## I. INTRODUCTION
Though cervical cancer (CC) is largely preventable, it is still the second most common female cancer internationally and the leading cause of cancer deaths among females in African countries[1]. Despite the considerable success of research results based on visual inspection and cytological analyses, cervical cancer still remains a public health problem in Cameroon. Cervical precancerous lesions are often correlated to Human Papilloma Virus infections[2], human papillomavirus (HPV) is currently the most common pathogen responsible for female cancers[3]. Indeed, HPV are viruses that belong to the Papillomaviridae family, consisting of an icosahedral capsid of $55~\mathrm{nm}$ in diameter and a double-stranded DNA of 8,000 base pairs[2]. HPVs have exclusive tropism for metaplastic cells in the junctions of squamous and glandular epithelia and elicit cytopathogenic effects with transformation of keratinocytes into koilocytes [4]. In Cameroon, very few people work on HPV, so very few studies have been conducted. No data are clear concerning viral genotypes circulating in this country and there is no information on risk factors available. In 2009, a US research team headed by Desruisseau, demonstrated that; oncogenic HPV subtypes 45 and 58 were more prevalent in Cameroon [4]. Several other studies conducted in Africa by other researchers had presented different dominant genotypes. In Ethiopia for example, Human papilloma virus type 16 was the most prevalent genotype identified from the subjects screened[5]. In Angola, The most prevalent HPV genotypes were HPV16, HPV6 and HPV 18[6]. Morocco data presented, high rates of infection with HPV genotypes in sexually active Moroccan women making molecular investigation for HPV16, 18 and 31 essential in clinical approach. However, HPV 33, 35 and 45 are less frequent in this population[7], [8]. In Mozambique, among women with cervical cancer HPVs 16 and HPV 18 were the two most frequently identified genotypes $(47.0\%$ and $31.3\%$ respectively), followed by HPV Types 51, 52, 45, 35, 33 and 31[9]. Studies in south Africa demonstrated high prevalence of HPV and multiple HPV infection among HIV-positive women compared to HIV-negative women across all ages [10]. Study carry out in Zambia shows that among high-risk (HR) types, HPV 52 $(37.2\%)$, 58 $(24.1\%)$ and 53 $(20.7\%)$ were more common overall than HPV 16 $(17.2\%)$ and 18 $(13.1\%)$ in women with high-grade squamous intraepithelial lesions or squamous cell carcinoma (SCC) on cytology[11]. All this Human Papilloma Viruses diversity demonstrates the importance of accurately determining HPV genotypes and subtypes that prevail in the sub-Saharan zone, especially in Cameroon.
## II. MATERIAL AND METHODS
### a) Study sites
This was a cross-sectional study, which took place in hospitals in the selected areas of three region of Cameroon (the South, The Far North and the central regions).
### a. District of Niete
Niete is an agro-industrial locality located in southern Cameroon near the Atlantic coast in the Ocean Division and the Southern Region. The commune of Niete has about 40 894 inhabitants in 28 villages. The communal population is composed of 19 137 men, 11 154 women, 5 655 and 4 948 young people aged 5-16 years and less than 5 years respectively. The work was carried out in three subdivision medical centers, namely the V15 hospital, the V4 hospital and the ADJAP hospital.
b.District of Mokolo
Mokolo is a city located in the Far-North region, near the border with Nigeria. It is the county town of the Mayo-Tsanaga Division. The study was conducted at the District Hospital of Mokolo. The commune of Mokolo is one of the largest municipalities in the Far North with an area of $1650~\mathrm{km}^2$ for a population estimated at 310,000 inhabitants in 106 villages. The economy of Mokolo is relied mainly on agriculture (rainy season sorghum, dry season sorghum, groundnut, cowpea, soybean, sweet potato, and vegetable and fruit crops), livestock (cattle, goats, sheep, and poultry), small trade and crafts.
## c. District of Yaoundé 1
The district of Yaounde III, Department of Mfoundi, Central Region. It covers an area of 5552 hectares for a population estimated at 281 586 inhabitants, ie a density of about 507habitants/km2. Gender related statistics estimate 141,525 for males and 140,011 for females; Leading to a sex ratio of $101.05\%$. It is managed by a municipal council of 41 members, a communal executive composed of a mayor and four deputies. There are several hospitals and dispensaries, where we collected the data.
### b) Study population and sample size
These analyses targeted all women with or without cancer at different stages of development.
## i. Selection criteria
# a. Inclusion
All Cameroonian women over the age of 18 were eligible. They should not have undergone hysterectomy, they had to be willing to participate in the study, the must have signed the informed consent form, they must be sexually active.
b.Exclusion
All pregnant women;
All women with cervical cancer.
## ii. Sampling method
A convenient sampling technique in which potential participants were consecutively recruited at the different sites.
## iii. Questionnaire
An investigator administered semi structured questionnaire was used to collect data of each woman through 15-20 minutes' individual interview. The first part of questionnaire focused on sociodemographic information such as age, level of occupation or religion. The second part allowed for collecting obstetric information as well as those related to sex behavior.
## iv. Cervix sample collection and visual inspection
After counseling participants were placed in gynecological position on an examination table. A clean, sterile non-lubricated speculum was gradually introduced into their vagina for eye examination of cervix. 2 Samples were obtained by collecting exfoliated cells, from the transformation zone of the cervix using cytobrush and ayre spatula. Thereafter, these cells from ayre spatula were transferred directly onto a slide and fixed using the conventional technique and the cells from cytobrush were used for PCR. The visual inspection with acetic acid was performed according the atlas of cytology, with Lugol's iodine recommendations. Results were classified as: normal cervix, abnormal cervix, and cervix with suspected cancer. The cytological analysis has been performed using the Papanicolau test, and the Bethesda classification system for interpretation of results has been used.
## v. Cytological analysis
Pap smears were obtained by collecting exfoliated from the transformation zone of the cervix using ayre spatula. The cells were transferred directly to a slide and fixed using the conventional technique. The Bethesda system was used to interpret results from slides.
### c) DNA extraction
The cells collected with cytobrush were used for virus isolation.DNA extraction of fresh cervical cells was made using the QIAGEN extraction kit, which is a commercially available. Extraction was made according to the manufacturer.
### d) HPV genotyping strategy
# a. Primers
The table in supplementary material shows the newly designed primers that were used throughout this study.
The beta globin gene was used to check the quality of the reaction.
DNA sequences of the HPV genotypes targeted obtained from Genbank. The primers were modeled for each type of HPV which the literature showed a high prevalence in Cameroon, as presented above. With the Primer 3 online program (https://primer3.ut.ee/), we were able to generate the primers for 6 low risk genotypes (LR), (6, 11, 61, 62, 70 and 81) and 6 high risk genotypes (HR) (16, 18, 35, 45, 58 and 84)
### b.Mix and PCR
The master mix contains pre-optimized concentrations of HotStarTaq DNA Polymerase and MgCl2, plus dNTP, and a PCR buffer that contains the new MP factor. The use of a master-mix format reduces the time and handling for the reaction configuration and increases the reproducibility by eliminating many possibilities.
The following conditions were used for PCR amplification on a Thermal Cycler (Applied Biosystems): Denaturation for 3 minutes at $91^{\circ}\mathrm{C}$, followed by 42 cycles of 27 seconds at $94^{\circ}\mathrm{C}$, 45 seconds at $50^{\circ}\mathrm{C}$, and 10 minutes at $64^{\circ}\mathrm{C}$, and a final elongation step of 5 minutes at $65^{\circ}\mathrm{C}$. After amplification, the reaction mixture was transfer for electrophoresis to $8^{\circ}\mathrm{C}$.
Analyzing of the PCR product was done using agarose gel electrophoresis, which separates DNA products on the basis of size and charge, it allows for the determination of the presence and the size of the PCR product. Visualization was done using transilluminator.
## c. Control quality
The $\beta$ -globin was a quality control gene for PCR amplification used to show the smooth progress of the PCR. The $\beta$ -globin (-) represents the samples whose $\beta$ -globin internal control gene could not be amplified. In our study, $5.16\%$ of the samples were $\beta$ -globin (-); and as a result were they were declared unsatisfactory. $\beta$ -globin (+) represents samples whose $\beta$ -globin internal control gene has been successfully amplified which accounted for $94.84\%$.
### e) Ethical statement
The study was carried out in conformance with the guidelines for human experimental models in clinical research as stated by the Cameroon Ministry of Public Health and the Helsinki declaration. To do so, ethical clearance was issued by the National Ethics Committee of Cameroun with registration number
2014/08/485/CE/CNERSH/SP. likewise, administrative clearance was issued by regional delegations. The aim and objectives of the study were explained to each woman in the language they understood best (English or French), and their questions were answered. Only women who signed an informed consent form for their participation were enrolled. Participation in the study was strictly voluntary and women were free to decline answering any question or totally withdraw if they so wished at any time.
### f) Statistical analysis
Data were keyed and verified for consistency into Excel spreadsheet and thereafter analyzed with Graph Pad Prism version 6. Independence Chi-square test and one-way ANOVA, were used to compare results. Qualitative variables were presented as percentage with confidence interval at $95\%$ in tables and graphics. XLSTAT 2015 software was used to perform principal component analysis (PCA) in order to establish correlations between $\geq 2$ quantitative variables. Significance was set at $P \leq 0.05$.
## III. RESULTS
### a) Characterization of sample collected
The table 2 below presents some patients with atypical profiles who were selected for molecular analysis and cytology and PCR results. The aim was to clarify the involvement of HPV among lesions observed. Most of cervix cell alterations were represented by ASCUS (43.2%). Out of 1836 $\beta$ -globin positive samples, HPV genotypes were found in 1484 samples giving a HPV prevalence of $76.2\%$. Low Risk HPV genotypes were identified in $75.51\%$ of all positive samples. 297 cases of multiple infections were recorded in the study and were mainly represented by co-infection with genotypes of low risk. This table presents characterization of women for molecular analysis. We selected 1947 women with positive uterine cell alteration according to VIA/VILI or Pap Test. Out of 4063 women fulfilled inclusion criteria. 1947 presented uterine cell alterations according to VIA/VILI or Pap test and were therefore included in this study. The prevalence of ASCUS (61.8%) (P value $< 0.001$ ), LSIL (43.2%) (P value $< 0.001$ ), and HSIL (59.4%) (P value $< 0.001$ ), were higher in women from Yaounde than in their counterparts from, Niete and Mokolo. In addition, ASCH lesions were more frequent in Mokolo (40.6%) as summarized in table below.
Table 1: Cytology and PCR results
<table><tr><td></td><td></td><td>Effective</td><td>Percentage</td><td>95% CI</td><td>P value</td></tr><tr><td colspan="6">Cytology Diagnose</td></tr><tr><td></td><td>ASCUS</td><td>841</td><td>43.2</td><td>35.3-51.7</td><td></td></tr><tr><td></td><td>ASCH</td><td>653</td><td>33.5</td><td>26.1-40.4</td><td></td></tr><tr><td></td><td>LSIL</td><td>319</td><td>16.4</td><td>9.4-23.6</td><td>< 0.001</td></tr><tr><td></td><td>HSIL</td><td>134</td><td>6.9</td><td>2.6-15.1</td><td></td></tr><tr><td colspan="6">β-globine Presence</td></tr><tr><td></td><td>β-globine+</td><td>1836</td><td>94.2</td><td>82.4-97.3</td><td>< 0.001</td></tr><tr><td></td><td>β-globine-</td><td>111</td><td>5.8</td><td>3.2-13.6</td><td></td></tr><tr><td colspan="6">HPV Statut</td></tr><tr><td></td><td>HPV+</td><td>1484</td><td>76.2</td><td>70.1-82.5</td><td></td></tr><tr><td></td><td>HPV-</td><td>256</td><td>13.1</td><td>7.4-17.9</td><td></td></tr><tr><td></td><td>Unsatisfactory smears</td><td>207</td><td>10.6</td><td>4.6-18.3</td><td>< 0.001</td></tr><tr><td colspan="6">HPV Genotypes according to classes</td></tr><tr><td></td><td>LR-HPV</td><td>1138</td><td>75.5</td><td>71.2 - 78.1</td><td></td></tr><tr><td></td><td>HR-HPV</td><td>346</td><td>23.4</td><td>17.3 - 29.5</td><td>< 0.005</td></tr><tr><td colspan="6">Multiple Infections</td></tr><tr><td></td><td>LR-HPV</td><td></td><td></td><td></td><td></td></tr><tr><td></td><td>+</td><td>91</td><td>31</td><td>25.1 - 36.8</td><td>NS</td></tr><tr><td></td><td>HR-HPV</td><td></td><td></td><td></td><td></td></tr><tr><td></td><td>LR-HPV</td><td>144</td><td>48.2</td><td>45.7 - 54.4</td><td></td></tr><tr><td></td><td>HR-HPV</td><td>62</td><td>20.6</td><td>14.7- 24.8</td><td></td></tr><tr><td>Study sites</td><td>ASCUS N (%) (n=1357)</td><td>ASCH N (%) (n=1439)</td><td>LSIL N (%) (n=957)</td><td>HSIL N (%) (n=310)</td><td></td></tr><tr><td>NYETE</td><td>229 (16.8)</td><td>321 (22.3)</td><td>314 (32.8)</td><td>65 (20.9)</td><td></td></tr><tr><td>MOKOLO</td><td>288 (21.2)</td><td>586 (40.7)</td><td>231 (24.1)</td><td>63 (20.3)</td><td></td></tr><tr><td>YAOUNDE</td><td>840 (61.9)</td><td>532 (36.9)</td><td>412 (43)</td><td>182 (58.7)</td><td></td></tr><tr><td>P value</td><td>< 0.001</td><td></td><td></td><td></td><td></td></tr></table>
### b) Frequency of High Risk genotypes (HR-HPV) identified
Table 2 below present different cervical lesions and HR-HPV genotypes identified. The prevalence of low risk genotypes was higher than of high risk ones in ASCUS, ASCH and LSIL lesions. Conversely, high risk genotypes were more frequently associated with HSIL lesions than Low risk genotypes (20%, versus 6.1%) as presented in table 2. However, the difference was not statistically significant (P value=0.0847). ASCUS (60%); was more representative among women with Genotype 16 no patient with LSIL had the HR 16 genotype. The HR 18 genotype was identified in among all women presenting lesions, with the most prevalence for HSIL (30%). HPV 35 was identified mostly among women with LSIL (80%). The final general analysis shows a high frequency of HR 45 genotypes among all the women selected; in fact, HPV 45 was more representative among women who had presented ASCUS (54.5%).
Table 2: HR-HPV genotypes and precancerous cervical lesions
<table><tr><td colspan="2">HR Genotypes</td><td>HPV 16 (n=54)</td><td>HPV 18 (n=100)</td><td>HPV 35 (n=52)</td><td>HPV 45 (n=105)</td><td>HPV 58 (n=0)</td><td>HPV 84 (n=35)</td></tr><tr><td></td><td></td><td>N(%)</td><td>N(%)</td><td>N(%)</td><td>N(%)</td><td>N(%)</td><td>N(%)</td></tr><tr><td rowspan="6">Precancerous cervical Lesions</td><td>ASCUS</td><td>32 (59.3)</td><td>20 (20)</td><td>0(0)</td><td>55 (52.4)</td><td>0 (0)</td><td>11(31.4)</td></tr><tr><td>ASCH</td><td>12 (22.2)</td><td>28 (28)</td><td>0(0)</td><td>10 (9.5)</td><td>0(0)</td><td>9 (25.7)</td></tr><tr><td>LSIL</td><td>0(0)</td><td>24 (24)</td><td>42 (80.8)</td><td>23 (21.0)</td><td>0(0)</td><td>10 (28.6)</td></tr><tr><td>HSIL</td><td>10 (18.5)</td><td>28 (28)</td><td>10 (19.2)</td><td>17 (17.1)</td><td>0(0)</td><td>5 (14.3)</td></tr><tr><td>Total</td><td>54 (100)</td><td>100 (100)</td><td>52 (100)</td><td>105 (100)</td><td>0(0)</td><td>35 (100)</td></tr><tr><td>P value</td><td></td><td></td><td>< 0.001</td><td></td><td></td><td></td></tr></table>
### c) Prevalence of Human papilloma virus infection High risk among Cameroonian women
Figure 1 below presented different high risk virus identified during the study. HPV 45 and HPV 18 were the most represented with $30.3\%$ and $28.9\%$ respectively.
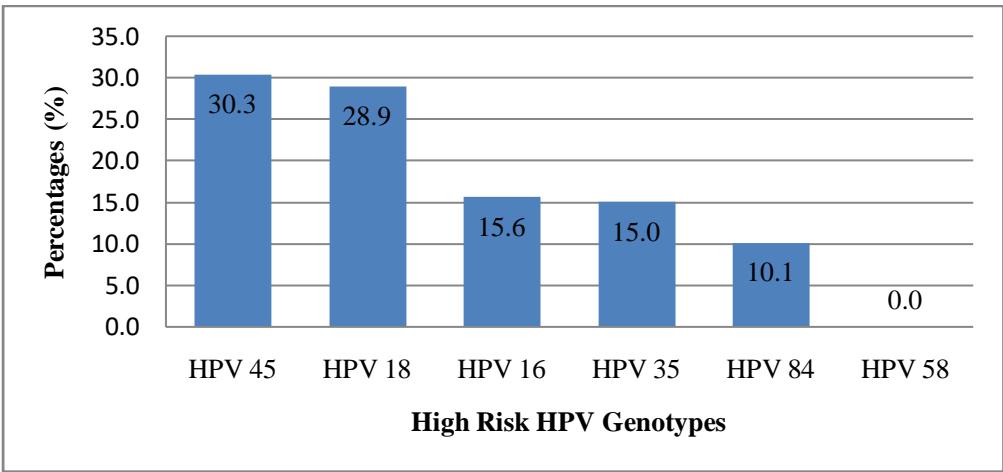 Figure 1: Prevalence of different HR-HPV genotypes targeted in the study population
### d) Frequency of Low Risk genotypes (HR-HPV) identified
Table3 below presented cervical lesion according to HPV genotype. The distribution of all low risk HPV genotypes targeted in this was significantly unbalanced as presented in Table 5 (P-value $< 0.0001$ ).
Table 3: LR-HPV genotypes and precancerous cervical lesions
<table><tr><td colspan="2">LR Genotypes</td><td>HPV 6 (n=294)</td><td>HPV 11 (n=190)</td><td>HPV 61 (n=218)</td><td>HPV 62 (n=138)</td><td>HPV 70 (n=139)</td><td>HPV 81 (n=159)</td></tr><tr><td></td><td></td><td>N (%)</td><td>N (%)</td><td>N (%)</td><td>N (%)</td><td>N (%)</td><td>N (%)</td></tr><tr><td rowspan="6">Precancerous cervical Lesions</td><td>ASCUS</td><td>110 (37.4)</td><td>71 (37.4)</td><td>90 (41.3)</td><td>37 (26.8)</td><td>80 (57.6)</td><td>28 (17.6)</td></tr><tr><td>ASCH</td><td>91 (31)</td><td>25 (13.2)</td><td>50 (22.9)</td><td>26 (18.8)</td><td>52 (37.4)</td><td>69 (43.4)</td></tr><tr><td>LSIL</td><td>83 (28.2)</td><td>52 (27.4)</td><td>78 (35.8)</td><td>75 (54.3)</td><td>0 (0)</td><td>53 (33.3)</td></tr><tr><td>HSIL</td><td>10 (3.4)</td><td>42 (22.1)</td><td>0 (0)</td><td>0 (0)</td><td>7 (5)</td><td>9 (5.7)</td></tr><tr><td>Total</td><td>294 (100)</td><td>190 (100)</td><td>218 (100)</td><td>138 (100)</td><td>139 (100)</td><td>159 (100)</td></tr><tr><td>P value</td><td></td><td></td><td>< 0.001</td><td></td><td></td><td></td></tr></table>
### e) Prevalence of Human papilloma virus infection Low risk among Cameroonian women
Figure 2 below present distribution of HPV. On the six low risk HPV genotypes targeted, HPV 6 and HPV 61 were the most represented with $25.8\%$ and $19.2\%$ respectively.
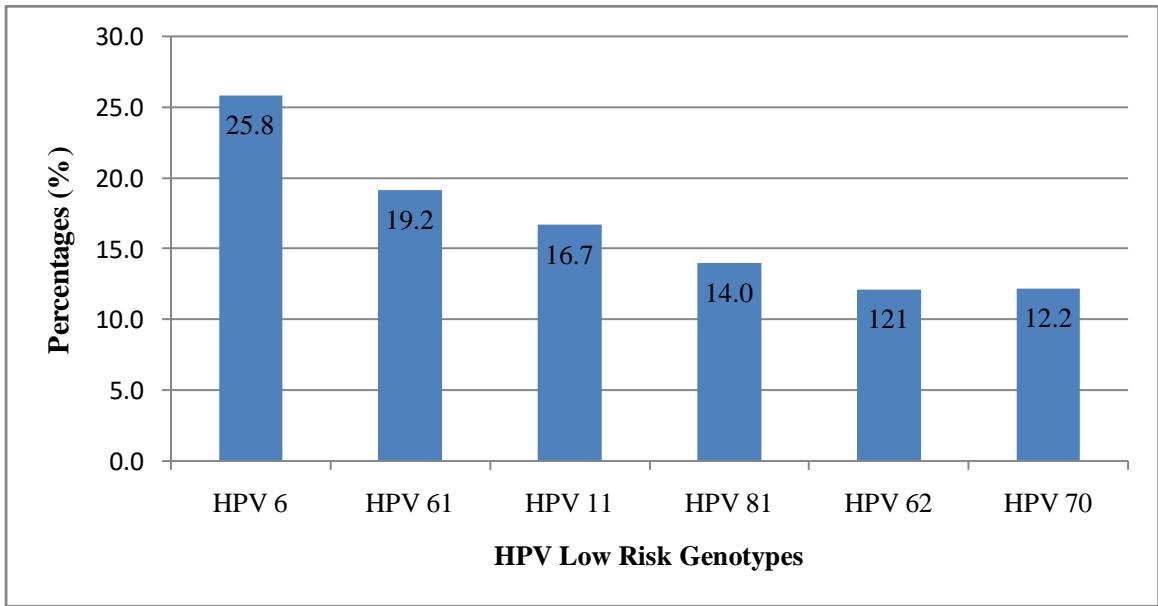 Figure 2: Frequency of distribution of LR-HPV genotypes targeted in the study population
### f) Genotype and cervical lesions general association
This figure3 present association between HPV genotypes and precancerous lesions; according to this PCA (principal component analysis) above, many HPV infected are related to specific cervical cells lesions.
Appearance of ASCUS is most correlated with HPV genotypes 84 (P value $< 0.001$ ). HSIL and LSIL could be respectively associated with HPV genotype 45 and genotype 16 significantly (P value $< 0.001$ ).
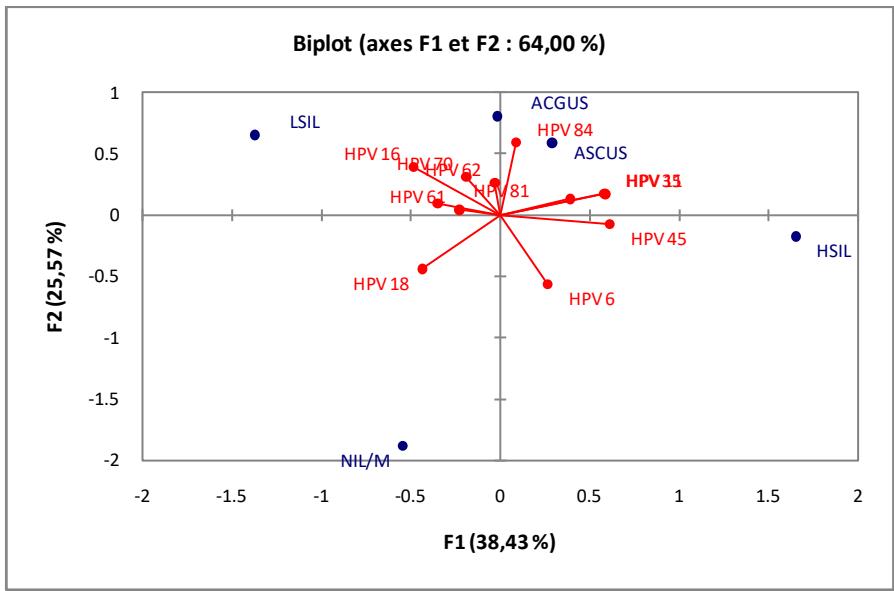 Figure 3: Principal component analysis (PCA) of HPV genotype and precancerous cervical lesions
## IV. DISCUSSION
The aim of this study was to characterize Human Papilloma Virus genotypes and their prevalence among Cameroonian women living with precancerous cervical lesions. For this purpose, additional analyses were carried out in samples of patients with lesions (ASCUS to HSIL). Out of 4063 patients with ASCUS,
ASCH, LSIL and HSIL we selected, 1947samples were PCR positive. Various lesions were identified and were classified according to the Bethesda classification system. $76\%$ of the samples analyzed were positive for at least one genotype of Human Papilloma Virus. However, remaining samples were negative after PCR. This finding is consistent with previous studies [6], [11]. Indeed, HPV presence on these samples presents his part of contribution during carcinogenesis. Multiples infections with HR and LR-HPV accounted for $31.03\%$ of all mixed infections[13]. Low Risk HPV genotype 6 was the most prevalent in Cameroon, previous studies presented similar finding[14].HSIL precancerous lesions were found in $20\%$ of women carrying High Risk HPV; almost identical to those found by Desruisseau and collaborators in 2009 [3]. Assessment of HPV infection according to lesions shows that, more women with cervical lesions were also infected by HPV subtypes; this situation can be explain by capacities of lesions to allow HPV penetration on the cervical cell. Results obtained in Iran showed almost the same results [15]. This frequency of lesions is not unique and china study has presented similar data[16]. The HR HPV 18 genotype was identified among women presenting LSIL and HSIL; many studies identified the HPV 18 as the most characterized on HSIL samples [15], this may be due to the dynamic of the virus to modify his polymorphism according to the environment. The final global analysis shows a high frequency of HR genotypes 45 $(31.42\%)$, followed directly by the genotype 18 $(28.57\%)$. HPV 45 and HPV 18 are two HPV genotypes which belong to HPV High Risk Group; their most prevalence could be attributed to interact with host cell domain and lead to degradation of PDZ-domain-containing proteins through its Carboxy-terminal motif [2].
Among women with low-risk genotypes, the highest frequency was observed among women with indeterminate lesions ASCUS and ASCH (63.9%) as in other studies [16]; this observation present the capacity of the virus to destroy the cervical tissue and to induce dysplasia, as shown Muentes et al.,[18]. However, a study by Castle et al showed that the most prevalent HPV types in the vaginal samples were HPV71 (4.0%), HPV61 (3, 7%) and HPV58 (3.4%) [19]. No patient with genotype 11 had LSIL; this result presents genotype 11 not so implicated in the development of LSIL in this Country, may be because the genotype 11 is identify as low risk and less dangerous. The final general analysis shows a high frequency of LR 6 genotypes (25.4%), followed directly by genotype 61 (18.42%). Genotype 16 and other genotypes have a considerable frequency of occurrence in the study population. The highest frequency of HPV types found in this study were HPV45 and HPV18, HPV6 do not differ too much from those described in Africa[19], in Ethiopia[20], Morocco[7], Uganda [21], Algeria [8]. However, the higher frequency of HPV6 found here is different from previous reports in other African countries that found the most widespread type of HPV, HPV58 in Botswana [22], HPV45 in Cameroon [3], VPH53 in Gabon[23], HPV35 in Burkina Faso [24], HPV2 in Kenya [25]. Multiple infections cases were identified. The HPV-LR 11 genotype was found in two cases of co-infection, as in many studies [26]. Other HPV-LR genotypes were also observed in co-infection cases such as HPV-LR 61/VPH-HR 84 (11.11%); HPV-LR 62/HPV-HR 58 (11.11%); HPV-LR 81/HPV-HR (11.11%). Cuschieri and collabor demonstrated that the prevalence of multiple HPV infections is often high $43.3\%$ and the most prevalent type of HPV multiple infection was only HR-HPV types, with $23.3\%$ and a frequency of multiple infections LR-HPV of $0.8\%$ [18].
## V. CONCLUSION
The aim of this study was to characterize HPV genotypes circulating among Cameroonian women and to identify precancerous cervical lesions involved. Results presented that, Cameroon contain a big diversity of HPV. Global results concerning assessment of prevalence of Low Risk-HPV and High Risk, presented that Human Papilloma Viruses are present in Cameroon with various genotypes, it is important to take into consideration these genotypes during the implementation of prophylactic strategy.
Declarations
### Ethics approval and consent to participate
The study protocol was written based on the Helsinki ethical principles for medical researches and approved by the National Ethics Committee for Human Health Research (n° 2014/08/485/CE/CNERSH/SP).
#### Consent for publication
Informed consent was obtained from all individual participants included in the study
#### Availability of data and materials
#### Funding statement
This study did not receive any funding in any form.
#### ACKNOWLEDGEMENT
The authors are strongly grateful to all women who accepted to participate in the study. They also express their gratitude to all administrative and traditional officials who facilitate the implementation of this study in each study site.
#### Conflict of interest statement
"The authors declare no potential conflicts of interest."
Yuki Yoshimatsu,Tomomi Nakahara,Katsuyuki Tanaka,Yuki Inagawa,Mako Narisawa‐saito,Takashi Yugawa,Shin‐ichi Ohno,Masatoshi Fujita,Hitoshi Nakagama,Tohru Kiyono (2017). Roles of the <scp>PDZ</scp> ‐binding motif of <scp>HPV</scp> 16 E6 protein in oncogenic transformation of human cervical keratinocytes.
A Kombe (2020). Epidemiology and Burden of Human Papillomavirus and Related Diseases, Molecular Pathogenesis, and Vaccine Evaluation.
Andrew Desruisseau,Delf Schmidt-Grimminger,Edith Welty (2009). Epidemiology of HPV in HIV-Positive and HIV-Negative Fertile Women in Cameroon, West Africa.
W Mihret (2014). A pilot study on detection and genotyping of humanpapilloma virus isolated from clinically diagnosed Ethiopian women having cervical intraepithelial neoplasia.
P De,A Damião (2016). Human Papillomavirus types distribution among women with cervical preneoplastic, lesions and cancer in Luanda, Angola.
Zaitouna Alhamany,Mohammed El Mzibri,Aicha Kharbach,Abderrahman Malihy,Redouane Abouqal,Hassan Jaddi,Ali Benomar,Mohammed Attaleb,Najat Lamalmi,Nadia Cherradi (2010). Prevalence of human papillomavirus genotype among Moroccan women during a local screening program.
Noureddine Chaouki,F Bosch,Nubia Muñoz,Chris Meijer,Brahim El Gueddari,Abbes El Ghazi,Judith Deacon,Xavier Castellsagué,Jan Walboomers (1998). The viral origin of cervical cancer in Rabat, Morocco.
Xavier Castellsagué,Joellen Klaustermeier,Carla Carrilho,Ginesa Albero,Jahit Sacarlal,Wim Quint,Bernhard Kleter,Belen Lloveras,Mamudo Ismail,Silvia De Sanjosé,F Bosch,Pedro Alonso,Clara Menéndez (2008). Vaccine‐related HPV genotypes in women with and without cervical cancer in Mozambique: Burden and potential for prevention.
Zizipho Mbulawa,David Coetzee,Anna-Lise Williamson (2015). Human papillomavirus prevalence in South African women and men according to age and human immunodeficiency virus status.
V Sahasrabuddhe,M Mwanahamuntu,S Vermund,W Huh,M Lyon,J Stringer,G Parham (2007). Prevalence and distribution of HPV genotypes among HIV-infected women in Zambia.
Tian-Yu Zhong,Ji-Chun Zhou,Rong Hu,Xiao-Na Fan,Xiao-Ying Xie,Zhao-Xia Liu,Min Lin,Yi-Guo Chen,Xiao-Mei Hu,Wei-Hua Wang,Long Li,Hua-Ping Xiao (2017). Prevalence of human papillomavirus infection among 71,435 women in Jiangxi Province, China.
A Valibeigi (2017). Prevalence of Human Papilloma Virus in Sinonasal Papilloma in Southern Iranian Population.
Themba Ginindza,Cristina Stefan,Joyce Tsoka-Gwegweni,Xolisile Dlamini,Pauline Jolly,Elisabete Weiderpass,Nathalie Broutet,Benn Sartorius (2017). Prevalence and risk factors associated with sexually transmitted infections (STIs) among women of reproductive age in Swaziland.
Mostafa Salehi-Vaziri,Farzin Sadeghi,Firoozeh Hashemi,Hayedeh Haeri,Farah Bokharaei-Salim,Seyed Monavari,Hossein Keyvani (2016). Distribution of Human Papillomavirus Genotypes in Iranian Women According to the Severity of the Cervical Lesion.
Li Ma,Xiao Cong,Mai Shi,Xiu-Hong Wang,Hai-Yan Liu,Mei-Lu Bian (2017). Distribution of human papillomavirus genotypes in cervical lesions.
Rebecca Ogembo,Philimon Gona,Alaina Seymour,Henry Soo-Min Park,Paul Bain,Louise Maranda,Javier Ogembo (2015). Prevalence of Human Papillomavirus Genotypes among African Women with Normal Cervical Cytology and Neoplasia: A Systematic Review and Meta-Analysis.
G Muentes (2016). Genotypes distribution of human papillomavirus in cervical samples of Ecuadorian women.
Philip Castle,Ana Rodriguez,Carolina Porras,Rolando Herrero,Mark Schiffman,Paula Gonzalez,Allan Hildesheim,Robert Burk (2007). A Comparison of Cervical and Vaginal Human Papillomavirus.
Teweldetesfaye Gebremariam (2016). Human Papillomavirus Related Cervical Cancer and Anticipated Vaccination Challenges in Ethiopia.
W Mihret (2014). A pilot study on detection and genotyping of humanpapilloma virus isolated from clinically diagnosed Ethiopian women having cervical intraepithelial neoplasia.
Iain Macleod,Belinda O'donnell,Sikhulile Moyo,Shahin Lockman,Roger Shapiro,Mukendi Kayembe,Erik Van Widenfelt,Joseph Makhema,M Essex,Carolyn Wester (2011). Prevalence of human papillomavirus genotypes and associated cervical squamous intraepithelial lesions in HIV‐infected women in Botswana.
Ali Si‐mohamed,Angélique Ndjoyi‐mbiguino,Kate Cuschieri,Isabelle Onas,Isabelle Colombet,Francis Ozouaki,Jérôme Goff,Heather Cubie,Laurent Bélec (2005). High prevalence of high‐risk oncogenic human papillomaviruses harboring atypical distribution in women of childbearing age living in Libreville, Gabon.
Theodora Zohoncon,Jacques Simpore (2013). PREVALENCE OF HPV HIGH-RISK GENOTYPES IN THREE COHORTS OF WOMEN IN OUAGADOUGOU (BURKINA FASO).
H Vuyst (2010). The prevalence of human papillomavirus infection in Mombasa, Kenya.
H Vuyst (2003). Distribution of human papillomavirus in a family planning population in nairobi, kenya.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Embolo Enyegue Elisée Libert. 2026. \u201cGenetic Profile of Human Papilloma Virus Circulating in Cervical Precancerous-Lesions of Cameroonian Women\u201d. Global Journal of Medical Research - F: Diseases GJMR-F Volume 23 (GJMR Volume 23 Issue F1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.