The adhesion of the polymer paint Lakroten E-244 was investigated on oxidized aluminum A5154 and magnesium MA-20 alloys. It is shown that the adhesion of the polymer coating on the chemically oxidized surface of the aluminum alloy is significantly greater than on the anodic film of the magnesium alloy. Using photoelectron XPS spectroscopy and SEM analysis, the chemical composition and morphology of the alloys have been investigated. The dependence of these factors on the adhesion force of the polymer coating to the oxidized surface of aluminum and magnesium alloys has been established. Methods of modification of conversion coatings with ethylen glycol, which significantly improve the adhesion of polymer layers, have been proposed. The chemical composition and morphology of these coatings have been investigated. An explanation of the mechanism of improvement of adhesion properties of modified coatings on aluminum and magnesium alloys is given. The mechanism of improving the adhesive properties of modified coatings on aluminum and magnesium alloys is explained.
## I. INTRODUCTION
Aluminum and magnesium alloys are widely used as structural materials in the aerospace, electronics and automotive industries due to their low density and high strength. The disadvantage of these alloys is their low corrosion resistance in aqueous electrolytes, which limits their use without special preparation. To protect aluminum and magnesium alloys from corrosion, paints and varnishes are commonly used, which are applied to conversion coatings. To improve the adhesion of aluminum and magnesium alloys to paints, alkaline treatment is often recommended to increase the concentration of -Me-OH atomic groups on their surface. To enhance the adhesive activity of aluminum and magnesium alloys to paints, alkaline treatment is often recommended in order to increase the concentration of Me-OH groups of atoms on their surface. At the same time, an increase in the adhesive force is associated with the chemical interaction of surface metal hydroxides with paintwork components. Most coatings in their composition contain monomers with the general formula R-OH and R-COOH, where R is aliphatic or cyclic hydrocarbons. In this case, the chemical interaction of the paint with metal occurs as a result of the condensation reaction, which can be represented in the form of equations:
$$
\mathrm{Me} - \mathrm{OH} + \mathrm{R} - \mathrm{OH} = \mathrm{Me} - \mathrm{OR} + \mathrm{H2O}
$$
$$
\mathrm{Me} - \mathrm{OH} + \mathrm{R} - \mathrm{COOH} = \mathrm{MeOCOR} + \mathrm{H2O}
$$
Thus, for the paint to form chemical bonds with the conversion coating, chemically active hydroxyl groups of atoms must be present in its composition. Most studies on the modification of conversion coatings are aimed at changing their structure rather than their chemical composition, based on the assumption that the main factor that increases adhesion is the mechanical bonding of the paint to the pore walls. However, the rapid development of technologies based on the use of organosilane primers indicates the significant role of chemical interaction in the formation of strong bonds of coating materials with the surface of metals. Conversion coatings on aluminum and magnesium alloys consist mainly of metal oxides and hydroxides. Only metal hydroxides participate in the formation of chemical bonds by the mechanism of hydrolytic condensation, so the adhesion strength should depend on the MeO/MeOH ratio in the conversion coating. In this work, the chemical compositions of anodized layers on aluminum A5154 and magnesium MA-20 alloys will be investigated by XPS methods in order to determine the influence of the chemical composition of conversion coatings on the adhesion force to paints. The coatings impregnation was carried out with acrylic paint based on Lakroten E-244 dispersion. The copolymer of this dispersion is acrylic acid which can interact with metal hydroxides by reaction (2).
## II. MATERIAL AND METHODS
The work used samples of aluminum and magnesium alloys, the composition of which is shown in Table 1.
Table 1: Compositions of aluminum alloy A 5154 and magnesium alloy MA-20
<table><tr><td></td><td colspan="8">Mass fraction of elements,%</td></tr><tr><td>Alloy grade</td><td>Fe</td><td>Cu</td><td>Mn</td><td>Mg</td><td>Cr</td><td>Zn</td><td>Ce</td><td>Al</td></tr><tr><td>A5154</td><td>0.25</td><td>0.4</td><td>0.1</td><td>0.1</td><td>3.1</td><td>15</td><td>0.2</td><td>93.8</td></tr><tr><td>MA20</td><td>0.05</td><td>0.03</td><td>0.04</td><td>97.94</td><td>1.5</td><td>0.12</td><td>0.02</td><td>0.2</td></tr></table>
The following substances were used to compose inhibitory compositions:
- Ethylene glycol (EG), HO-CH2-CH2-OH (Chem.Russia);
- Vinyltrimethoxysilane (VS) (WitcoCo);
Compositions of oxidation electrolytes of aluminum alloy A 5154 and magnesium alloy MA-20 is shown in Table 2.
Table 2: Compositions of oxidation electrolytes of aluminum alloy A 5154 and magnesium alloy MA-20
<table><tr><td>Sample</td><td>Conversion Coating</td><td>Modified Conversion Coating</td></tr><tr><td>A5154</td><td>35g/1 NaH2PO4, 10gl AlNO3, 5 r/ll NaOH</td><td>35g/1 NaH2PO4, 10gl AlNO3, 5 r/ll NaOH, 50 r/ll EG</td></tr><tr><td>MA20</td><td>55g/1 NaOH, 35g/1 Na2HPO4, 10g/l Al(NO3)3</td><td>55g/1 NaOH, 35g/1 Na2HPO4, 10g/l Al(NO3)3, 50 r/ll EG</td></tr></table>
### a) Adhesion Measurement
 Fig. 1: Electronic adhesion meter PSO-MG4g
- The coating adhesion test during separation was carried out using an electronic adhesion meter PSP-MP4 (Russia), shown in Fig.
1. Samples with conversion coating were placed in a fixed holder. Super glue was applied to the surface of an aluminum rod with a diameter of $10\mathrm{mm}$ and then brought into contact with the test samples. After curing the glue, the joint was stretched at a constant speed of $5\mathrm{mm / min}$. For each test, five repeating samples with the specified mean value were used.
b) X-ray Photoelectron Spectroscopy (XPS) Study
X-ray photoelectronic spectra (XPS) were recorded using the Omicron+ X-ray photoelectron spectrometer (FRG). The pressure in the analyzer chamber did not exceed 10-8 Torr. The radiation of an X-ray Al-anode (radiation energy 1486.6 eV, power 200 W) was used as a source. The transmission energy of the analyzer is set to $20\mathrm{eV}$. The binding energy of the electrons was calibrated according to the XPS peak of C1s electrons, the binding energy of which was assumed to be $285.0\mathrm{eV}$ and which is due to the settled layers of diffusion oil vapor. Photoionization cross sections of the corresponding electron shells taken from [19] were used for quantitative evaluation. The integral peak intensities were obtained after subtracting the background using the Shirley method [20] and by fitting the observed peaks with Gauss curves with the contribution of the Lorentz component. To obtain information about the thicknesses of the layers formed on the surface, the MultiQuant program was used [21], the photoionization sections of the corresponding electron shells given by Schofield [22] were selected. To calculate the thicknesses of the layers, the values of electron free path lengths (or average attenuation


coefficients) calculated by the method proposed by Kampson and Sih were used [23].
### c) SEM of the Study
SEM images were obtained by a JSM-6400 scanning microscope with an electron-beam intensity of 20 keV, unless otherwise written. Analysis of the elements was performed with SEM equipped with energy-dispersive X-ray analysis (EDX) with a WinEDS EUMEX analyzer (Germany). The thickness of the polymer siloxane coatings was determined using the ZAF correction algorithm for Ka-ratios samples with a known coating thickness.
### d) EIS Study
An electrochemical complex consisting of a potentiostat with a frequency analyzer (FRA) is used to measure the impedance. The measurements were carried out in a three-electrode electrochemical cell with a corrosion potential using the Solartron electrochemical complex with a change in the frequency of alternating current from 60,000 to $0.1\mathrm{Hz}$ ( $\Delta E = 10\mathrm{Mv}$ ). The impedance data was analyzed using ZView® electrochemical impedance software (version Scribner Associates, Inc., USA).
## III. EXPERIMENTAL
The adhesion of coating materials to metal surfaces depends mainly on the chemical composition of the surface, the thickness and porosity of the adhesion layer. The thickness and porosity of conversion coatings for aluminum and magnesium alloys differ to a lesser extent than the chemical composition. Anodic coatings on magnesium alloy AZ91D contain mainly $\mathrm{MgO}$, $\mathrm{Al_2O_3}$ and $\mathrm{MgAl_2O_4}$ [24,25]. The adhesion of the paint to such a surface should depend on the thickness and porosity of the conversion coating, since the chemical interaction is minimized. The addition of reagents to anodizing electrolytes that are susceptible to hydrolysis increases the hydroxide ion content of the conversion coating and thus, according to equation (1.2), the adhesion of the coating to the alloy surface. Since the adhesive strength of the coating depends on the chemical composition of the substrate, XPS studies of conversion coatings were carried out.
### a) XPS and SEM Investigation of the Chemical Composition of the Conversion Coating on the MA-20 Alloy
The elemental composition of the conversion coating obtained by anodizing the MA-20 alloy in an alkaline solution is shown in Table 3.
Table 3: The elemental composition of the conversion coating on the MA-20 alloy
<table><tr><td>Sample</td><td>Mg,% at</td><td>Al,% at</td><td>C,% at</td><td>O,% at</td><td>Thickness, mkm</td></tr><tr><td>MA-20</td><td>30.3</td><td>8.4</td><td>19.2</td><td>42.1</td><td>23.0±5.0</td></tr></table>
From the results of the chemical analysis given in Table 2, it can be seen that the composition of the conversion coating includes significant amounts of aluminum compounds that fall into the coating from the substrate or electrolyte during oxidation. Figure 2 shows the XPS spectra of Mg2p and Al2p in the conversion coating. After decomposition of the spectra into components, it is seen that the conversion coating consists mainly of magnesium and aluminum oxides. A study of the adhesion of acrylic paint on anodised magnesium alloy surfaces showed extremely low adhesion strength. To increase the adhesive strength of aluminium and magnesium alloys, it is often recommended to hydroxylate the surface by treatment in alkaline solutions. It is possible to increase the concentration of adhesively active R-OH radicals in the conversion coating by adding polyfunctional alcohols to the anodizing electrolytes.
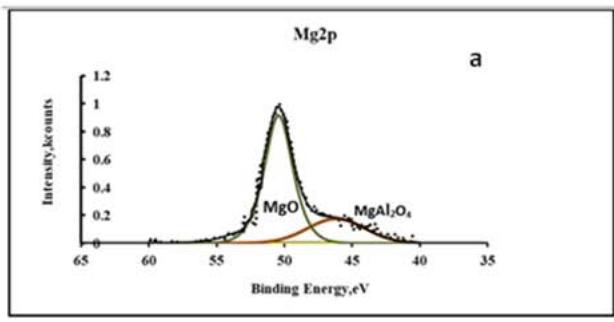
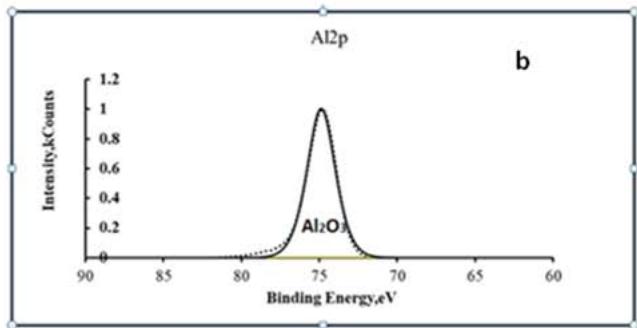 Fig. 2: X-ray spectra of Mg2p (a) and C1s (b) in the conversion coating
In the articles [26,27], studies were carried out on the anodizing of magnesium alloys in aqueous solutions of ethylene glycol and glycerin. The authors of these works found that the conversion coatings obtained in these solutions are formed at sufficiently high speeds, have a porous structure, have high hardness and wear resistance. However, studies of the chemical composition and adhesive strength were not carried out in these works. Morphology and chemical composition of the magnesium layers obtained with ethylene glycol additives in anodizing solutions were studied. Figure 4 shows SEM images of the surface of the anodized alloy before and after modification with ethylene glycol.

 Fig. 3: SEM images of the surface of the anodized alloy before and after modification with ethylene glycol
The addition of ethylene glycol to the electrolyte leads to an increase in the roughness and thickness of the coating. The increase in roughness is apparently due to the formation of magnesium glycolate macromolecules [29] linked together by a bridging bond. Table 4 shows the chemical composition of the modified composite coating.
Table 4: Elemental composition of the modified composite coating
<table><tr><td>Sample</td><td>Mg,% ar.</td><td>Al,% ar.</td><td>C,% ar.</td><td>O,% ar.</td><td>Thickness, mkm</td></tr><tr><td>MA-20</td><td>22,3</td><td>2.4</td><td>43.2</td><td>32,1</td><td>32.4.0±5.0</td></tr></table>
The results of chemical analysis show that the composite coating contains a significant amount of carbon. According to [29], magnesium hydroxide molecules interact with ethylene glycol to form magnesium ethylene glycolate by reaction (3). Magnesium glycolates can interact with each other to form macromolecules by reaction (4).
$$
\begin{array}{c c c} & \text{OH} \\2\mathrm{Mg}_{\mathrm{OH}}^{-\mathrm{OH}} + \mathrm{H}-\mathrm{O}-\mathrm{CH}_{2} & \mathrm{Mg}-\mathrm{O}-\mathrm{CH}_{2} + 2\mathrm{H}_{2}\mathrm{O} \\\mathrm{OH} & \mathrm{H}-\mathrm{O}-\mathrm{CH}_{2} & \mathrm{Mg}-\mathrm{O}-\mathrm{CH}_{2} \\& & \mathrm{OH} \end{array} \tag{3}
$$
$$
n[\mathrm{HO}-\mathrm{MgO}(\mathrm{CH}_{2})\mathrm{OMg}-\mathrm{OH}] \longrightarrow [ -\mathrm{O}-\mathrm{Mg}-\mathrm{O}(\mathrm{CH}_{2})\mathrm{OMgO}- ]_{n} + n\mathrm{H}_{2}\mathrm{O}
$$
Macromolecules, together with magnesium oxides, are embedded in the structure of globules that make up conversion coating [29,30]. Polyglycolates contain in their composition adhesive active groups - OH, through which interaction with paints and individual globules with each other is carried out. On the XPS spectra of Mg2, magnesium ethylene glycolates can be observed by the appearance of a peak with $E_{\mathrm{b}} = 53.5$ eV (Fig. 5), and on the spectra of C1s with $E_{\mathrm{b}} = 288.2$ eV (Fig. 6).
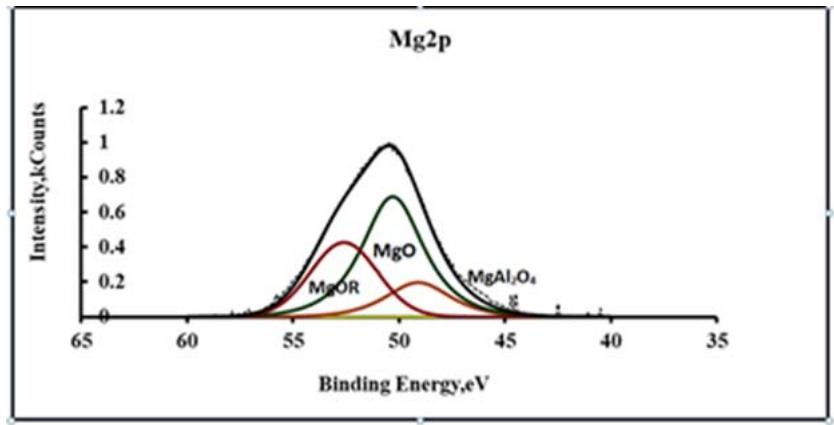 Fig. 4: XPS spectra of Mg2p in a modified inversion coating
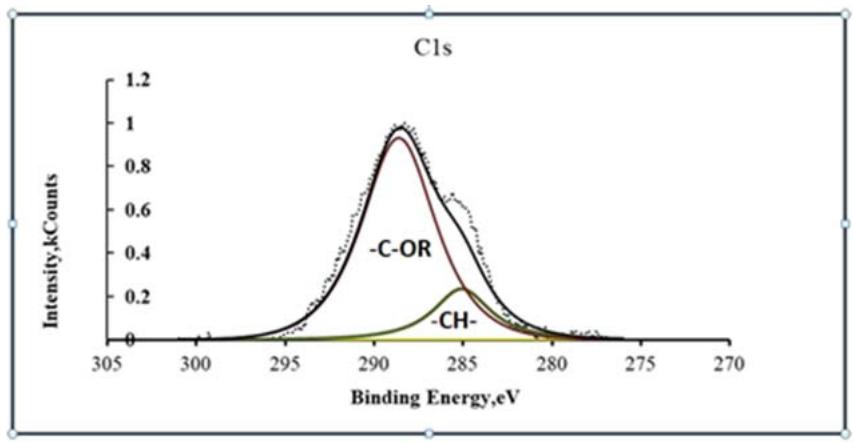 Fig. 5: XPS spectra of C1S carbon in a modified conversion coating
The amounts of ethylene glycolates and magnesium hydroxides in the original and modified conversion coating are shown in Table 5.
Table 5: Chemical composition and tensile strength of the initial and modified MA-20 alloy
<table><tr><td>Type of coating</td><td>MgO,% ar.</td><td>Mg glycolate% ar.</td><td>MgAlO4% ar.</td><td>Adhesion strength KPa</td></tr><tr><td>1. Initial coating</td><td>84,6</td><td>-</td><td>15.4</td><td>300±50</td></tr><tr><td>2. Modified</td><td>57.2</td><td>36.2</td><td>18.6</td><td>1180±50</td></tr></table>
The inclusion of magnesium glycolates in the conversion coating should change the concentration of chemically active groups -OH. Figure 6 shows the XPS spectra of O1s oxygen in the modified conversion coating.
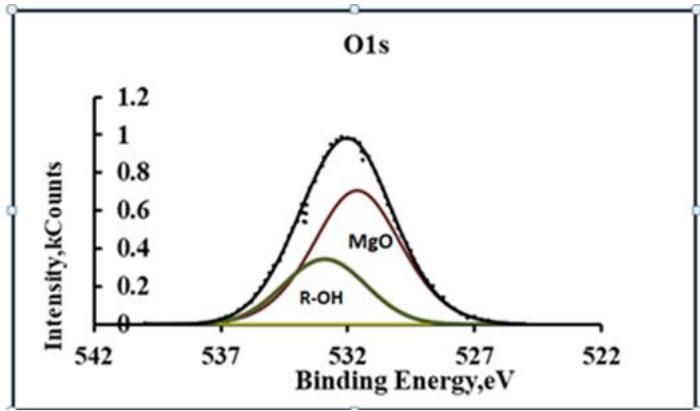 Fig. 6: XPS-spectra of O1s oxygen in the modified conversion coating
Quantitative calculations have shown that the ratio of $[O] / [OH]$ concentrations in the conversion coating changes towards an increase in hydroxide ions mainly due to magnesium ethylene glycolate. An increase in the adhesive strength of the modified conversion coating may be due to an increase in roughness or due to the formation of chemical bonds with the paint. With the help of vapor-gas deposition of vinyltrimethoxysilane (VS) [31], its roughness was reduced on the surface of the modified conversion coating. The coating thickness was 150-200 nm. To assess the roughness of the conversion coating, EIS studies were conducted.
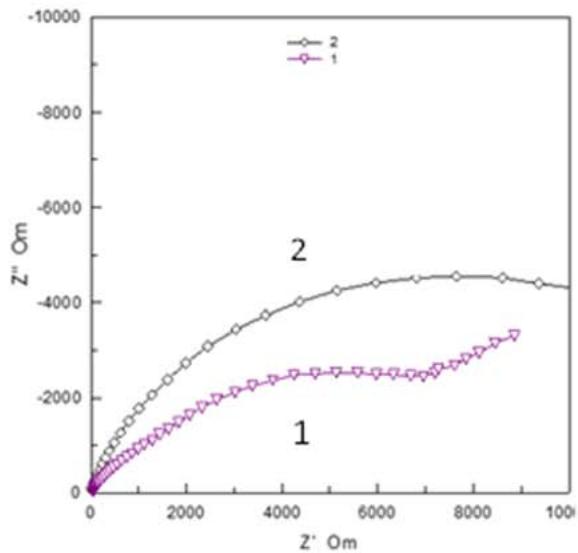
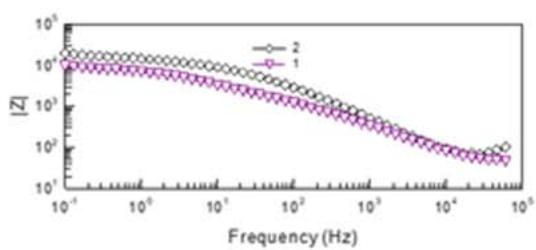
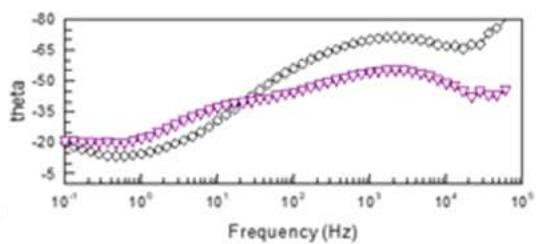 Fig. 7: Nyquist and Bode plots for modified conversion coating (plot 1) and after VS impregnation (plot 2)
In plot 1 (Fig. 7), there is a rectilinear section with an angle of $45^{\circ}$ in the high frequency region. This form of the Nyquist graph is characteristic of mass transfer in porous systems. After impregnation, the Nyquist graph has the form of a hemisphere, characteristic of a smoothed surface. The Bode graphs for the initial conversion coverage show the presence of two well-defined time constants. In the high frequency range the time constant can be explained by the barrier properties of coatings. In the low-frequency region, 10-50 Hz is attributed to corrosion processes at the boundary of the coating and metal. After the vapor-gas modification of the conversion coverage, the through pores are filled with polymer and a wide area of change in the phase angle of the barrier coating remains. Silane coating practically does not change |Z|, which indicates partial filling of the pores and a slight change in the structure of the coating. The decrease in the roughness and pore volume of the conversion coverage should have reduced the adhesive force at the same time, it increased after steam-gas treatment by \\sim 20-30%, which indicates the predominant influence of chemical interaction over mechanical. Impregnation of the coatings with vinylsilane increases the number of functional groups involved in the formation of chemical bonds in connection with which it can be used to enhance the adhesion of the coating with paint. Table 6 shows the adhesion strength of Lakroten E-244 to oxidized alloys MA-20 and A5154 with paint.
Table 6: Adhesive strength of Lakroten E-244 to oxidized alloys MA-20 and A5154
<table><tr><td>Conversion coverage on MA-20</td><td>Adhesion strength, KPa</td></tr><tr><td>1. Initial</td><td>300±50</td></tr><tr><td>2. Modified with ethylene glycol</td><td>1180±50</td></tr><tr><td>3. Modified with ethylene glycol+VS</td><td>1350±100</td></tr><tr><td>Conversion coverage on A 5154</td><td></td></tr><tr><td>4. Initial</td><td>1600±100</td></tr><tr><td>5. Conversion coverage on A 5154</td><td>1850±100</td></tr></table>
From the data given in Table 6, it can be seen that the adhesion of Lakroten E-244 on the modified chemically oxidized alloy A5154 is maximum. To clarify the influence of the morphology and chemical composition of the aluminum alloy on the adhesion of the paint, XPS and SEM studies were conducted.
### b) XPS and SEM Study of the Chemical Composition of the Conversion Coating on Aluminum Alloy A5154
The conversion coating on chemically oxidized alloy A5154 consists mainly of $\mathrm{Al(OH)}_4$, which is obtained by the equation:
$$
\mathrm {A l} _ {2} \mathrm {O} _ {3} + 2 \mathrm {O H} ^ {-} + 3 \mathrm {H} _ {2} \mathrm {O} \rightarrow 2 \mathrm {A l} (\mathrm {O H}) _ {4} \tag {5}
$$
This explains the high adhesion of coatings to these alloys. Chemical oxidation is used along with electrochemical, but is inferior to the latter in terms of performance characteristics, primarily mechanical strength and wear resistance. Modification of a chemically oxidized conversion coating with ethylene glycol can increase its mechanical properties due to additional chemical bonds between individual amorphous $\mathrm{Al(OH)}_4$ globules of which conversion coating consists [32]. Along with high adhesive activity, such coatings could significantly expand their application areas.
The conversion coating on alloy A 5154 was modified with ethylene glycol by adding it to oxidation solutions. Figure 8 shows a SEM images of the surface of the conversion coating on the original and modified alloy.
 Panel label: a.
 b Fig. 8: SEM images of the surface of the conversion coating on the original (a) and modified (b) alloy
It can be seen that the structure of conversion coating A5154 is fine-grained with vertical pores of 5-10 microns in size, in contrast to magnesium MA-20. On the modified alloy the structure is also granular but the number and diameter of the pores are larger. Inclusions of white colour according to X-ray microanalysis data are microphases of magnesium glycolate. The chemical composition of the alloys is given in Table 7.
Table 7: Chemical composition of the initial and modified conversion coating on alloy A5154
<table><tr><td>Conversion coating on alloy A5154</td><td>Mg,% ar.</td><td>Al,% ar.</td><td>C,% ar.</td><td>O,% ar.</td><td>Thickness, mKm</td></tr><tr><td>Initial</td><td>2,3</td><td>14.4</td><td>25.1</td><td>58.2</td><td>3.2</td></tr><tr><td>Modified</td><td>1.8</td><td>11.6</td><td>42.8</td><td>43.8</td><td>2.8</td></tr></table>
Comparing the chemical analysis data of the original and modified coating, it is clear that the latter contains a significant amount of carbon belonging to magnesium and aluminum glycolates. Aluminum glycolates can be detected by the appearance of a peak on the Al 2p spectra with an energy of $71.4\mathrm{eV}$ and a peak with an energy of $288.2\mathrm{eV}$ on the C1s spectra (Fig. 9).

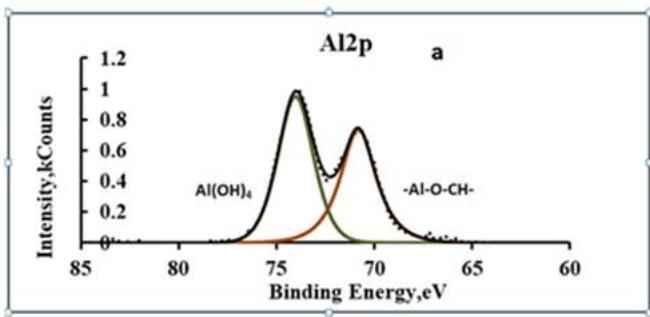
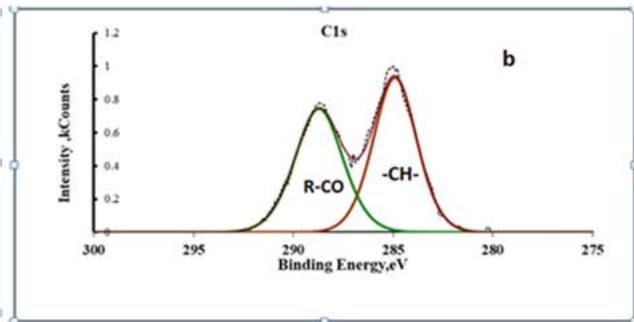 Fig. 9: XPS spectra of Al 2p(a) and C1s (b) for a modified conversion coating on alloy A 5154
A symmetrical doublet on the Al2p spectrum indicates the existence of two types of chemical compounds of aluminum distributed in the conversion coating as separate phases. One of these phases is $\mathrm{Al(OH)}_4$, the other is aluminum glycolate. Quantitative calculations of the XPS spectra belonging to aluminum hycolates have shown that two aluminum atoms account for approximately the same number of carbon atoms. This ratio corresponds to the reaction of the formation of aluminum glycolate according to equation (6).
$$
\begin{array}{c c c c c}&&&\mathrm{HO}&\mathrm{OH}\\&\mathrm{OH}&\mathrm{HO-CH_{2}}&\mathrm{Al-O-CH_{2}}\\2\mathrm{HO-Al}&+&|&\rightarrow&|\\&\mathrm{OH}&\mathrm{HO-CH_{2}}&\mathrm{Al-O-CH_{2}}\\&&&\mathrm{HO}&\mathrm{OH}\end{array}+2\mathrm{H_{2}O}\tag{6}
$$
Aluminum glycolate molecules can interact with each other to form macromolecules and individual phases in the conversion coating. A significant amount of aluminum glycolate in the coating indicates a high chemical activity of ethylene glycol to aluminum hydroxides. The inclusion of aluminum glycolate in the composition of the conversion coating changes its structure and possibly mechanical properties in the direction of increasing strength due to the formation of new chemical bond. The adhesive strength of the modified conversion coating is slightly higher than the initial one due to mechanical adhesion, since the concentration of chemically active $-\mathrm{OH}$ groups varies slightly.
## IV. RESULTS AND DISCUSSION
Published studies of paint adhesion to aluminium and magnesium alloys are random and adhesion strength is mainly attributed to mechanical adhesion in the pore volume. At the same time, our studies have shown that the adhesion strength of paint to the surface of these alloys can vary by an order of magnitude. In this work, the surface of the alloys was subjected to chemical and anodic oxidation. Conversion coatings on aluminium alloy A5154 were obtained by chemical oxidation in alkaline solutions. Oxide coatings on magnesium alloys Ma-20 were obtained by anodising in alkaline solutions. From the point of view of the theory of mechanical adhesion of paints with the alloy surface, conversion coatings on Ma-20 alloy are more preferable because they have a more developed structure on the surface and inside the oxide layer. However, studies have shown that the adhesion force on aluminium alloys is 5-6 times greater than on magnesium alloys. It has been suggested that this is due to the chemical composition of the conversion layers. On aluminium alloys they consist of aluminium hydroxides $\mathrm{Al(OH)_4}$ and on magnesium alloys of oxides. According to equations (1,2) the formation of strong chemical bonds with paints is possible only on aluminium alloy. Modification of the conversion coating with ethylene glycol leads to an increase in its concentration of -OH groups capable of chemical interaction with paintwork materials, as a result of which the adhesive force increases. It is possible to increase the concentration of hydroxides in the coating by introducing compounds that form -R-OH groups of atoms as a result of hydrolysis, such as organosilanes, into the anodising electrolyte. Modification of aluminium alloys with ethylene glycol does not lead to a significant increase in adhesion strength because the conversion coating consists of aluminium hydroxides. However, there is an increase in the number and size of pores, which increases the adhesive strength.
## V. CONCLUSIONS
Studies of the adhesion of the acrylic paint Lakroten E-244 to aluminum A5154 and magnesium Ma-20 alloys have been carried out. Significant differences in the adhesive activity of these alloys to polymer coatings were found. With the help of XPS and SEM, studies of the chemical composition and morphology of alloys were carried out. It is shown that the adhesion of coatings to the surface of alloys depends mainly on the ratio of MeO/MeO in the conversion coating. Modification of the conversion coating on a magnesium alloy with ethylene glycol significantly increases the adhesive strength of the alloy to paints. The chemical composition of magnesium ethylene glycolate macromolecules and their amount in the conversion coating were determined with the help of XPS studies. The addition of ethylene glycol increases the thickness of the conversion coating and its strength due to the formation of additional chemical bonds of magnesium ethylene glycol macromolecules with inside and on the border of globules. Studies of the chemical composition, morphology and adhesive activity of conversion coatings on the surface of aluminum alloys modified with ethylene glycol have been carried out. It is shown that the increase in the adhesive activity of modified aluminum alloys is mainly due to an increase in the number and size of pores. The chemical composition and quantity have been established macromolecules of aluminum ethylene glycolate in the conversion coating of aluminum alloy. Modification of the ethylene glycol conversion coating on magnesium and aluminum alloys changes their structure and chemical composition, which contributes to a significant increase in their adhesive activity.
Generating HTML Viewer...
References
31 Cites in Article
S Kulinich,A Akhtar (2012). On conversion coating treatments to replace chromating for Al alloys: Recent developments and possible future directions.
J-P Dasquet,D Caillard,E Conforto,J-P Bonino,R Bes (2000). Investigation of the anodic oxide layer on 1050 and 2024T3 aluminium alloys by electron microscopy and electrochemical impedance spectroscopy.
J Yoganandan,Balaraju Synergistic effect of V and Mn oxyanions for the corrosion protection of anodized aerospace aluminum alloy.
S Golru,M Attar,B Ramezanzadeh (2015). Effects of surface treatment of aluminium alloy 1050 on the adhesion and anticorrosion properties of the epoxy coating.
Wen Zhu,Wenfang Li,Songlin Mu,Nianqing Fu,Zhongmiao Liao (2017). Comparative study on Ti/Zr/V and chromate conversion treated aluminum alloys: Anti-corrosion performance and epoxy coating adhesion properties.
Y Li,P Zhang,P Bai,L Wu,B Liu,Z Zhao (2018). Microstructure and properties of Ti/TiBCN coating on 7075 aluminum alloy by laser cladding.
M Sabouri,S Mousavi Khoei (2018). Plasma electrolytic oxidation in the presence of multiwall carbon nanotubes on aluminum substrate: Morphological and corrosion studies.
N Meis,L Van Der Ven,R Van Benthem,G De With (2014). Extreme wet adhesion of a novel epoxy-amine coating on aluminum alloy 2024-T3.
Fabiola Pinedac,Carola Martínezb,Mamié Sancyb,Marcela Floresf,María Encinasg,Maritza Páeza (2019). Improving the interaction between aluminum surfaces and polymer coating.
Ruiyue Zhang,Shu Cai,Guohua Xu,Huan Zhao,Yan Li,Xuexin Wang,Kai Huang,Mengguo Ren,Xiaodong Wu (2014). Crack self-healing of phytic acid conversion coating on AZ31 magnesium alloy by heat treatment and the corrosion resistance.
R Zhang,J Bu,C Lin,G Song (2012). Recent progress in corrosion protection of magnesium alloys by organic coatings.
R Zhang,S Zhang A B,N Yang,L Yao B,F He,Y Zhou B,X Xu B,L Chang B,S Bai (2012). Journal of Alloys and Compounds Influence of 8hydroxyquinoline on properties of anodic coatings obtained by micro arc oxidation on AZ91 magnesium alloys.
P Srinivasan,J Liang,C Blawert,M Störmer,W Dietzel (2010). Characterization of calcium containing plasma electrolytic oxidation coatings on AM50 magnesium alloy.
Ximei Wang,Liqun Zhu,Weiping Li,Huicong Liu,Yihong Li (2009). Effects of half-wave and full-wave power source on the anodic oxidation process on AZ91D magnesium alloy.
R Zhang A B,Yu Zuo,Guochao Nie (2020). The enhanced properties of anodic films on AZ91D magnesium alloy by addition of oxide nanoparticles.
Gen Zhang,Aitao Tang,Liang Wu,Zeyu Zhang,Hongxin Liao,Ying Long,Lingjie Li,Andrej Atrens,Fusheng Pan (2019). In-situ grown super- or hydrophobic Mg-Al layered double hydroxides films on the anodized magnesium alloy to improve corrosion properties.
Akeem Yusuf Adesina B,A Madhan Kumar B,Mohammad Mizanur Rahman B,J Ram (2020). Enhancing the biodegradability and surface protective performance of AZ31 Mg alloy using polypyrrole/gelatin composite coatings with anodized Mg surface.
Yurong Ding,Xing Guo,Yong Jiang,Xiao Zeng,Wen Ding,Yan Zhu (2005). Effects of Triethanolamine on the Performances of the Anodicing Film of AZ31B Magnesium Alloy.
D Shirley (1972). High-resolution X-ray photoemission spectrum of the valence bands of gold.
M Mohai (2004). XPS MultiQuant: multimodel XPS quantification software.
R Waldo (1988). An Iteration Procedure to Calculate Film Compositions and Thicknesses in Electron-Probe Microanalysis.
P Cumpson,M Seah (1997). Elastic Scattering Corrections in AES and XPS. II. Estimating Attenuation Lengths and Conditions Required for their Valid Use in Overlayer/Substrate Experiments.
Pei Zhang,Yu Zuo,Guochao Nie (2020). The enhanced properties of anodic films on AZ91D magnesium alloy by addition of oxide nanoparticles.
H Hsiao,W Tsai (2005). Characterization of anodic films formed on AZ91D magnesium alloy.
Hiroki Habazaki,Fumitaka Kataoka,Khurram Shahzad,Etsushi Tsuji,Yoshitaka Aoki,Shinji Nagata,Peter Skeldon,George Thompson (2015). Growth of barrier-type anodic films on magnesium in ethylene glycol electrolytes containing fluoride and water.
A Němcová,O Galal,P Skeldon,I Kuběna,M Šmíd,E Briand,I Vickridge,J-J Ganem,H Habazaki (2016). Film growth and alloy enrichment during anodizing AZ31 magnesium alloy in fluoride/glycerol electrolytes of a range of water contents.
Makarychev Yu,Yu Kuzenkov,D Chugunov,O Grafov,Yu,A Aliev (2023). Vapor-phase deposition of polymer siloxane coatings on aluminum and magnesium alloys.
M Santamaria,F Di Quarto,S Zanna,P Marcus (2007). Initial surface film on magnesium metal: A characterization by X-ray photoelectron spectroscopy (XPS) and photocurrent spectroscopy (PCS).
Wanqiu Zhou,Dayong Shan,En-Hou Han,Wei Ke (2008). Structure and formation mechanism of phosphate conversion coating on die-cast AZ91D magnesium alloy.
Makarychev Yu,B Luchkin,A Yu,Grafov Yu,Andreev Vapor-phase deposition of polymer siloxane coatings on the surface of copper and lowcarbon steel.
G Yoganandan,J Balaraju Synergistic effect of V and Mn oxyanions for the corrosion protection of anodized aerospace aluminum alloy.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Makarychev Yu.B.. 2026. \u201cStudies of the Mechanism of Adhesion of Polymer Coatings on the Oxidized Surface of Aluminum and Magnesium Alloys\u201d. Global Journal of Research in Engineering - A : Mechanical & Mechanics GJRE-A Volume 24 (GJRE Volume 24 Issue A1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.