PD-L2 Levels and Functions
PD-1 is overexpressed on M1 and M2 PD-1 is a surface glycoprotein and is presented on CD4+ and CD8+ T cells, natural killer [NK] cells, natural killer T [NKT] cells, B cells, macrophages, and dendritic cells [DC] subsets. Also; PD-1 is expressed on tumor cells and TAMs [tumor associated macrophages] of many cancer types such as melanoma, non-small cell lung cancer, and head and neck squamous cell cancer [Baumeister et al. 2016]. PD-1 surface receptors should be suitable because it controls the immune balance towards self-antigens. Its deficiency or increased level causes altered immune response, lethal immune response and autoimmune disorder. PD-1 deficiency in murine models by genetic knockdown or blocking its signaling pathway results in serious immunopathology during acute infection via elevated levels of cytokines that result in tissue damage [Barber et al. 2006, Frebel et al. 2012]. Other harmful possibilities can occur, such as autoimmune dilated cardiomyopathy and autoimmune encephalomyelitis. [Sage et al. 2018] PD-1 has an inhibitory function on binding with PD-L1 and PD-L2 expressing cells
[Latchman et al. 2001]. PD-L1 and PD-L2 receptors are expressed on hematopoietic cells such as CD8+, CD4 + T cells, B cells, dendritic cells, macrophages and non-hematopoietic cells like hepatocytes, vascular endothelial cells, epithelial cells, myocytes, pancreatic islet cells, placenta and eye cells. Also, PD-L1 and PD-L2 are expressed on tumor cells and stromal tumor cells [Sun et al. 2018]. PD-1 inhibitory signals play a critical immune modulatory response by induction regulatory [Treg] and natural [T reg]. As result, immune modulatory molecules, such as anti-inflammatory cytokines transforming growth factor-b [TGF-b] and interleukin-10 [IL-10], are secreted [Attanasio et al. 2016]. Activated Treg cells show high PD-1 levels, and their blockage will inhibit Treg cells` essential function.
PD-1 is overexpressed on M1 and M2 macrophages within the tumor tissue that represent tumor-associated macrophages [TAMs]. M1 macrophages have an early tumorigenic effect, while M2 macrophages stimulate metastasis [Tamura et al. 2018, Pollari et al. 2018] PD-1 receptors within the tumor different cells inactivate T cells, B cells, Natural killer cells and dendritic cells, by that way it inhibits the phagocytic action of T cells and other cellular immune response against tumor cells [Gordon et al. 2017]. The cytoplasmic tail of PD-1 entails two structural motifs: ITIM and ITSM. Once binding to PD-L1/PD-L2, the tyrosine residues are phosphorylated, which permits the efficacy of cytoplasmic tyrosine phosphatases such as SHP2. These phosphatases attenuate the signal of the TCR and CD28 [Berraondo, 2019]. PD-1 expressing tumor cells by that mechanism can convert CD8+cytotoxic cells into exhausted cells. PD-1 can stimulate and induce T regulatory cells to consider tumor antigens as self-antigens and escape from phagocytosis within the tumor microenvironment [Jiang et al. 2015]. Also, PD-1 disturbs T cell metabolism by glycolysis suppression and lipolysis stimulation [Patsoukis et al. 2015].
### b) PD-L1/ PD-1 Overexpression and Association with Stat1 Level
Tumor cells can induce stromal cells and TAMs to express PD-L1 directly by cell to cell contact or indirectly through secretion specific mediators such as IL-4, IL-6, IL-10, IL-13, CXCL8, SPP1 and IFN- $\gamma$ [Lu et al.
2019]. In that way, tumor cells recruits surrounding cells for macrophage resistance by PD-L1 increased transcription. STAT1 is activated by the same activators of PD-L1 such as IL-4, IL-13 [Wang et al. 2004], CXCL8 [Chen et al. 2019] $\alpha$ and TNF-[Wang et al. 2000].
The number of PD-1 membrane receptors is increased by IFN- $\gamma$ that activates Janus Kinases [JANs] that phosphorylate STAT1; in turn, activated STAT1 is transferred to the nucleus and acts as a transcriptional factor to enhance interferon-stimulated genes replication [ISGs] [Walter MR 2020]. Also, Anti-PD-1 antibody activates STAT1 through IL-12 activation [Lu et al. 2109].
STAT1-Pt molecule selection to be included in a therapeutic approach after linking to nivolumab can be an effective therapy. As illustrated above, PD-1/PD-L1 transcriptional cascade reactions by cancer cells share the same activators of STAT1. So, PD-L1 and PD-1 enhancement is resulted by malignant cells' mediators against nivolumab, helping STAT1-Pt transported to the nucleus [fig 1]. So endocytosis of the nivolumab-activated STAT1-Pt complex into the cancer cell will permit deposition of platinum loaded on STAT1 onto the Nucleus [in response to malignant cell defense], specifically PD-1 and PD-L1 gene promoter regions. Also that can be upregulated by the advantage of nivolumab that can be endocytosed more than other anti-PD1 antibodies [Ben Saad et al. 2020].
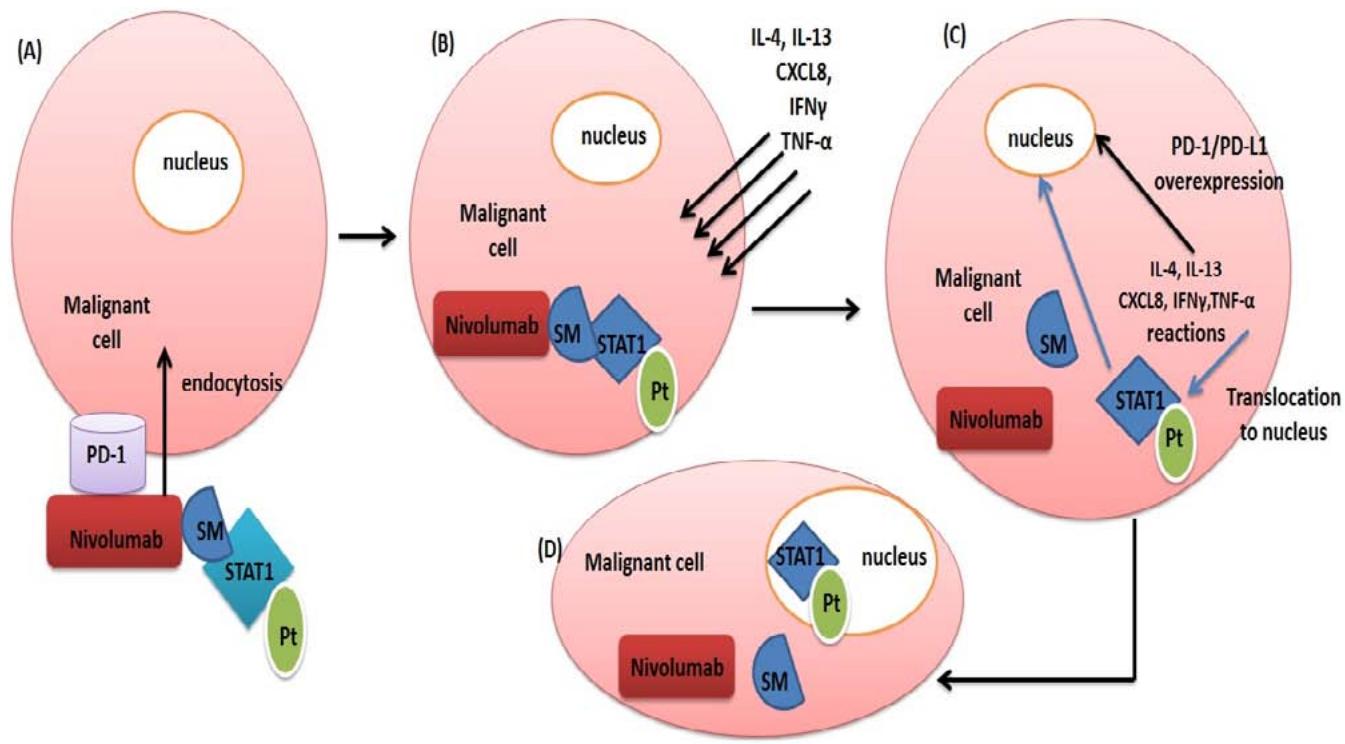
Figure [1]: Stages of nivolumab-STAT Pt endocytosis and functioning.[A] Endocytosis of the molecule into the malignant cell after binding to PD-1 receptor.[B] Nivolumab is dissociated from STAT1-Pt by the action of malignant cell glycosidase enzymes and releasing solamarginine polymer [SM] free within the malignant cell cytoplasm. Malignant cells secrete IL-4, IL-13, CXCL8, TNF- $\alpha$ and IFN $\gamma$ for PD-1/PD-L1 genetic stimulation by various intracellular signals. [C] IL-4, IL-13, CXCL8, TNF- $\alpha$ and IFN $\gamma$ stimulate signaling pathways for elevation PD-1 surface proteins and simultaneously STAT-1-Pt translocation to the malignant cell nucleus.[D] STAT1-Pt attaches to the malignant cell nucleus [PD-1 promoter region] the platinum molecules loaded on STAT1 making adducts with PD-1 promoter region adenine and guanine bases, so damaging the base of PD-1 gene.
### c) Solamargine Specific Anti-Tumor Properties
Solamargine's selective anti-cancer efficacy makes it a candidate for directing nivolumab specifically to malignant cells. Solamargine used in treatment of cancer cell lines of Ehrlich Carcinoma, Leukemia [K562], Colon Cancer [HT-29, HCT-15], Liver Cancer [HepG2, PLC/PRF/5, SMMC-7721], Lung Cancer [A549], Gastric Carcinoma [AGS], Pancreatic Carcinoma [MIA, PaCa-2], Renal Adenocarcinoma [786-0], Uterine Adenocarcinoma [HeLa 229], Ovarian Carcinoma
[JAM], Mesothelioma [NO36], Glioblastoma, Astrocytoma [U87-MG], Prostate Carcinoma [DV-145, LNCap, PC-3], Melanoma [A2058], Breast Cancer [T47D, MDA-MB-231], Osteosarcoma [U20S] and Squamous Cell Carcinoma [A431, SCC4, SCC9, SCC25][Bill, 2013].
Solamargine has multiple anti-cancer mechanisms such as stimulation the intrinsic and extrinsic pathways of apoptosis, increased function of external death receptors [TNFR-1, Fas receptor, TNFR-
1-associated death domain [TRADD], Fas-associated death domain [FADD], elevation of the intrinsic ratio of Bax to Bcl-2 and oncosis [Sun et al 2010].
Solamargine polymer will be the bridging molecule between nivolumab [after glycosylation its Fc portion] and STAT1-Pt complex. [Chemical and biochemical reactions will be discussed later] Nivolumab, like other immune checkpoint inhibitors, are related to exaggerate immune-related Side effects, such as colitis, hepatitis and skin disorders because of crossreaction with healthy PD-1 presenting hematopoietic cells [Dyck et al. 2017]. Those autoimmune adverse reactions can be avoided by solamargine, in other words, it will restrict nivolumab binding with normal PD-1 presenting cells and will facilitate selectivity towards PD-1 expressing malignant cells and TAMs only.
Solamargine molecular formula is C45H73NO15 with a mass of 868.04 Da. Its systematic name is [22R, 25R]-spiro-5-ene-3βL-α-L-rhamnopyranosyl-[1 2glu]-0-α--yl-rhamnopyranosyl-[14glu]-β-D-glucopyranose. [fig.2]
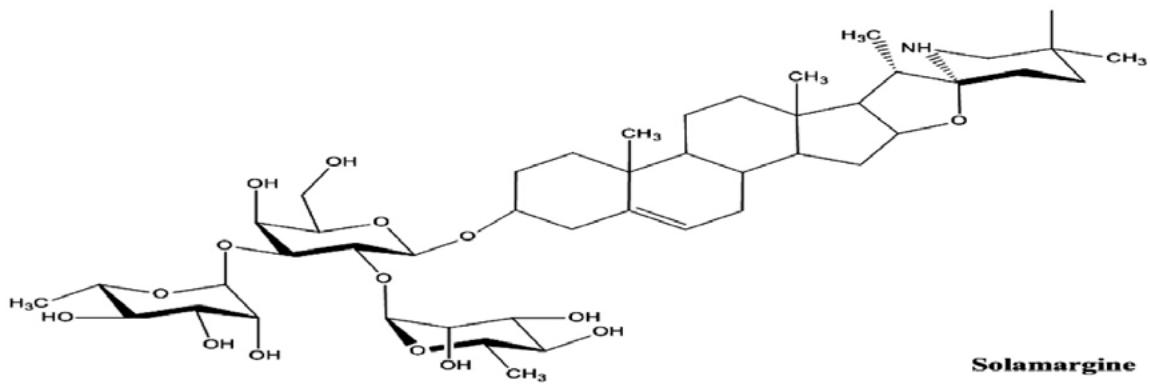
Figure [2]: Molecular formula of solamargine.
### d) Structure of Nivolumab-Stat1- Platinum Molecule
The innovated molecule consists of nivolumab [anti PD-1 MAB] glycosylated with glucopyranose of solamargine. Solamargine $\beta$ -solamargine polymer through its Fc region with is bound to glycosylated cisplatin molecules loaded on seven lysine residues of biochemically activated synthetized STAT1.[Fig 3]
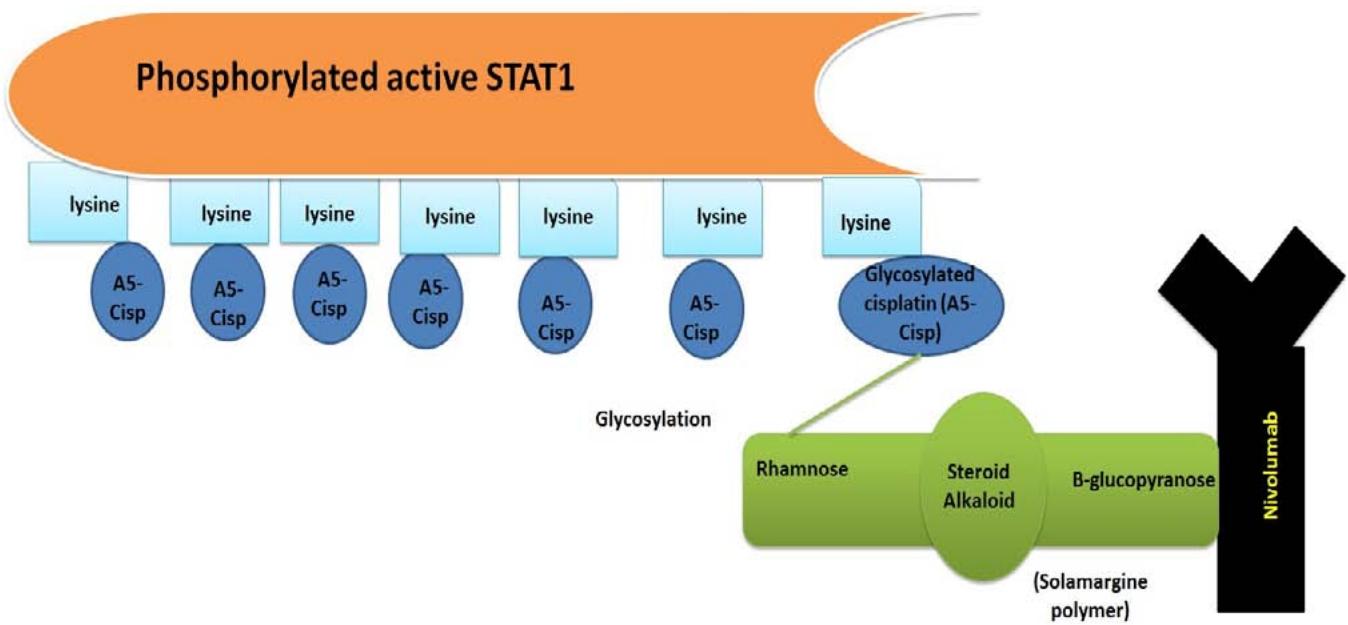
Figure [3]: Structure of nivolumab-STAT1 Pt molecule. Nivolumab Fc portion is glycosylated glucopyranose residue]. Glycosylated cisplatin molecules are $\beta$ -with solamargine polymer [loaded on STAT1 lysine residues [reaction discussed later]. Glycosylated cisplatin is attached to solamargine polymer [rhamnse moiety] by rhamnosyl transferase.
### e) Nivolumab-Stat1 Pt Therapeutic Mechanisms
On Nivolumab-STAT1 Pt administration, it runs within the body circulation towards PD-1 presenting malignant cells due to the presence of multiple targeting elements. The first is nivolumab's nature a monoclonal antibody [Anti-PD1] however, it can be directed towards PD-1 expressing hematopoietic cells such as CD4+ and CD8+ T cells, natural killer [NK] cells, natural killer T [NKT] cells, B cells, macrophages, and dendritic cells resulting in immunosuppression. Also, it can bind to hepatocytes, vascular endothelial cells, epithelial cells, myocytes, pancreatic islet cells, placenta and eye, initiating autoimmune adverse reactions. Here the role of solamargine polymer comes. Solamargine glycoside is considered as an attracting factor for cancer cell requirements for proliferation and spread. Also, it is characterized by selective tumor cell binding. The third is the activated STAT1 which is the needed transcriptional factor for malignant cells to overexpress PD-1 as a defending pathway against nivolumab. So cancer cells and TAMs will uptake the activated STAT1 because it is their rescue to escape from the immune system. After all, it is responsible for increasing PD-1 expression on their surface membranes.
Also, nivolumab-STAT1 Pt molecule is a concentrated anti-cancer therapeutic molecule. Nivolumab is a human immunoglobulin G4 PD-1 immune checkpoint inhibitor antibody that attenuates PD-1 interaction with PD-L1/PD-L2 receptors and stimulates anticancer immunity. It showed good therapeutic parameters [prolonged PFS and increased response rate] in the treatment of non-small-cell lung cancer [NSCLC], melanoma, renal cell carcinoma [RCC] and other cancers. [Guo et al. 2016]. Nivolumab [IgG4] Fc region consists of double heavy-chain Cγ2 and Cγ3 constant domains that are bound to two Fabs, comprising VH and Cγ1 [heavy chain] and VL and Cj/k [light chain] domains, through a hinge. The Fc region has the dominant role for functioning. A biantennary oligosaccharide moiety, covalently attached to Asn297 in the Cc2 domain, contains two N-acetylglucosamine residues, and a branching mannose residue to which α[1-3] and α[1-6] 'arms' of mannose and N-acetylglucosamine residues are attached. The oligosaccharide moiety can additionally contain a fucose residue, attached to the first N-acetylglucosamine residue, and galactose and sialic acid residues attached to the α[1-3] and α[1-6] arms [Davies and Sutton 2015].
Activated STAT1-Pt molecule will be a trap for malignant cells. Malignant cells use activated STAT1 for tumor spread and immunity resistance [Messi et al. 2017]. This is observed in the reduction of NK cellular activity in multiple myeloma, acute myeloid leukemia [AML], and acute lymphoblastic leukemia [ALL] [Bellucci et al. 2015] after IFN $\gamma$ stimulation. While in head and cancer, wild type of EGFR induces
JAK2/STAT1 activation that promotes the antitumor effect by PD-L1 over-transcription [Concha-Benavente et al. 2016] Interferon regulatory factor 1 [IRF1], which is a downstream activator of STAT1 just after IFN $\gamma$ stimulation, has an enhancing effect of PD-L1 genetic activity [Lee et al. 2006].
Activated STAT1 contains 2-acetyl serine which is essential for protein integrity [Bienvenut et al. 2012]. Lysine residues 114, 175, 29, 366, 525, 637 and 665 are methylated. Methylation gives the advantage of the antiviral function. Methylation is done by methyltransferase SETD2 [Chen et al. 2015]. Lysine residues without methylation are the target ones to be conjugated with glycosylated cisplatin molecules. That conjugation will not affect STAT1 function in malignancy. Glutamic acid residues 657, 705 are ADP-ribosylated by PARP 14. Glutamic acid ADP ribosylation suppresses STAT1 phosphorylation [Iwata et al 2016]. During synthesis and purification of activated STAT1-Pt molecule, ADP ribosylation of glutamic acid will be avoided. Tyrosine 701 residue is phosphorylated in response to Janus protein-tyrosine kinase and epidermal growth factor receptor stimulation after IFN $\gamma$ induction [Quelle et al. 1995, Iwata et al 2016]. Tyrosine 701 phosphorylation also is one by KIT-Asp [816] mutants in neoplastic mast cell lines [Chaix et al. 2011]. Serine residues 708, 745 phosphorylation occurs through IFN- $\alpha/\beta$ induction by IKK $\varepsilon$, so serine residues 708,745 phosphorylation is essential for STAT1 activation [Perwitasari et al. 2011]. Serine 727 phosphorylation and tyrosine 701 phosphorylation is really necessary for STAT1 activation. Serine 727 phosphorylation occurs by the action of etoposide and PKCdelta. [Brodie and Blumberg 2003, Wen et al. 1995]
Glycosylated cisplatin are combined with purified active STAT1 lysine residues. Glycosylated cisplatin [platinum IV] are prodrugs that undergo activation to platinum II by malignant cell reductants such as ascorbic acid and glutathione. Being a prodrug and activation inside malignant cells only minimize possible side effects to a significant extent. Glycosylation helps attachment to lysine residues and at the same time, glycosylation to rhamnose residues of solamargine [fig.2]. Also, platinum IV drugs are favored other than platinum II ones because they are more stable and have longer half-life than platinum II drugs. Glycosylation adjusts steric hindrance and length to enable cisplatin for a reduction potential and positive shift to the cancer cells. It is not forgettable that platinum IV drugs have lipophilicity more than platinum II. Lipophilicity permits more access of platinum IV drugs for tumoral cellular uptake and DNA adenine-guanine platination. The used platinum IV drug in STAT1-Pt is A5 complex of cisplatin [fig. 4]. It is known that A5 has more efficacy towards HeLa, A549, MCF-7 and PC3 cancer cell lines other than cisplatin and oxaliplatin. [Jing, et al., 2016].
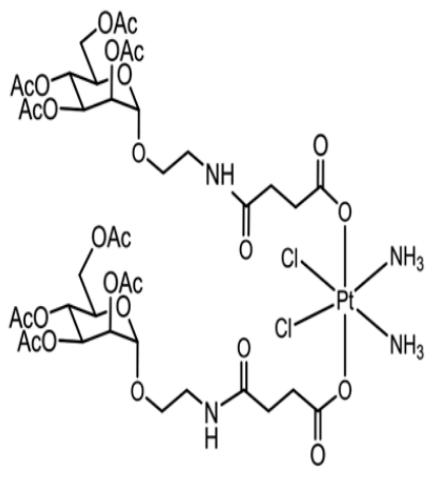
A5 Figure [4]: Structure of A5 cisplatin complexes.
### f) Chemical and Biochemical Steps for Nivolumab-Stat1-Platinum Molecule
1. Purification of activated STAT1: HeLa cells have a major role in STAT1 cultivation. HeLa cells will be incorporated with Lysine 6- dehydrogenase gene delivered by exosome pDNA [plasmid DNA] containing the enzyme gene [Munagala et al. 2021], then keeping PH 10.1 and temperature 70 o C [Heydari et al. 2004]. $70^{\circ}\mathrm{C}$ will not affect STAT1 protein integrity because its denaturation temperature is $95^{\circ}\mathrm{C}$ [Sisler et al. 2015]. The resulted STAT1 within HeLa cells will be unphosphorylated and deaminated lysine residues. Then IFN stimulation of HeLa cells for tyrosine phosphorylation. [Kim and Maniatis 1996]. However, it is inactivated in the nucleus by unknown tyrosine phosphatase PTP and purified as Stat1-PTP from HeLa nuclear extract. [ten Hoeve et al. 2002] Then E6-E6AP complex [one of the Human Papilloma Virus E6 oncoproteins] can be used to combine with Stat1-PTP and degrade it in vitro to yield purified phosphorylated STAT1 [Jing et al. 2007]. The purified STAT1 is phosphorylated at its tyrosine 701 and serine 727 because of IFN stimulation besides deaminated lysine residues 114, 175, 296, 366, 525, 637 and 665, and that is the wanted form to be used in our molecule [fig.5].
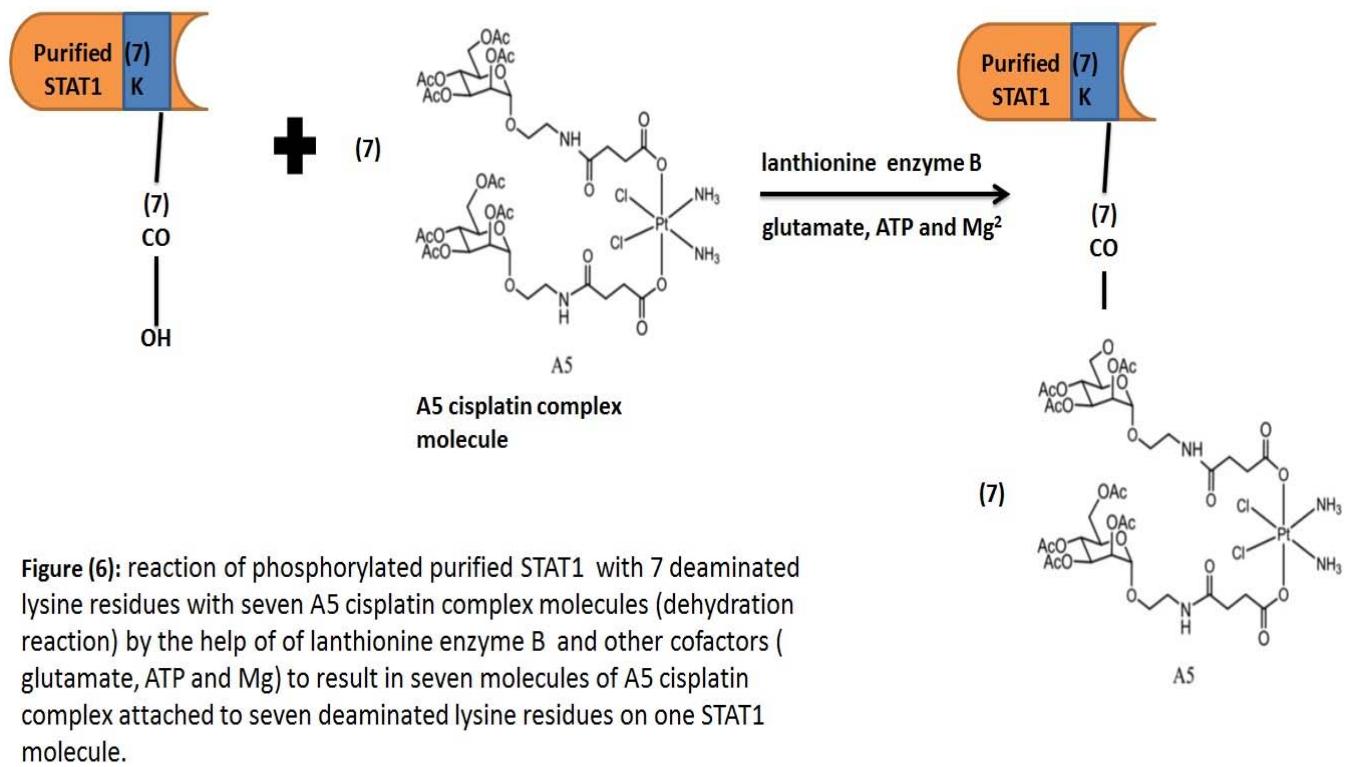
2. A5 complex molecules of cisplatin will be reacted with deaminated Lysine residues of purified STAT1 [not methylated] by lanthionine biosynthetic enzyme B [LanB] proteins in the presence of glutamate, ATP and $\mathrm{Mg}^{2+}$. [Garg et al. 2103]. This in vitro dehydration reaction between the glycosidic component of 7 molecules of A5 complex cisplatin and 7deaminated lysine residues of phosphorylated STAT1 to result in STAT1-A5 cisplatin molecule. [fig.6]
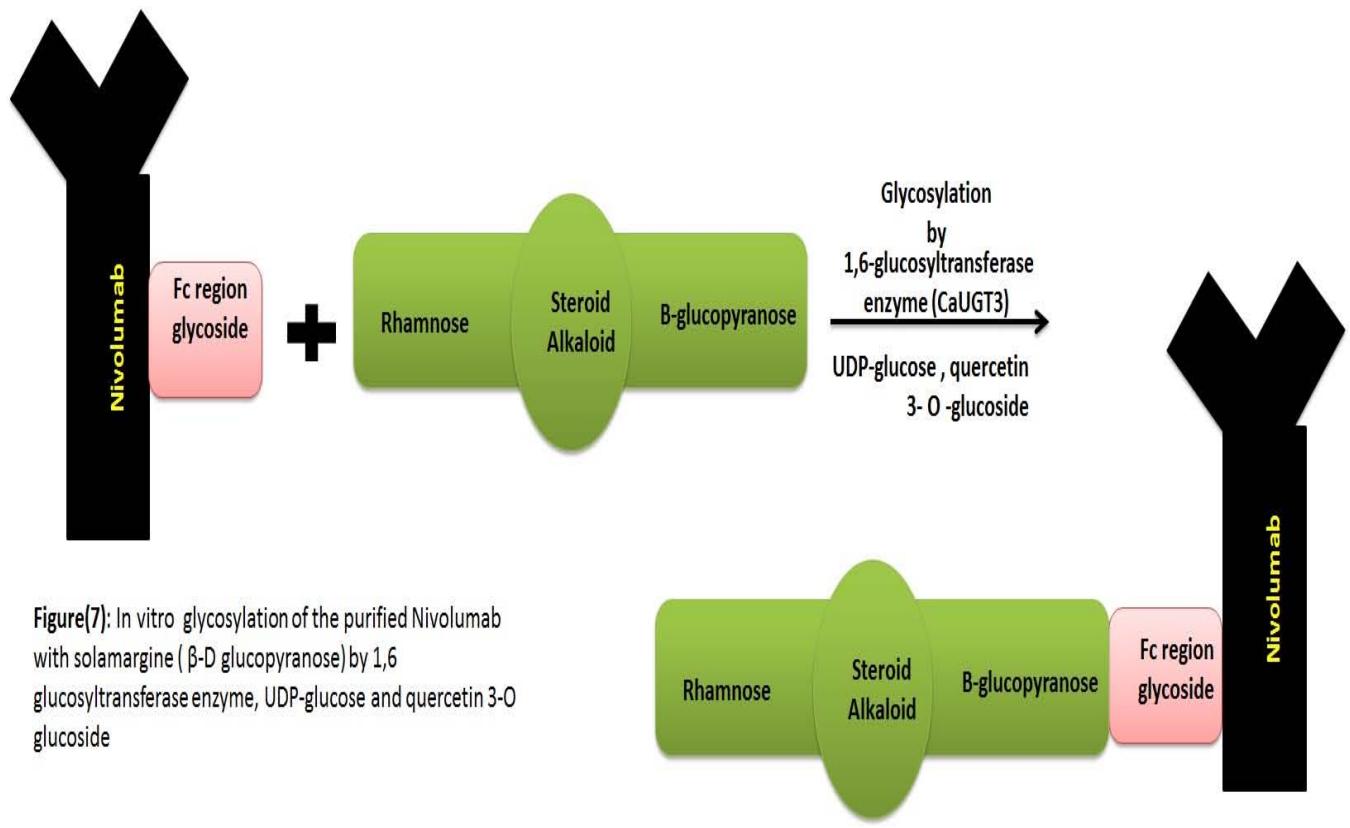
3. Production of glycosylated Nivolumab with solamargine polymer: Transgenic immunization for human immunoglobulin loci with genetically recombinant Chinese hamster ovarian cells expressing human PD-1/PD-L1/human IgG1 Fc fusion protein. [Mimura et al. 2018]. The core complex biantennary heptasaccharide attached to the purified nivolumab Fc region is GlcNAc2Man3GlcNAc2. Previously mentioned heptascchride can be attached to G0, G1 or G2 saccharide according to the number of galactose residues.G0 has no galactose residue, and G1 has one galactose terminal, while G2 has two galactose residues. [Mimura et al. 2018]. 6-glucosyltransferase enzyme [CaUGT3] can elongate the heptasacchride G0/G1/G2 of nivolumab as a sugar acceptor to $\beta$ -D-glucopyranose of solamargine. [Masada et al. 2009]. The enzymatic assay is used with the purified nivolumab using quercetin 3-O-glucoside as an acceptor substrate in the presence of UDP-glucose. The same retention time and UV absorption of quercetin 3-O-gentiobioside result in nivolumab [one side Fc region] with solamargine polymer [Masada et al. 2009]. [fig.7]
4. Solamargine rhamnose moiety is transferred to one A5 complex molecule of cisplatin [attached to STAT1] by rhamnosyltransferases besides the nucleotide diphosphate-sugar UDP-rhamnose [UDP-Rha] as a substrate to result in STAT1-Pt-Nivolumab molecule [Lairson et al. 2008] [fig.3].
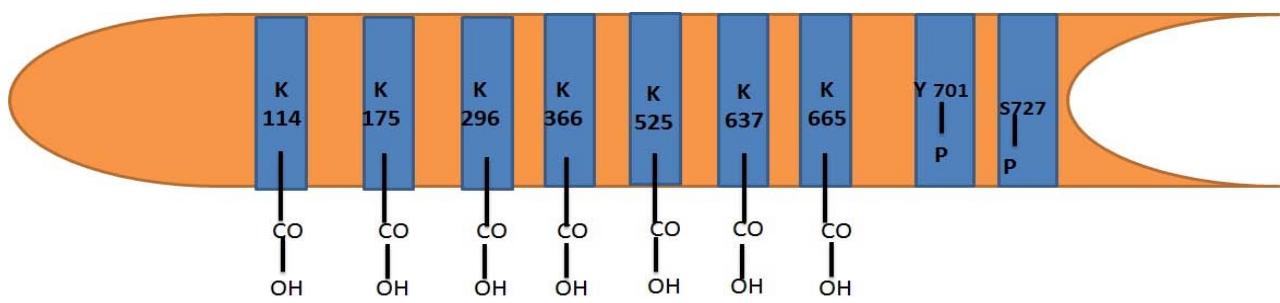
Figure (5): Structure of purified STAT1 that contains phosphorylated tyrosine 701, phosphorylated serine 727 and seven (114, 175, 296, 366, 525, 637, 665) deaminated lysine residues
## II. CONCLUSION
STAT1-Pt nivolumab molecules are targeted towards malignant cells only by the bridging solamargine glycoside. After binding nivolumab to PD-1 expressing malignant cell, endocytosis occurs. Here, glycosidic bonds of solamargine - nivolumab and solamargine-A5 cisplatin STAT1 are hydrolyzed by malignant cell glycosidase enzymes. STAT1- Pt [A5 cisplatin molecules] are transported to the nucleus and seven molecules of platinum IV of A5 cisplatin complex molecule are reduced by malignant cell reductants [glutathione and ascorbic acid] to functioning cytotoxic platinum II. The active form of the used STAT1 in the therapeutic molecule is essential because it does facilitate its nuclear translocation upon different cytokines and IFN-y secretion by malignant cells [malignant cells use those mediators for over recruitment PD-1/PD-L1 genes to resist nivolumab]. So the more mediators secretion, the more STAT1-A5 cisplatin movement to the malignant cell nucleus. In the end, the aim of the molecule is reached, which is damaging PD-1/PD-L1 genetic promoter regions by multiple concentrated platinum containing STAT1 molecules. Also anti-tumor role of endocytosed solamargine is not forgotten as it becomes free after glycosidic bonds hydrolysis. While nivoulmab exerts anti PD-1 signaling pathway, it can be considered a targeting molecule besides solamargine towards PD-1 expressing malignant cell for initiating the cytotoxic reactions of the innovated therapeutic molecule.
Conflict of Interest
Authors have no conflict of interest.
Generating HTML Viewer...
References
44 Cites in Article
J Attanasio,E Wherry (2016). Costimulatory and coinhibitory receptor pathways in infectious disease.
D Barber,E Wherry,D Masopust,B Zhu,J Allison,A Sharpe (2006). Restoring function in exhausted CD8 T cells during chronic viral infection.
S Baumeister,G Freeman,G Dranoff,A Sharpe (2016). Coinhibitory pathways in immunotherapy for cancer.
Roberto Bellucci,Allison Martin,Davide Bommarito,Kathy Wang,Steen Hansen,Gordon Freeman,Jerome Ritz (2015). Interferon-γ-induced activation of JAK1 and JAK2 suppresses tumor cell susceptibility to NK cells through upregulation of PD-L1 expression.
Pedro Berraondo (2019). Mechanisms of action for different checkpoint inhibitors.
Willy Bienvenut,David Sumpton,Aude Martinez,Sergio Lilla,Christelle Espagne,Thierry Meinnel,Carmela Giglione (2012). Comparative Large Scale Characterization of Plant versus Mammal Proteins Reveals Similar and Idiosyncratic N-α-Acetylation Features.
Bill Cham (2013). Drug therapy: Solamargine and other solasodine rhamnosyl glycosides as anticancer agents.
C Brodie,P Blumberg (2003). Regulation of cell apoptosis by protein kinase c δ.
C Sun,R Mezzadra,T Schumacher (2018). Regulation and function of the PD-L1 checkpoint.
Amandine Chaix,Sophie Lopez,Edwige Voisset,Laurent Gros,Patrice Dubreuil,Paulo De Sepulveda (2011). Mechanisms of STAT Protein Activation by Oncogenic KIT Mutants in Neoplastic Mast Cells.
Kun Chen,Juan Liu,Shuxun Liu,Meng Xia,Xiaomin Zhang,Dan Han,Yingming Jiang,Chunmei Wang,Xuetao Cao (2017). Methyltransferase SETD2-Mediated Methylation of STAT1 Is Critical for Interferon Antiviral Activity.
Zhiming Chen,Lei Mou,Yiheng Pan,Chi Feng,Jingjing Zhang,Junjun Li (2019). <p>CXCL8 Promotes Glioma Progression By Activating The JAK/STAT1/HIF-1α/Snail Signaling Axis</p>.
Fernando Concha-Benavente,Raghvendra Srivastava,Sumita Trivedi,Yu Lei,Uma Chandran,Raja Seethala,Gordon Freeman,Robert Ferris (2016). Identification of the Cell-Intrinsic and -Extrinsic Pathways Downstream of EGFR and IFNγ That Induce PD-L1 Expression in Head and Neck Cancer.
Lydia Dyck,Kingston Mills (2017). Immune checkpoints and their inhibition in cancer and infectious diseases.
A Davies,B Sutton (2015). Human IgG4: a structural Perspective.
Ben Elham,Andres Saad,Christopher Oroya,Rudd (2020). Anti PD-1 induces the endocytosis of the coreceptor from the surface of T-cells: Nivolumab is more effective than pembrolizumab.
Helge Frebel,Veronika Nindl,Reto Schuepbach,Thomas Braunschweiler,Kirsten Richter,Johannes Vogel,Carsten Wagner,Dominique Loffing-Cueni,Michael Kurrer,Burkhard Ludewig,Annette Oxenius (2012). Programmed death 1 protects from fatal circulatory failure during systemic virus infection of mice.
Neha Garg,Luis Salazar-Ocampo,Wilfred Van Der Donk (2013). In vitro activity of the nisin dehydratase NisB.
Mojgan Heydari,Toshihisa Ohshima,Naoki Nunoura-Kominato,Haruhiko Sakuraba (2004). Highly Stable <scp>l</scp> -Lysine 6-Dehydrogenase from the Thermophile <i>Geobacillus stearothermophilus</i> Isolated from a Japanese Hot Spring: Characterization, Gene Cloning and Sequencing, and Expression.
Ming Jing,Joanna Bohl,Nicole Brimer,Michael Kinter,Scott Vande Pol (2007). Degradation of Tyrosine Phosphatase PTPN3 (PTPH1) by Association with Oncogenic HumanPapillomavirus E6 Proteins.
Jing Ma,Qingpeng Wang,Xiande Yang,Wenpei Hao,Zhonglv Huang,Jiabao Zhang,Xin Wang,Peng Wang (2016). Glycosylated platinum(<scp>iv</scp>) prodrugs demonstrated significant therapeutic efficacy in cancer cells and minimized side-effects.
Tae Kim,Tom Maniatis (1996). Regulation of Interferon-γ-Activated STAT1 by the Ubiquitin-Proteasome Pathway.
L Lairson,B Henrissat,G Davies,S Withers (2008). Glycosyltransferases: Structures, Functions, and Mechanisms.
Yvette Latchman,Clive Wood,Tatyana Chernova,Divya Chaudhary,Madhuri Borde,Irene Chernova,Yoshiko Iwai,Andrew Long,Julia Brown,Raquel Nunes,Edward Greenfield,Karen Bourque,Vassiliki Boussiotis,Laura Carter,Beatriz Carreno,Nelly Malenkovich,Hiroyuki Nishimura,Taku Okazaki,Tasuku Honjo,Arlene Sharpe,Gordon Freeman (2001). PD-L2 is a second ligand for PD-1 and inhibits T cell activation.
Seung-Jin Lee,Byeong-Churl Jang,Soo-Woong Lee,Young-Il Yang,Seong-Il Suh,Yeong-Min Park,Sangtaek Oh,Jae-Gook Shin,Sheng Yao,Lieping Chen,In-Hak Choi (2006). Interferon regulatory factor‐1 is prerequisite to the constitutive expression and IFN‐γ‐induced upregulation of B7‐H1 (CD274).
Liting Guo,Haijun Zhang,Baoan Chen (2017). Nivolumab as Programmed Death-1 (PD-1) Inhibitor for Targeted Immunotherapy in Tumor.
K Meissl,S Macho-Maschler,M Muller,B Strobl (2017). The good and the bad faces of STAT1 in solid tumours.
Radha Munagala,Farrukh Aqil,Jeyaprakash Jeyabalan,Raghuram Kandimalla,Margaret Wallen,Neha Tyagi,Sarah Wilcher,Jun Yan,David Schultz,Wendy Spencer,Ramesh Gupta (2021). Exosome-mediated delivery of RNA and DNA for gene therapy.
Mimura Yusuke,Toshihiko Katoh,Radka Saldova,O' Roisin,Tomonori Flaherty,Yuka Izumi,Toshiaki Mimura-Kimura,Yoichi Utsunomiya,Kenji Mizukami,Tsuneo Yamamoto,Pauline Matsumoto,Rudd (2018). Glycosylation engineering of therapeutic IgG antibodies: challenges for the safety, functionality and efficacy.
Masada Sayaka,Kazuyoshi Terasaka,Yukie Oguch,Seiji Okazaki,Tunehiro Mizushima,Hajime Mizukami (2009). Functional and Structural Characterization of a Flavonoid Glucoside 1, 6-Glucosyltransferase from Catharanthus roseus.
Olivia Perwitasari,Hyelim Cho,Michael Diamond,Michael Gale (2011). Inhibitor of κB Kinase ϵ (IKKϵ), STAT1, and IFIT2 Proteins Define Novel Innate Immune Effector Pathway against West Nile Virus Infection.
Frederick Quelle,William Thierfelder,Bruce Witthuhn,Bo Tang,Stanley Cohen,James Ihle (1995). Phosphorylation and Activation of the DNA Binding Activity of Purified Stat1 by the Janus Protein-tyrosine Kinases and the Epidermal Growth Factor Receptor.
Ryota Tamura,Toshihide Tanaka,Yohei Yamamoto,Yasuharu Akasaki,Hikaru Sasaki (2018). Dual Role of Macrophage in Tumor Immunity.
Sydney Gordon,Roy Maute,Ben Dulken,Gregor Hutter,Benson George,Melissa Mccracken,Rohit Gupta,Jonathan Tsai,Rahul Sinha,Daniel Corey,Aaron Ring,Andrew Connolly,Irving Weissman (2017). PD-1 expression by tumour-associated macrophages inhibits phagocytosis and tumour immunity.
Peter Sage,Frank Schildberg,Raymond Sobel,Vijay Kuchroo,Gordon Freeman,Arlene Sharpe (2018). Dendritic Cell PD-L1 Limits Autoimmunity and Follicular T Cell Differentiation and Function.
Jennifer Sisler,Magdalena Morgan,Vidisha Raje,Rebecca Grande,Marta Derecka,Jeremy Meier,Marc Cantwell,Karol Szczepanek,William Korzun,Edward Lesnefsky,Thurl Harris,Colleen Croniger,Andrew Larner (2015). The Signal Transducer and Activator of Transcription 1 (STAT1) Inhibits Mitochondrial Biogenesis in Liver and Fatty Acid Oxidation in Adipocytes.
Lingmei Sun,Ying Zhao,Huiqing Yuan,Xia Li,Aixia Cheng,Hongxiang Lou (2010). Solamargine, a steroidal alkaloid glycoside, induces oncosis in human K562 leukemia and squamous cell carcinoma KB cells.
Johanna Ten Hoeve,Maria De Jesus Ibarra-Sanchez,Yubin Fu,Wei Zhu,Michel Tremblay,Michael David,Ke Shuai (2002). Identification of a Nuclear Stat1 Protein Tyrosine Phosphatase.
I Wang,Huamao Lin,Samuel Goldman,Michiko Kobayashi (2004). STAT-1 is activated by IL-4 and IL-13 in multiple cell types.
Y Wang,T Wu,S Cai,T Welte,Y Chin (2000). Stat1 as a component of tumor necrosis factor alpha receptor-1 TRADD signaling complex to inhibit NF-KappaB activation.
Mark Walter (2020). The Role of Structure in the Biology of Interferon Signaling.
Z Wen,Z Zhong,J Darnell (1995). Maximal activation of transcription by STA1 and STAT3 requires both tyrosine and serine phosphorylation.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Waleed O. Atta. 2026. \u201cUsage of Nivolumab – Platinum Containing STAT1 Molecule for Suppression PD-1/PD-L1 Genes in PD-1/PD-L1 Expressing Cancer Cells\u201d. Global Journal of Medical Research - B: Pharma, Drug Discovery, Toxicology & Medicine GJMR-B Volume 22 (GJMR Volume 22 Issue B2).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.