On the basis of a unified theory of the processes of transfer and transformation of any form of energy, called “energy dynamics” by the author, the possibility of synthesizing the methods of non equilibrium thermodynamics and continuum mechanics is shown. A unified substantiation of the main provisions of both the theory of irreversible processes and classical mechanics, free from postulates and hypotheses, is given. At the same time, a new method for studying real processes is proposed, which does not exclude from consideration any (reversible or irreversible) part of them. This made it possible to generalize all three Newton’s principles, to find a short-range form of his law of gravity, and for the first time to substantiate the principle of least action, which mutually enriches both thermodynamics and mechanics.
## I. INTRODUCTION
The development of physics in the twentieth century is characterized by the emergence of three new theories of a revolutionary nature. The first two of them are well known: they are quantum mechanics (QM) and the theory of relativity (SRT and GRT) [^1]. Meanwhile, along with them in the first third of the same century, another no less revolutionary theory arose - the thermodynamics of irreversible processes (TIP), whose contribution to the theoretical thought of the twentieth century was marked with two Nobel prizes. This theory arose on the basis of the pioneering works of Lars Onsager as a theory of the rate of relaxation processes, which he called "quasithermodynamics" [2]. She returned to thermodynamics the concepts of the driving force and speed of the process, and thereby gave thermodynamics the ability to find the causes of the occurrence of a particular process, to identify the direction and speed of its flow, to identify the sources of dissipative losses and their magnitude. This theory explained a number of processes that contradict classical thermodynamics such as "active transport" of substances in biological systems and "ascending diffusion" in alloys (transfer of matter in the direction of increasing its concentration, as well as the emergence of "conjugated" cyclic Belousov-Zhabotinsky reactions and processes of "self-organization" systems far from equilibrium [3] All this made it possible to consider TIP a theory no less revolutionary than QM and RT.
Each of the named theories had its own conceptual system, its own mathematical apparatus and its own model ideas about the object of research. Each was based on a number of additional hypotheses and postulates, some of which, in principle, could not be confirmed by experience with the existing level of experimental and observational tools. Therefore, they developed independently, having practically no points of contact. The disagreements between them were so great that the synthesis of at least the first two of them - QM and RT - was assessed by A. Einstein as "Great Unification".
In this regard, it is of interest to study mechanics and other fundamental disciplines from the standpoint of the deductive method (from the general to the particular), especially since its most productive "principle of least action" also needs to be substantiated.
This article proposes to do this on the basis of "thermokinetics" as a unified theory of non-equilibrium processes of transfer and transformation of any forms of energy, proposed by us in our doctoral dissertation [4], and its further generalization in a new discipline, called energy dynamics for brevity [5]. The main goal of this article is to show that the synthesis of the thermodynamics of irreversible processes and the mechanics of continuous media leads to their mutual enrichment and a number of non-trivial consequences of a fundamental nature.
## II. METHODOLOGICAL FEATURES OF ENERGODYNAMICS
If we adhere to the methodology of thermodynamics as a deductive and phenomenological discipline, which is alien to model concepts of the microscopic "mechanism" of processes, as well as hypotheses and postulates, then the thermodynamics of nonequilibrium (non-static) processes should be built on its own conceptual basis. Such a basis is provided by the law of conservation of the energy of the system $U$ in the form proposed by the Russian professor N. Umov back in 1874 [6]:
$$
d U / d t = - \oint j _ {e} \cdot d f, \tag {1}
$$
where $j_{e}$ is the density of the internal energy flux through the vector element $df$ of the closed surface $f$ of the system a certain volume $V$ in the direction of the external normal n (Figure 1).
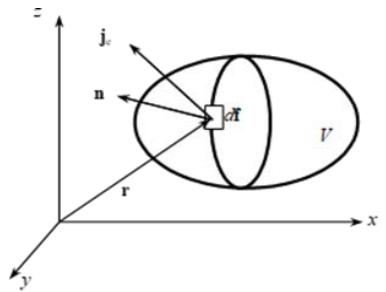 Figure 1: Energy flow across system boundaries
This form of the law of conservation of energy takes into account the kinetics of real processes, without making any assumptions about the mechanism of energy transfer and the internal structure of the system, i.e., considering it a continuous medium. According to him, the internal energy $U$ does not just disappear at some points in space and arises at others, but is transferred by some of its carriers $\Theta_{j}$ through the boundaries of the system. Such "energy carriers" are moles of $k$ -th substances $N_{k}$, their momenta $P_{k}$, charges $\Theta_{e}$, entropies $S_{k}$, etc. In this case, the energy flux $j_{e}$ consists of the energy flux $j_{ej}$ of the $j$ -th kind $U_{j}$, carried by the $j$ -th energy carriers $\Theta_{j}$. Each of these flows is expressed, as is known, by the product of the energy carrier flow $j$ by its potential $\psi_{j} = dU_{j} / dM_{j}$ (specific energy):
$$
j_{e} = \Sigma j_{e j} = \Sigma_{j} \Psi_{j} j_{j} = \Sigma_{j} \Psi_{j} \rho_{j} v_{j}.
$$
Using the Gauss-Ostrogradskiy theorem, we transform $\oint j_{e} \cdot df$ into an integral over its volume $\int \nabla \cdot j_{e} dV$. Then, after decomposing $\nabla (\psi_{j} j)$ into independent components $\Sigma_{j} \psi_{j} \nabla \cdot j_{j} + \Sigma_{j} j_{j} \nabla \psi_{j}$, the energy conservation law (1) takes the form:
$$
d U / d t + \Sigma_ {j} \int \psi_ {j} \nabla \cdot j _ {j} d V - \Sigma_ {j} \int j _ {j} \cdot \mathbf {x} _ {j} d V = 0 (\mathrm {B T}), \tag {3}
$$
where $\mathbf{x}_j \equiv -\nabla \psi_j$ are quantities that have the meaning of the field strength of the potential $\psi_j$ and play the role of local thermodynamic forces in the TIP.
Taking out on the basis of the mean value theorem for the integral sign some average value $\Psi_{j}$ of the potential $\psi_{j}$ and the average value $X_{j}$ of thermodynamic forces $x_{j}$, we find:
$$
d U / d t = - \Sigma_ {j} \Psi_ {j} J _ {j} + \Sigma_ {j} X _ {j} \cdot J _ {j}, \tag {4}
$$
Here $J_{j} = \int j_{j}dV = \Theta_{j}\overline{\pmb{v}}_{j}$ is the vector flow, which has the meaning of the momentum of the energy carrier; $J_{j} = \int \nabla \cdot j_{j}dV$ is the scalar flow of the same energy carrier through the system boundaries, which has the meaning of its consumption.
This equation can be transformed to a form more familiar to mechanics, if we take out the mean value $\overline{\pmb{v}}_j$ velocity $\pmb{v}_j$ of the flow $j_{j} = \rho_{j}\pmb{v}_{j}$ outside the integral sign and replace the thermodynamic force $X_{j}$ with the resultant forces of the $j$ -th kind in its general physical understanding $F_{j} = \int \rho_{j}\pmb{x}_{j}dV = \Theta_{j}X_{j}$:
$$
dU/dt=\Sigma_{j}F_{j}\cdot\overline{\boldsymbol{b}}_{j}-\Sigma_{j}\Psi_{j}J_{j}.
$$
This form of the law of conservation of energy makes it possible to express the strength of any force field $H_{j} \equiv \partial F_{j} / \partial V = -\nabla \rho_{uj}$ in a unified way as the density gradient of the $j$ -th form of the "partial" energy $U_{j} = \int \rho_{uj} dV$. Thus, we emphasize once again that any force fields are generated by the uneven distribution of the corresponding energy carrier in space, and are not any form of matter.
It is easy to see that equations (4) and (5) describe the processes of both the transfer of energy (1st sum) and its transformation (2nd sum), i.e., they combine the laws of conservation of energy related to both classical thermodynamics (where $J_{j} = 0$ and $J_{j} = -d\Theta_{j} / dt$ ) and to classical mechanics (where $J_{j} = 0$ ). This opens up the possibility not only of bringing their conceptual system and mathematical apparatus closer together, but of obtaining, on their basis, all the main provisions of these disciplines as consequences of energetodynamics.
## III. NON-EQUILIBRIUM THERMODYNAMICS AS A CONSEQUENCE OF ENERGODYNAMICS
Energodynamics differs from TIP in that it does not exclude from consideration any (reversible or irreversible) component of real processes and finds fluxes $J_{j}$ and forces $X_{j}$ not on the basis of the principle of increasing entropy, but on a more general basis of the law of conservation of energy in the form (4). This makes it possible to study on its basis both the processes of energy transfer and the processes of its transformation, i.e., This allows us to consider energetodynamics as a theory of the rate of real processes, as a generalization of TIP to processes of useful energy conversion.
The subject of TIP research - relaxation processes - was included in the competence of classical thermodynamics [2]. However, the latter was limited to considering internally equilibrium (homogeneous) systems and quasi-static (infinitely slow) processes, that is, it was actually a thermostat. TIP, however, was considered from the very beginning as a method for describing the kinetics of irreversible processes. To this end, L. Onsager assumed the existence in an adiabatically isolated system of some extensive parameters $A_{j}$, characterizing the deviation of its state from equilibrium, and introduced the concept of the generalized rate of the $j$ -th relaxation process $J_{j} = dA_{j} / dt$ as a derivative of this parameter with respect to time $t$, calling it stream. Then he introduced the concept of thermodynamic force $X_{j} = dS / dU_{j}$ as a derivative of the entropy $S$ over these flows. This made it possible to represent the rate of increase in the entropy $dS / dt$ of the system under consideration as the sum of the products of fluxes $J_{j}$ and forces $X_{j}$ of different nature:
$$
dS/dt=\Sigma_{j}X_{j}J_{j}.
$$
Thus, the concept of force and the ability to explain the cause of a particular relaxation process, to indicate its direction and rate of flow, the power of scattering processes $TdS/dt$ and the final result, returned to thermodynamics.
Fundamentally new for thermodynamics and physical kinetics was another postulate, according to which each of these flows $J_{i}$ arises under the action of all forces $X_{j}$ present in the system. At the same time, it linearly increases with an increase in any of them [2]:
$$
J_{i}=\sum_{j}L_{ij}X_{j}.
$$
where $L_{ij}$ are constant proportionality coefficients characterizing the conductivity of the system with respect to the $j$ -th applied force $X_{j}$ and called by him "phenomenological".
Linear Onsager laws (7) had a matrix form with the number of coefficients $L_{ij}$ equal to $n^2$ ( $j = 1,2, \ldots, n$ ), i.e., $J_i$ was considered as a superposition of fluxes $J_{ij} = L_{ji}X_j$, excited by independent thermodynamic forces $X_j$. This made it possible to explain the well-known at that time thermomechanical, thermochemical thermoelectric and other effects as a result of their "superposition" but required knowledge of a much larger number of empirical coefficients than before. To somewhat compensate for the increase in their number, L. Onsager substantiated the existence of the so-called reciprocity relations:
$$
L _ {i j} = L _ {j i} \tag {8}
$$
To substantiate them L. Onsager needed the theory of fluctuations, the principle of microscopic reversibility, the principle of detailed equilibrium and an additional postulate about the linear nature of the laws of decay of fluctuations [2]. All these provisions went beyond thermodynamics, which is why he called his theory "quasithermodynamics". Due to these relations, the number of coefficients $L_{ij}$ decreased from $n^2$ to $n(n + 1)/2$.
However, since the parameters $A_{j}$ in equilibrium thermodynamics of that time were obviously absent, the practical application of Onsager's theory was difficult. The situation changed when I. Prigogine proposed to find fluxes $J_{j}$ and forces $X_{j}$ on the basis of the expression "production" of entropy due to irreversibility $dS/dt > 0$ and the equations of balance of energy, mass, charge, momentum and its moment, compiled on the basis of other fundamental disciplines, taking into account kinetics of processes [9]. However, since these disciplines lacked the concept of entropy and, moreover, its source $d_{i}S$, he had to make a number of additional assumptions. The main one was the hypothesis of local equilibrium, according to which the volume elements of the nonequilibrium continuum $dV$ are in a state of equilibrium (despite the absence of its sign - the termination of macroprocesses), and their state is characterized by the same set of variables as in equilibrium (despite the appearance of potential gradients), so that the equations of equilibrium thermodynamics are valid for them (despite their inevitable transition to inequalities). Nevertheless, this hypothesis made it possible to find the "entropy production" $d_{i}S / dt > 0$ in stationary irreversible processes supported by "external compulsion" (external forces $X_{j}$ ). Such a theory of irreversible processes (TIP) aroused keen interest of researchers in many countries [10-18] and was evaluated by the award of two Nobel Prizes (L. Onsager, 1968, I. Prigogine, 1977).
It would seem that TIP opened the way to accounting for irreversibility in other fundamental disciplines, which until then were limited to the study of reversible processes and the so-called "conservative systems" in which the sum of external kinetic and potential energies was assumed to be constant. However, this did not happen, since TIP was based on the principle of entropy increase $d_{\mathrm{r}}S / dt > 0$, thereby deliberately excluding from consideration the reversible part of real processes associated with the performance of useful work. Meanwhile, the processes of performing this work, that is, converting energy from one form to another, are primarily of interest to mechanics and representatives of other engineering disciplines. Therefore, the task of developing a more general theory, which would not exclude from consideration any (reversible or irreversible) component of real processes, remains topical. This is exactly what energy dynamics is, which is based on the law of conservation of energy (6) and takes into account the irreversibility of real processes. Its application to continuous systems with any finite number of degrees of freedom makes it possible to detect the presence of internal sources not only in the entropy $S$ or in the number of moles $N_{k}$ of any $k$ -th substance arising in the course of chemical reactions, but also in purely mechanical quantities. It is known, for example, that in the processes of cutting metals or crushing materials, the amount of released heat of dissipation $Q^{d}$ is always less than the expended work $W$ due to the transfer of part of the ordered energy into other forms of their internal energy. At present, this is taken into account in calculations with a "heat output coefficient" less than one. In the general case, internal sources can be found in all energy carriers and in all "emergent" degrees of freedom acquired by the system in the process of evolution and lost in the process of its involution (degradation). This reveals the inconsistency of the attempts of R. Clausius and his followers to make the entropy S a "scapegoat" for dissipation and irreversibility. It becomes obvious that the problem of thermodynamic inequalities is solved by passing to scalar $J_{j}$ and vector $J_{j}$ energy carrier flows through the system boundaries, as follows from equations (6) and (7) [19]. These equations already include relation (11) and therefore are also valid for non-static (irreversible) processes. It is also important that they already contain the required flows $J_{j}$ and forces $X_{j}$, giving them an unambiguous definition and a very specific meaning. This makes it superfluous to compose cumbersome and complex balance equations for $k$ -th substances, charge, momentum, momentum, energy and entropy. In addition, this approach eliminates the arbitrariness in dividing their product into factors, which distorts the contribution of each process to the overall rate of energy dissipation. However, the main advantage of this approach is that it allows one to obtain the main content of TIP without resorting to any hypotheses, postulates and considerations of a statistical-mechanical nature, which comes to a number of non-trivial consequences [20].
First of all, in this way it can be shown that the Onsager reciprocity relations follow directly from the energy conservation law (4). Based on the independence of the mixed derivative from the order of differentiation with respect to the variables $X_{i}$ and $X_{j}(i,j = 1,2,n)$, we have:
$$
\partial^ {2} U / \partial X _ {i} \partial X _ {j} = \partial^ {2} U / \partial X _ {j} \partial X _ {i} \tag {9}
$$
This directly implies the relationship between unlike flows and forces, which we call differential reciprocity relations:
$$
(\partial J_{i} / \partial X_{j}) = (\partial J_{j} / \partial X_{j}).
$$
These relations are applicable to both linear and nonlinear transport laws and admit any dependence of the coefficients $L_{ij}$ on the parameters of the equilibrium state $\psi_i$ and $\Theta_i$. Their application to linear Onsager laws (7) directly leads to the symmetry of the matrix of phenomenological coefficients $L_{ij} = L_{ji}$:
$$
(\partial J_{i} / \partial X_{j}) = L_{ij} = (\partial J_{j} / \partial X_{j}) = L_{ji}.
$$
Their justification shows that these relationships are a consequence of more general reasons than the reversibility of microprocesses. It is equally important that in this way it is possible to simplify Onsager's laws by translating them from matrix form (7) into a diagonal one containing the resulting force $X_{f}[20]$:
$$
J_{i}=L_{i}X_{j}.
$$
where $L_{i} = \Sigma_{j}(\Theta_{j} / \Theta_{i})$ $L_{j}$ are empirical coefficients containing both thermodynamic parameters $\Theta_{i},\Theta_{j}$ and kinetic coefficients $L_{j}$ of the type of thermal conductivity, electrical conductivity, diffusion, etc. This explains why the phenomenological Onsager coefficients $L_{ij}$ in consumer goods neither the one nor the other make sense[13].
Further, the diagonal form of the transfer laws (12) allows us to propose a new method for finding the so-called "superposition effects", according to which these effects can be found as a consequence of the onset of incomplete equilibrium $(J_{i} = 0)$. The specificity of this method is easier to understand by the example of the diffusion of the kth substance in continuous ones that are inhomogeneous in composition (concentration of components $c_{j}$, temperature $T$ and pressure $p$ ). According to (12), the diffusion law has the form:
$$
J _ {k} = - D _ {k} \nabla \mu_ {k}, \tag {13}
$$
where $D_{k}$ is the diffusion coefficient of the $k$ -th substance; $\mu_{k}$ is its chemical potential.
This expression differs from the diffusion law $J_{k} = -\Sigma_{j}D_{kj}\nabla \mu_{kj}$, proposed by Onsager himself, by the absence of the sum of such terms, which makes the problem of finding the diffusion coefficients $D_{kj}$ mathematically incorrect [21]. If we now represent $\nabla \mu_{k}$ in terms of its derivatives with respect to the concentrations $c_{j}$ of independent components, their temperature and pressure, then equation (12) can take the form:
$$
J _ {k} = - D _ {k} \left(\Sigma_ {j} \mu_ {k j} ^ {*} \nabla c _ {j} + s _ {k} ^ {*} \nabla T + v _ {k} ^ {*} \nabla p\right). \tag {14}
$$
where $\mu_{kj}^{*} = (\partial \mu_{k} / \partial c_{j})$ $s_k^* = (\partial \mu_k / \partial T)$ $\upsilon_{k}^{*}\equiv (\partial \mu_{k} / \partial p)$
Three components of the resulting force $F_{k}$ on the right side of this expression are responsible for the usual (concentration) diffusion $F_{kc} = \sum_{j}\mu_{kj}^{*}\nabla c_{j}$, thermal diffusion $F_{kT} = -s_k^*\nabla T$ and barodiffusion $F_{kp} = \upsilon_{k}^{*}\nabla p$. This allows one to separate the thermodynamic $\mu_{kj}, s_k^*$, $\upsilon_{k}^{*}$ and kinetic $D_{k}$ factors of multicomponent diffusion and establish a number of relationships between them, which are experimentally confirmed [19]. To obtain such results based on the Onsager diffusion equation $J_{k} = -\Sigma_{j}D_{kj}\nabla \mu_{kj}$, with the existing experimental means, turned out to be a mathematically incorrect problem [21].
From expression (14) under conditions of diffusion equilibrium $(J_{k} = 0)$, a number of relationships between these forces follows. In particular, if a temperature difference $(\Delta T \neq 0)$, is created in an inhomogeneous system divided into two parts by a porous partition, then a gas or liquid flow through the partition $J_{k} = D_{k}(s_{k}^{*}\nabla T - v_{k}^{*}\nabla p)$, arises, leading to the occurrence of a pressure difference on both sides of the partition (Feddersen effect, 1873):
$$
(\Delta p / \Delta T)_{\mathrm{st}} = -q_{k}^{*} / T v_{k}^{*},
$$
where $q_{k}^{*} = Ts_{k}^{*}$ is the so-called heat of transfer of the $k$ -th substance.
The opposite phenomenon is also known - the appearance of a temperature difference on both sides of the partition when air or other gas is forced through it. Both of these effects have the same nature with the Knudsen effect (1910) - the appearance of a pressure difference in vessels connected by a capillary or a narrow slit and filled with gas of different temperatures, as well as with the Allen and Jones fountain effect (1938) in liquid helium II, consisting in the outflow of helium from a vessel closed with a porous stopper, at the slightest heating. The opposite phenomenon - the occurrence of a temperature difference when creating a pressure difference on both sides of the partition - is called the mechanocaloric effect (Daunt-Mendelssohn).
If the systems initially had the same pressure on both sides of the porous partition $(\Delta p = 0)$ and the same concentration of the kth substance $(\Delta \mathrm{ck} = 0)$, then when a temperature difference $\Delta T$ is created, a concentration difference occurs on both sides of it (Soret effect, 1881):
$$
(\Delta c_{k} / \Delta T)_{\mathrm{st}} = - q_{k}^{*} / T \mu_{kk}.
$$
The opposite phenomenon is also known - the appearance of temperature gradients during diffusion mixing of components, discovered by Dufour in 1872 and bearing his name. In isothermal systems $(\Delta T = 0)$, when a pressure difference $\Delta p$ is created on the membrane, the phenomenon of reverse osmosis occurs - the separation of a binary solution with the release of the $k$ -th component (usually a solvent) from it. This phenomenon is widely used in water treatment plants. The resulting concentration difference of the $k$ -th component is described by the expression:
$$
\left(\Delta C _ {k} / \Delta p\right) _ {\mathrm {s t}} = - v _ {k} / \mu_ {k k}. \tag {17}
$$
These results correspond to those obtained in the framework of TIP [13,15]. However, now they were the result of the imposition of forces, which followed from Newtonian mechanics. At the same time, it was not necessary to admit either the linearity of Onsager's phenomenological laws, or to resort to his reciprocity relations. All this not only simplifies TIP, but also expands the scope of its applicability to nonlinear systems and states that are far from equilibrium.
The advantages of the above method for finding the above effects lie not only in its simplicity, but also in the possibility of its application in nonlinear systems far from equilibrium, where the Onsager reciprocity relations are violated [20]. No less important advantage of the proposed method is a further reduction in the number of $L_{ij}$ coefficients subject to experimental determination from $n$ ( $n + 1$ /2 in consumer goods to $n$ ). Such a reduction is especially noticeable in the group of so-called "thermos-galvano-magnetic" effects, which are caused by the superposition of temperature, electric and magnetic fields, and especially for anisotropic media. The system of equations, which gives a purely phenomenological description of such processes, contains 36 empirical coefficients. Thanks to the use of reciprocity relations, the number of such coefficients can be reduced to 21. Energy dynamics allows you to go even further and, by establishing additional links, reduce the number of these coefficients to 6 [5]. Thus, serious progress is achieved in the study of nonequilibrium systems.
## IV. MECHANICS AS A CONSEQUENCE OF ENERGODYNAMICS
The applicability of the mathematical apparatus of energodynamics in the form of its basic equation (5) becomes especially obvious if the average transfer rate $\overline{\pmb{v}}_j$ of any energy carrier $\Theta_j$ is decomposed into a translational $\overline{\mathbf{w}}_j$ and rotational (circumferential) component $\overline{\pmb{\omega}}_j \times R_o$, expressed as a vector product of the angular velocity $\overline{\pmb{\omega}}_j$ and the instantaneous radius of rotation of the volume element $R_o$. In this case, an additional sum appears in equation (5), expressed as the scalar product of the average moment of forces $M_j = \int H_j \times R_o)dV$ and the average angular velocity:
$$
dU/dt = -\Sigma\Psi_j J_j + \Sigma F_j \cdot \overline{\boldsymbol{w}}_j + \Sigma M_j \cdot \overline{\boldsymbol{\omega}}_j.\tag{18}
$$
This (integral) form of the energy balance makes it possible to operate with the nonequilibrium parameters of the continuum system as a whole, without dividing it into an infinite number of elementary volumes $dV$. This approach makes it possible to preserve the "system-forming" properties of the object of study, the loss of which during the fragmentation of the system "was the biggest shock for physicists since the time of Newton" [22]. The study of continuum systems as a whole frees from the need to resort to any postulates such as the hypothesis of local equilibrium by I. Prigogine [9], especially since, according to (3), the splitting of the system does not reduce the magnitude of the gradients of the potential $\nabla \psi_{j}$ in volume elements and does not bring them closer to equilibrium. Therefore, it should be expected that the above approach to continuum mechanics from the standpoint of energy dynamics will lead to non-trivial consequences.
### a) Correction and generalization of Newton's laws from the standpoint of energetodynamics
It is known that bodies or a system of bodies can move uniformly and rectilinearly or rotate uniformly in the absence of applied forces $F_{j}$ or moments $M_{j}$, that is, "by inertia". This means that in the deductive construction of mechanics (from the general to the particular).
The first principle of mechanics (Newton's law of inertia) [23] should be formulated without excluding rotating systems from consideration: "Anybody continues to be held in its state of rest or uniform translational or rotational motion, until and since it is not forced by the applied forces $F_{j}$ or change this state at moments $M_{j}$ ". Ignoring the prevailing rotational motion in the Universe by the Poincaré - Einstein theory of relativity led to the requirement of invariance of the laws of physics in any inertial reference frames[^1](IRF) and ignoring the prevailing frame of reference inherent in rotating systems.
No less serious consequences have a generalization of Newton's 2nd postulate - the law of force, which states that "the change in the momentum is proportional to the applied force" $F$. This expression defines only the accelerating force, that is, it is too particular. Energodynamics, on the contrary, gives the most general definition of the concept of force $F_{j} = \int \rho_{j}\nabla \psi_{j}dV$ as a measure of the in homogeneity of the system and the cause of a particular process, expressing it through the gradient of the corresponding form of energy $\nabla U_{j}$. Such a definition of mechanical force emphasizes its vector nature, which could not be taken into account by I. Newton due to the absence of the concept of a vector at that time. This definition is applicable to mechanical and non-mechanical, external and internal, long-range and short-range, useful and dissipative, active (applied) and reactive (opposing) forces. In this case, all forces acquire a single meaning, a single analytical expression and a single dimension. This gives researchers a unified method for finding clearly distinguishable forces of nature and opens up the prospect of creating a theory of a unified field as a region of space in which any forces are found.
From the standpoint of energetodynamics, it becomes extremely clear that any force fields are generated not by the presence of any energy carrier, but by its uneven distribution in space. The same is the field of inertial forces $F_{j} = -Ma$, understood as the forces of reaction to the acceleration process. It is also generated by the in homogeneity of the velocity field $\nabla v$. This becomes clearer if we consider that it is impossible to accelerate a body without moving it in space. But along with the displacement, the density of the medium also changes in the place where the body moved, i.e., there is a redistribution of the fields of density and momentum in space. This is reflected in the vector-gradient of velocity $\nabla v$, which is a tensor of the 2nd rank. The presence of this tensor of a symmetric (translational) and antisymmetric (rotational) component of acceleration (similar to the translational and rotational component of velocity) confirms the possibility of rotational acceleration in the field of inertial forces, and thus the possibility of transferring this acceleration from one rotating body to another.
The difference between active forces and forces of inertia is also clearer. Their difference is manifested in the fact that the applied forces and inertial forces have not only opposite signs, but also different values. In ergodynamics, the derivative $\frac{dP_i}{dt} = F_i$, which determines the force of inertia $F_i$, is only one of the flows $J_i$ arising under the action of the applied force $F_j$, so that $F_i \neq F_j$. This requires the introduction of a phenomenological coefficient $L_{ij}$:
$$
J_{i}=L_{ij}F_{j}.
$$
This equation is nonlinear, since upon reaching the limiting speed$|\upsilon| = c$no force$F_{j}$can cause changes in momentum$J_{i}$. From this it follows that with an increase in speed, this coefficient changes, and not the mass$M$in the expression for the momentum$P = M \upsilon$, as postulated by the SRT. This solves the long-standing dispute about the existence of inertial forces and fields of inertia, which are quite real in the presence of the acceleration process but disappear when it stops. The latter is due to the fact that the forces$F_{j}$are functions of the state that do not depend on how it is achieved: by performing work against equilibrium in the system or by its relaxation, while the reaction forces of the system are a function of the process$F_{i} = -dP/dt$as its consequence (in the spirit of the Le Chatelier-Brown principle). Thus, the fields of inertial forces as functions of state, available at any moment of time, do not exist.
The application of enerodynamics to mechanics reveals the need to generalize and Newton's third law, according to which "an equal response always corresponds to an action," and the forces of action and reaction lie on the same straight line. If we denote active forces and reaction forces as $F^a$ and $F^r$, this position has the form $F^a = -F^r$. However, in polyvariant systems there are many active forces $F_{i}^{a}$ and reaction forces $F_{j}^{r}$, and in closed systems their sum always vanishes. This means that each active force $F_{i}$ is opposed by the resulting $F_{i}^{r}$ of reaction forces of various kinds:
$$
F_{i}^{a} = -\sum_{j} F_{j}^{r}, (i, j = 1, 2, n).
$$
According to this expression, equilibrium takes place only when the forces of action $F_{i}^{a}$ and reaction forces $F_{i}^{r}$ have the same nature. Otherwise, the process of transformation of energy of the $i$ -th kind into the $j$ -th form of energy occurs, in which their sum remains unchanged:
$$
F _ {i} \boldsymbol {v} _ {i} + F _ {j} \boldsymbol {v} _ {j} = 0. \tag {21}
$$
This expression indicates the possibility of "branching" the trajectory of the process in the space of the opposing forces $F_{j}$, which is the most common reason for the irreversibility of the process in its most general understanding as the impossibility of returning all nature to its original state even in the absence of dissipation. It is characteristic that, in this form, Newton's third law admits the possibility of the action of separate forces $F_{i}$ and $F_{j}$ not along one straight line, which is currently considered as a violation of the laws of mechanics.
Thus, a more general approach from the standpoint of energy dynamics allows one to give a generalized formulation of all three Newton's laws [24].
### b) Short-range (field) form of the law of gravitation
It is known that Newton's law of gravitation has no theoretical basis and is the result of processing the experimental data of Kepler and his predecessors [23]. For the case of gravitation of two bodies of mass $m$ and $M$, located in a void $(\rho = 0)$ at a distance of their centers of mass from each other $R$, this law of force had the form:
$$
F _ {g} = G m M / R ^ {2}, \tag {22}
$$
where $G$ is the constant of gravity.
This law is valid for astronomical scales, when the intrinsic dimensions of gravitating bodies are negligible compared to the distance between celestial bodies, and the interaction between them is of a "paired" nature (without interference from foreign bodies). Moreover, it is not applicable to continuous media in which there are no "field-generating" or "test" bodies. It is quite obvious that in the presence between the masses $M$ and $m$ of a medium with the same density $\rho$ as theirs, that is, in the absence of a gradient or density difference, the gravitational force would be different. It is all the more interesting to find this force as a consequence of energetodynamics for the general case of a continuous medium with an arbitrary density distribution $\rho(r)$ in space.
To do this, we will use the above definition of the local strength of any (including gravitational) field $H_{g} = \partial F_{g} / \partial V$ as a negative gradient of gravitational energy density $-\nabla \rho_{g}$. The derivative $\nabla \rho_{g} \equiv \frac{\partial \rho_{g}}{\partial r}$ can be represented in the form of the product $(\frac{\partial \rho_{g}}{\partial \rho}) \nabla \rho$, in which $(\frac{\partial \rho_{g}}{\partial \rho})$ is equal, according to the theory of acoustic vibrations [25], to the square of the propagation velocity of vibrations in any medium $c^{2}$ (in this case the speed of light $c$ ). This immediately implies that
$$
H _ {g} = - c ^ {2} \nabla \rho (\mathrm {H m} ^ {- 3}); g = - H _ {g} / \rho = c ^ {2} \nabla \rho / \rho , \mathrm {m c} ^ {- 2}. \tag {23}
$$
This law fundamentally differs from Newton's law in that it reveals the existence of gravitational forces of both attraction $(\nabla \rho > 0)$ and repulsion $(\nabla \rho < 0)$. Therefore, we called it bipolar [26]. In this respect, it does not differ from Coulomb's law, which indicates the unity of the "mechanism" of all interactions.
The most important prediction of this law of gravity is the discovery that the gravitational interaction is the strongest of all known interactions. This follows from the fact that with equal relative density gradients of the energy carrier $\nabla p / p$, the magnitude of the gravitational field strength $Hg$ and acceleration $g$ is maximum in the "physical vacuum", where the speed of light is $c$. For any material medium, it is lower, since the propagation velocity of perturbations in it is equal to the speed of light divided by its refractive index in it. This opens a direct path to the creation of a unified field theory, which A. Einstein dreamed of.
An equally important consequence of the bipolar law of gravity (23) is the prediction of the existence of a gravitational equilibrium corresponding to the condition $\nabla \psi_{9}, \nabla \rho = 0$. This condition is satisfied by the antinodes of waves and any wavelike structures. This explains the stability of various structures, without requiring the balancing of charges of opposite signs or the equality of centrifugal and gravitational forces.
Finally, this law does not require knowledge of parameters that cannot be measured by modern means. This (field) form of the law of gravity is indispensable where it is impossible to single out "field-forming" and "test" bodies with masses M and m. According to him, in the field environment, which is the ether, the "hidden" mass of the Universe, "dark" matter, physical vacuum or "dark energy", the forces of gravity are directed towards increasing the density of the medium, i.e. they are "pushing" in nature with respect to to denser regions of the Universe, including clusters of stars and galaxies. This gives it a meaning far beyond a mere generalization of Newton's law.
### c) Proof of the principle of least action
For the first time this principle was formulated by P. Maupertuis in 1744, proceeding from the theological ideas of that time that all processes occurring in nature occur with a specific purpose and proceed in the most rational (economical) way. Only after quite a long time, thanks to L. Euler and J. Lagrange, this principle acquired a concrete mathematical meaning, asserting that for the actual path of a material point in a conservative force field, the integral of the momentum of a particle, taken along a segment of the trajectory between any of its two points, is minimal in comparison with the same integrals taken over the segments of other curves.
$L(r_{i},p_{i},t)$ a function of the time $t$ and made it possible to write down the mentioned principle in the form of the requirement for the minimality of some functional
$$
\Phi(t) = \int L(r_i, p_i, t)\,dt = \min.
$$
For the first time this principle was formulated by P. Maupertuis in 1744, proceeding from the theological ideas of that time that all processes occurring in nature occur with a specific purpose and proceed in the most rational (economical) way. Only after quite a long time, thanks to L. Euler and J. Lagrange, this principle acquired a concrete mathematical meaning, asserting that for the actual path of a material point in a conservative force field, the integral of the momentum of a particle, taken along a segment of the trajectory between any of its two points, is minimal in comparison with the same integrals taken over the segments of other curves.
For the first time this principle was formulated by P. Maupertuis in 1744, proceeding from the theological ideas of that time that all processes occurring in nature occur with a specific purpose and proceed in the most rational (economical) way. Only after quite a long time, thanks to L. Euler and J. Lagrange, this principle acquired a concrete mathematical meaning, asserting that for the actual path of a material point in a conservative force field, the integral of the momentum of a particle, taken along a segment of the trajectory between any of its two points, is minimal in comparison with the same integrals taken over the segments of other curves.
The first who gave the principle of least action the status of a general law of mechanics was H. Helmholtz [27]. He took the "Lagrangian" $L(r_{i}, p_{i}, t)$ as the initial value as the difference between the kinetic $E^{\kappa}$ and potential $E^{p}$ energy of the system, expressed in terms of the generalized coordinates $r_{i}(t)$ and momenta $P_{i}(t)$ of all particles of the system. This made the Lagrangian a function of the time $t$ and made it possible to write down the mentioned principle in the form of the requirement for the minimality of some functional
$$
\Phi (t) = \int L \left(r _ {i}, p _ {i}, t\right) d t = \min .
$$
From the properties of the extremum of this function, Helmholtz succeeded in deriving the laws of motion for a number of systems. Gradually, this method of finding the laws of motion from a single mathematical function became one of the most widely used and most important physical principles, which, in capable hands, turned out to be applicable to most sections of theoretical physics [28].
Nevertheless, attempts to derive this principle from the general laws of the mechanics of conservative systems have so far been unsuccessful. It is all the more interesting to show that it is a consequence of the nonentropy evolution criteria proposed by energy dynamics [29]. To do this, we express the current nonequilibrium state of an arbitrary system by the integral of the power of the process of moving it away from equilibrium $dU / dt = \Sigma_{j}X_{j}\cdot J_{j}$, caused by external coercion taken from the initial equilibrium state:
$$
U = \Sigma_ {j} \int X _ {j} \cdot J _ {j} d t + U _ {o}, \tag {25}
$$
where $U_{o}$ is the energy of the system in a homogeneous (internally equilibrium) state.
The product $J_{j}dt = \Theta_{j}\overline{\boldsymbol{v}}_{j}dt$ included in this expression is the total differential of the extensive state parameter $Z_{j} = \Theta_{j}\Delta R_{j}$, characterizing the distance of the system from the state of equilibrium of the $j$ -th kind and called in ergodynamics "the moment of energy carrier distribution" $\Theta_{j}$ [5]. These are vector analogs of the very parameters that L. Onsager had in mind when he introduced scalar variables $A_{j}$, the time derivatives of which give flows $J_{j} = dZ_{j} / dt = \Theta_{j}dR_{j} / dt = \Theta_{j}\overline{\boldsymbol{v}}_{j}$. The meaning of the parameters $Z_{j}$ is easy to understand if we pay attention to the change in the position of the center of the quantity $\Theta_{j}$ when its density $\rho_{j}$ is redistributed over the volume of the system. This position in the current (inhomogeneous) $R_{j}$ and the initial (equilibrium) state $R_{j0}$ with density $\rho_{j0}$ is determined in a known way:
$$
R _ {j} = \Theta_ {j} ^ {- 1} \int \mathrm {p} _ {j} r d V; R _ {j o} = \Theta_ {j} ^ {- 1} \int \mathrm {p} _ {j o} r d V, \tag {26}
$$
where $r$ is the running (Euler) coordinate.
This immediately implies that
$$
Z_{j} = \Theta_{j} \left(R_{j^{-}} R_{j o}\right) = \int \left(\rho_{j} - \rho_{j o}\right) r d V,
$$
Thus, the inhomogeneous distribution of any field quantity in the volume occupied by the system is characterized by a displacement $\Delta R_{j} = R_{j^{-}}R_{j o}$ of its center from the equilibrium position coinciding with the center of this volume $V$. It is quite obvious that in a system that tends to establish equilibrium, this is the displacement tends to zero and, in the presence of external compulsion, takes on a minimum value compatible with it. This reflects the expression
$$
U = \Sigma \int X_{j} \, dZ_{j} = \min ,
$$
which would be more correct to call the principle of least coercion, especially since it has nothing to do with the concept of "action" in mechanics as the product of the force $F_{j}$ and the time $dt$ of its action.
The meaning of this principle is quite clear: it reflects the fact that the relaxing system "chooses" from all possible trajectories of the system's motion the one at which it remains closer to equilibrium $(Z_{j},\Delta R_{j} = \min)$. The approach of these parameters to zero $(dZ_{j},dR_{j} < 0)$ is a more visual and informative criterion of involution (approach to equilibrium) of the system for each inherent degree of freedom than the maximum of its entropy. This concern, in particular, the hydrodynamic degree of freedom of the system associated with the motion of the fluid, where $X_{j} = -\nabla v_{j}dZ_{j} = Pdt$. Then (34) takes on the meaning of the minimum of the kinetic energy of the flow $U^{k}$, which also includes the turbulent component. This removes any veil of "mystery" from the PLA. Moreover, it becomes obvious that this circumstance is universal in nature and is true not only for mechanical motion, but for processes of any other nature. It is also valid for non-conservative systems, since it follows from energetodynamics, all equations of which take into account irreversibility [30]. This greatly expands the scope of this principle. In particular, it becomes clear why the laminar flow of a viscous fluid remains stable until the moment of bifurcation associated with the appearance of a new degree of freedom in it - vortex motion, or why, under conditions of invariability of external "compulsion", the flow is steady. It also becomes clear why this principle could not be obtained within the framework of disciplines that are not related to evolution. All this refutes the opinion of I. Prigogine that "dynamics and thermodynamics are two different worlds" [3].
## V. CONCLUSION
1. The appearance in the twentieth century of three new fundamental theories - quantum mechanics (QM), the theory of relativity (RT) and thermodynamics of irreversible processes (TIP), containing poorly consistent and even mutually exclusive provisions, makes it necessary to search for alternative ways of synthesizing new knowledge within the framework of a unified physics.
2. The proposed method for the synthesis of continuum mechanics (MC) and nonequilibrium thermodynamics (TIP) by generalizing the law of conservation of energy to the processes of transfer and transformation of any forms of energy and inhomogeneous systems, with any finite number of degrees of freedom, allows you to do this without additional hypotheses and postulates.
3. A new version of nonequilibrium thermodynamics, called energy dynamics, reveals the inconsistency of the hypothesis of local equilibrium and generalizes the TIP by introducing additional parameters of the heterogeneity of the studied systems (moments of distribution of energy carriers), conjugated with potential gradients, just as these energy carriers themselves-with the generalized potentials of their fields.
4. The proposed approach makes it possible to find the driving forces $X_{j}$ and generalized velocities of various processes (flows $J_{j}$ ), proceeding not from the principle of increasing entropy, but on a more general basis of the law of conservation of energy, without excluding from consideration any (reversible or irreversible) component real processes.
5. Energodynamics allows to give a strictly thermodynamic substantiation of all the provisions of the TIP, without involving for this statistical-mechanical and molecular-kinetic theories, which extends the scope of its applicability to nonlinear processes and states far from equilibrium.
6. The new method proposed by energy dynamics for finding the effects of "superposition" of dissimilar processes makes it possible to further reduce the number of empirical coefficients from $n$ ( $n + 1$ )/2 in TIP to $n$ and give a new explanation of these effects.
7. The application of energy dynamics to the mechanics of the continuum reveals the unity of the nature of all force fields as a consequence of the uneven distribution of their energy carriers and offers a unified method for finding clearly distinguishable driving forces and their moments for a number of processes of mechanical and nonmechanical nature.
8. Approach to mechanics from the standpoint of energy dynamics reveals the existence, along with the force fields, of the fields of their torques, which allows generalizing all three Newton's laws to rotational motion and to irreversible processes generated by the counteraction of active forces on the part of "alien" reaction forces. This makes it possible to take into account irreversibility into the equations of mechanics without revising its foundations.
9. From the main equation of energy dynamics follows the existence of the bipolar law of gravity, which differs from the Newtonian law of gravitation by the presence of forces of both attraction and repulsion, the discovery of the phenomenon of gravitational equilibrium and the "super-strong" nature of gravitational interaction, its short-range action and applicability to continuous media, in which it is impossible to distinguish "Field-forming" bodies.
10. The synthesis of nonequilibrium thermodynamics and continuum mechanics reveals the nature of the principle of least action as a consequence of thermodynamic criteria for the evolution of nonequilibrium systems, dictating to the system the choice of a single process path with a minimum distance from equilibrium. This explains why this principle was not substantiated within the framework of mechanics and expands the scope of its applicability to non-conservative systems and dissipative processes.
11. The heuristic value of the synthesis of nonequilibrium thermodynamics and continuum mechanics consists in the mutual complementarity and enrichment of these disciplines, reducing the number of their initial postulates and expanding the scope of their applicability. The results of this synthesis refute the opinion of I. Prigogine that
dynamics and thermodynamics are two different worlds.
[^1]: The use of IFR excludes the search for a preferential FR. Meanwhile, the very existence of IFR is nothing more than a postulate, since, according to the same A. Poincaré, "we will never have the opportunity to make sure that they all move uniformly and rectilinearly" [20]. _(p.6)_
Generating HTML Viewer...
References
36 Cites in Article
Khamis Hassan M.H.,Volkova O.A.,Evdokimov N.V.,Kamalov Yu.T.,Kamalov T.F. (1965). Model of the Universe without Dark Matter.
L Onsager (1931). Reciprocal relations in irreversible processes.
L Chesalin,G Zastenker,E Krukovskaia,E Lakutina,V Lutsenko,N Shevyrev (1985). Onboard information service of the scientific program in the Russian-Ukrainian project Interball-Prognoz (high-apogee spacecraft Interball-3).
Etkinva (2010). Thermokinetics (Synthesis of Heat Engineering Theoretical Grounds).
Valeriy Etkin (1999). PLANK'S RADIATION LAW AS A CONSEQUENCE OF NONEQUILIBRIUM THERMODYNAMICS.
V Etkin,Energodynamics (2011). Thermodynamic Fundamentals of Synergetics).
V Etkin (2008). Solving the riddles of multiverse from the position of energodynamics.
A Umov,M L Selected Works (1950). Unknown Title.
Jha Poynting (1914). A text-book of physics: electricity and magnetism. Pts. I and II. Static electricity and magnetism.
I Bazarov (1991). Motion in Higher Dimensions.
I Prigogine (1947). Etude Thermodynamique des Phenomenes Irreversibles.
Hbg Cazimir (1945). Unknown Title.
K Denbig (1954). Thermodynamics of Irreversible Processes — Irreversible Thermodynamics.
I Meixner (1954). Zur Thermodynamik der irreversiblen Prozesse.
De Groot,S Mazur,P (1962). Nonequilibrium Thermodynamics.
I Gyarmati (1960). Introduction to Irreversible Thermodynamics.
Rolf Haase (1963). Thermodynamik der Irreversiblen Prozesse.
I Bakhareva (1967). Nonlinear nonequilibrium thermodynamics.
David Jou,José Casas-Vázquez,Georgy Lebon (2010). Extended Irreversible Thermodynamics: Evolution Equations.
Y Demirel (2014). Nonequilibrium Thermodynamics.
Valeriy Etkin (2021). SOLVING THE PROBLEM OF THERMODYNAMIC INEQUALITIES.
Valeriy Etkin (2020). SOLVING THE PROBLEM OF THERMODYNAMIC INEQUALITIES.
Valeriy Etkin (2021). Synthesis of Equilibrium and Non-Equilibrium Thermodynamics.
V Etkin (2021). Nonequilibrium phase transitions.
M Krishtal,A Volkov (1985). Unknown Title.
A Poincaré,; On Science (1983). Russian Summaries.
Isaac Newton (2016). The Principia: The Authoritative Translation.
V Etkin (2014). Generalization of the principles of mechanics.
C (1996). Deterioration and Dissolution.
V Etkin (2018). Mechanics as a Consequence of Energodynamics.
Crawford Waves (1968). Unknown Title.
V Etkin (2016). Gravitational repulsive forces and evolution of the universe.
E Iomdina,O Kiseleva,A Archakov,I Moiseeva,A Stein (1959). The impact of prostaglandin analogues on the biomechanical parameters of corneoscleral shell in primary open angle glaucoma.
Khamis Hassan M.H.,Volkova O.A.,Evdokimov N.V.,Kamalov Yu.T.,Kamalov T.F. (2004). Model of the Universe without Dark Matter.
Valeriy Etkin (2018). CRITERIA OF EVOLUTION AND INVOLUTION OF ANIMATE AND INANIMATE NATURE.
V Etkin (2018). Energodynamic Substantiaton of the Principle Least Action.
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
On the basis of a unified theory of the processes of transfer and transformation of any form of energy, called “energy dynamics” by the author, the possibility of synthesizing the methods of non equilibrium thermodynamics and continuum mechanics is shown. A unified substantiation of the main provisions of both the theory of irreversible processes and classical mechanics, free from postulates and hypotheses, is given. At the same time, a new method for studying real processes is proposed, which does not exclude from consideration any (reversible or irreversible) part of them. This made it possible to generalize all three Newton’s principles, to find a short-range form of his law of gravity, and for the first time to substantiate the principle of least action, which mutually enriches both thermodynamics and mechanics.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.