## I. INTRODUCTION
Laboratory diagnosis of bacteremia and imaging results are among the most reliable combinations for the diagnosis of infective endocarditis (IE). However, conventional microbial blood cultures in IE patients are frequently non-diagnostic, with both false positive and false negative tests. These may be caused by a number of confounding factors, such as growth difficulties, sampling issues, suspected pathogen characteristics, or inadequate preculture treatment strategies (early and inappropriate antimicrobial treatment). Given the same factors, it is also advisable to exercise caution when interpreting positive tests. $^{1-5}$
Venous blood culture represents the most common diagnostic tool currently available for the detection of bacteremia. However, the diagnostic reliability of blood cultures may potentially be constrained despite the unambiguous clinical presentation of generalized infection and sepsis. To overcome this limitation, the Russian and international clinical guidelines recommend collecting at least three blood samples (or preferably to obtain three positive identical blood cultures). Nevertheless, a considerable proportion of conventional blood cultures (16–80%) remain inconclusive, with both false negative and false positive results.[1,6,7] The difficulty in culturing certain microorganisms and the challenges in interpreting the results obtained are the two main reasons for this.[6,8] The advances in alternative diagnostic approaches, such as polymerase chain reaction (PCR) and sequencing, have not significantly altered the landscape of etiological diagnosis for IE. The ambiguity of the results[6,9-12] has reserved these diagnostic tools as supplementary options in both Russian and international clinical guidelines.[1,13]
Efforts to increase the probability of true positive blood cultures based on the infectious biology, etiology, and pathogenesis of IE have encouraged the authors of this paper to initiate both venous (as is generally accepted) and arterial blood sampling for microbiology. This was based on the assumption that the left heart (aortic and/or mitral valves, less often left atrial or left ventricular mural vegetations) is the most frequent location of IE. From these sites, bacteria are occasionally released into the arteries of the systemic circulation. The subsequent mechanical and biological blood filtration occurs through tissue ultracapillaries. This results in the blood passing into the venules and veins with only minimal microbial contamination.[2,6,14]

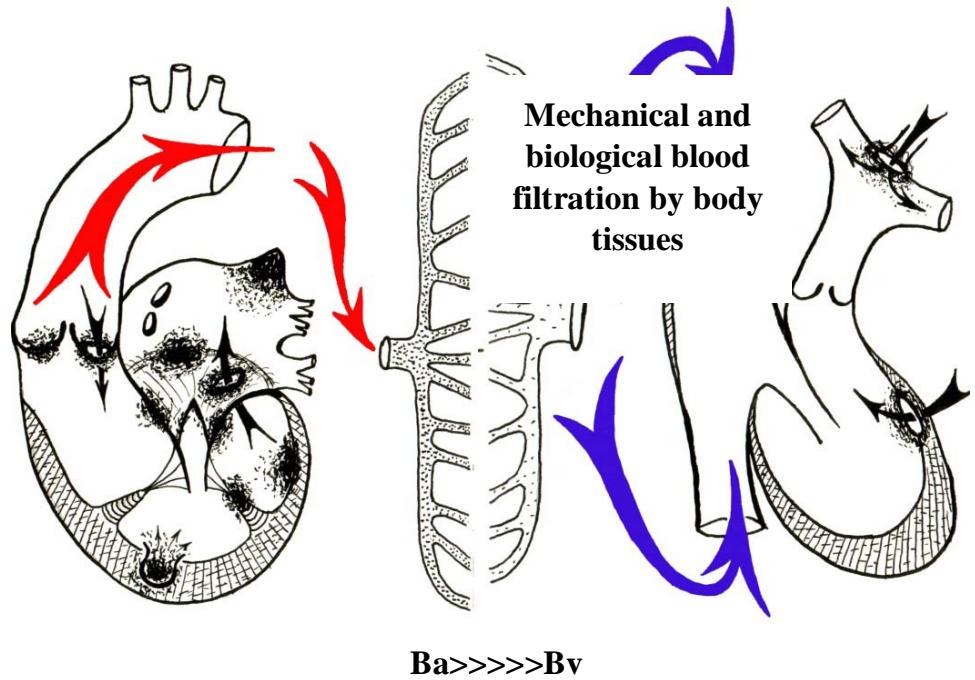 Figure 1: Schematics of the blood purification mechanism that elucidates the difference between arterial (Ba) and venous (Bv) bacteremia
It can be reasonably assumed that the bacterial load for arterial bacteremia is higher than for venous bacteremia. The arterial blood samples are thereby more likely to display higher positive detection rates than venous blood samples (Fig. 1).
## II. METHODS
The patients have given their informed consent for participation in the research study. The study was approved by the institutional research ethics committee and specifying the guidelines for care of animals that have been followed.
A dedicated experimental study was conducted to substantiate the hypothesis that IE is associated with a higher bacterial load in arterial blood than in venous blood (Fig. 2).
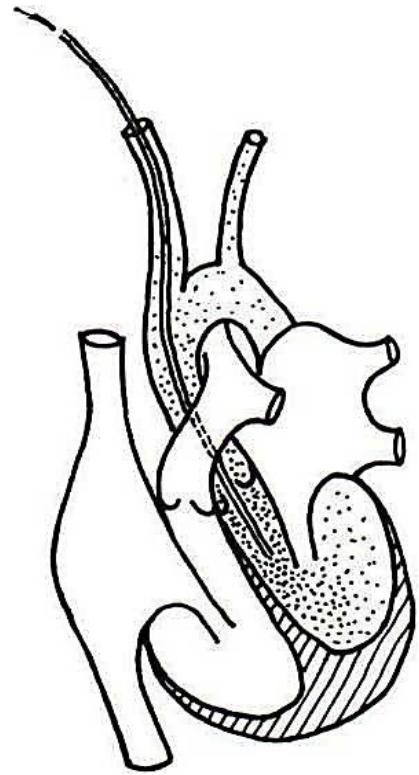 Figure 2: Schematics of experimental bacterial elimination from the LV into the arterial circulation
The qualitative assessment of the differences in bacterial loads between arterial and venous blood was performed in 18 dogs. A bacterial suspension was injected via a catheter into the left ventricle (LV) through the carotid artery in animals under thiopental anesthesia. Blood samples were collected from the femoral artery, and 12-15 seconds later, blood was drawn from the right atrium (RA) via a catheter inserted through the jugular vein. This model simulated transient bacteremia localized to the left heart, which is the most common site of IE. A volume of $50 \mathrm{~mL}$ of broth medium was inoculated with $5 \mathrm{~mL}$ of blood (Fig. 3).
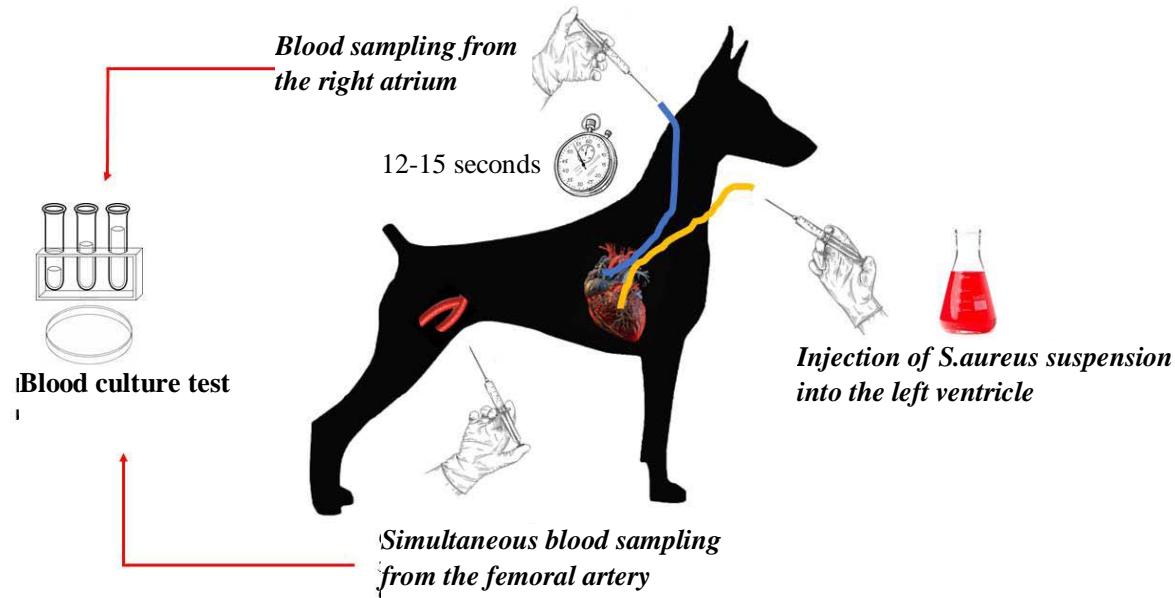 Figure 3: Graphical presentation of the first series of experiments (refer to the test for further clarification)
The second series of experiments was designed to measure the differences in the bacterial load between arterial and venous blood samples using the radionuclide method. The bacterial suspension of S. aureus isolates cultured in agar plates containing $^{131}$
labeled albumin was injected into the LVs of experimental animals. The arterial and venous blood samples were analyzed to determine the relative concentration of $^{131}\mathrm{I}$ (counts per minute in $1\mathrm{mL}$ of blood) in the well counter chamber (Fig. 4).
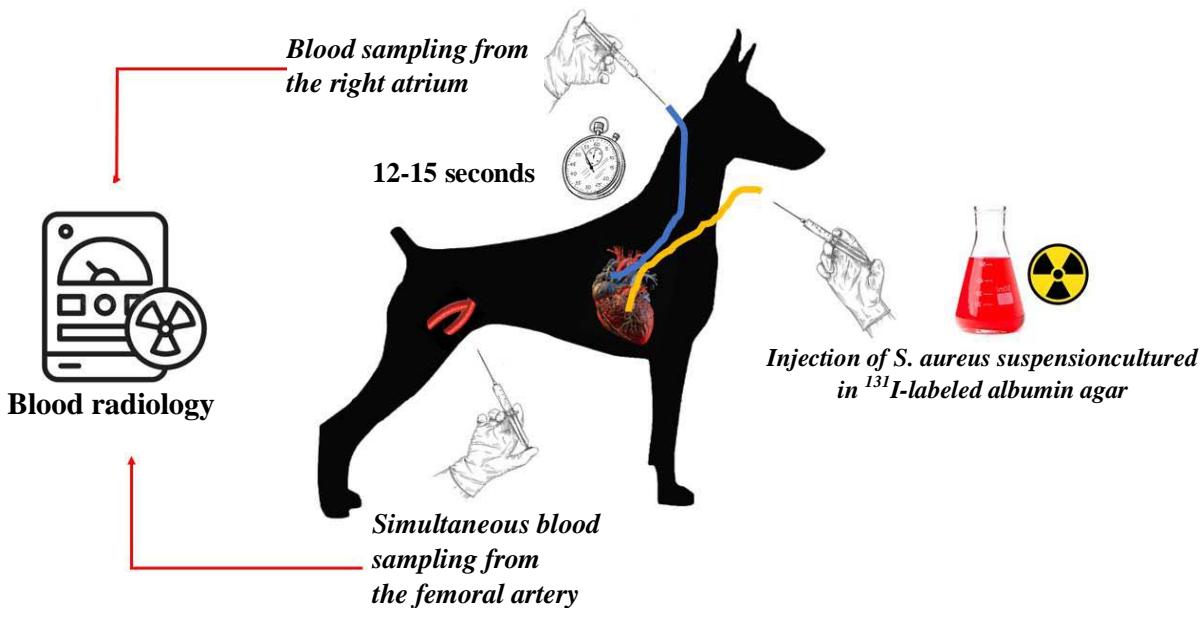 Figure 4: Graphical presentation of the second series of experiments (refer to the test for further clarification)
A comparable series of experiments was mechanical component in the overall blood filtration by conducted to ascertain the contribution of the tissues following the administration of ganglionic blockers (pentamine $5\%$ 2.0). This resulted in peripheral capillary dilation, which was assumed to increase the penetration of bacterial conglomerates into the venous vessels, thereby reducing the difference in the bacterial load between the arterial and venous blood (Fig. 5).
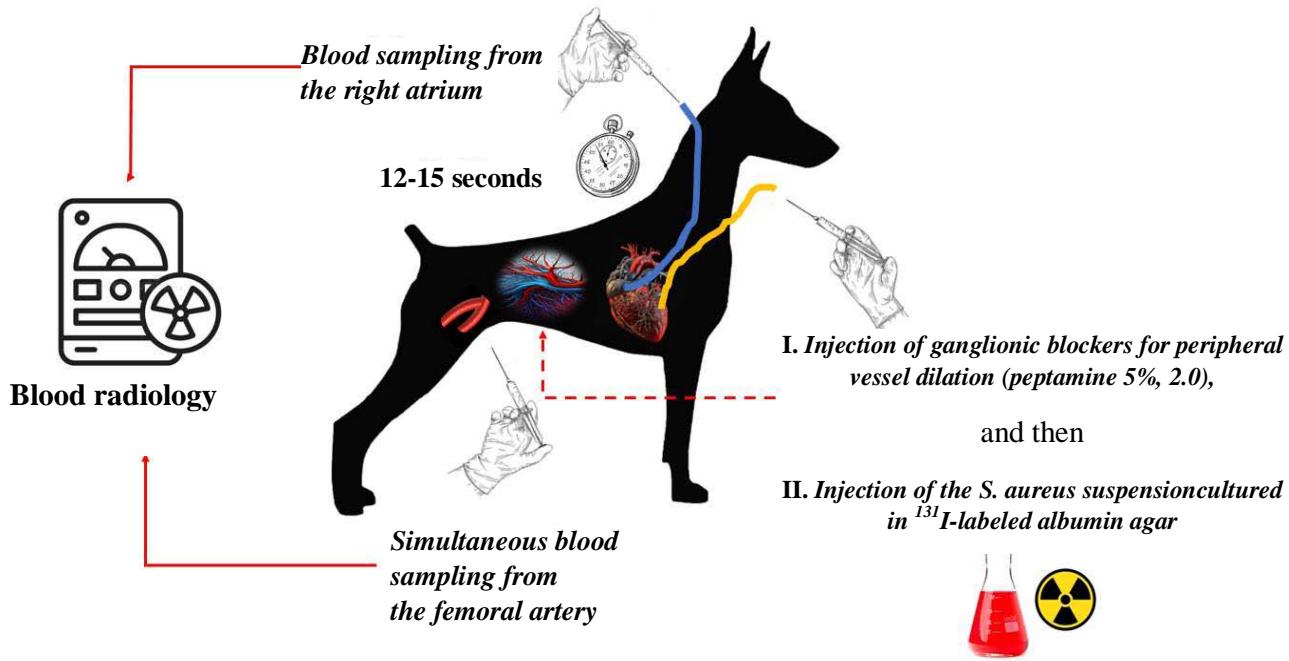 Figure 5: Graphical presentation of the third series of experiments (refer to the test for further clarification)
The fourth series of experiments attempted to evaluate the filtration capacity of the pulmonary circulation, as well as the difference in the blood load for arterial and venous bacteremia localized in the right heart. For this purpose, an isotopic bacterial suspension was injected into the peripheral vein of the animal's forelimb. Four or five seconds later, the first blood sample was drawn directly from the right ventricle through a catheter inserted through the jugular vein to measure an initial concentration. After 6-8 seconds of blood sampling from the right ventricle, the blood sample was obtained from the femoral artery. Following an interval of 12-15 seconds, venous blood samples were collected from the right ventricle (Fig. 6).
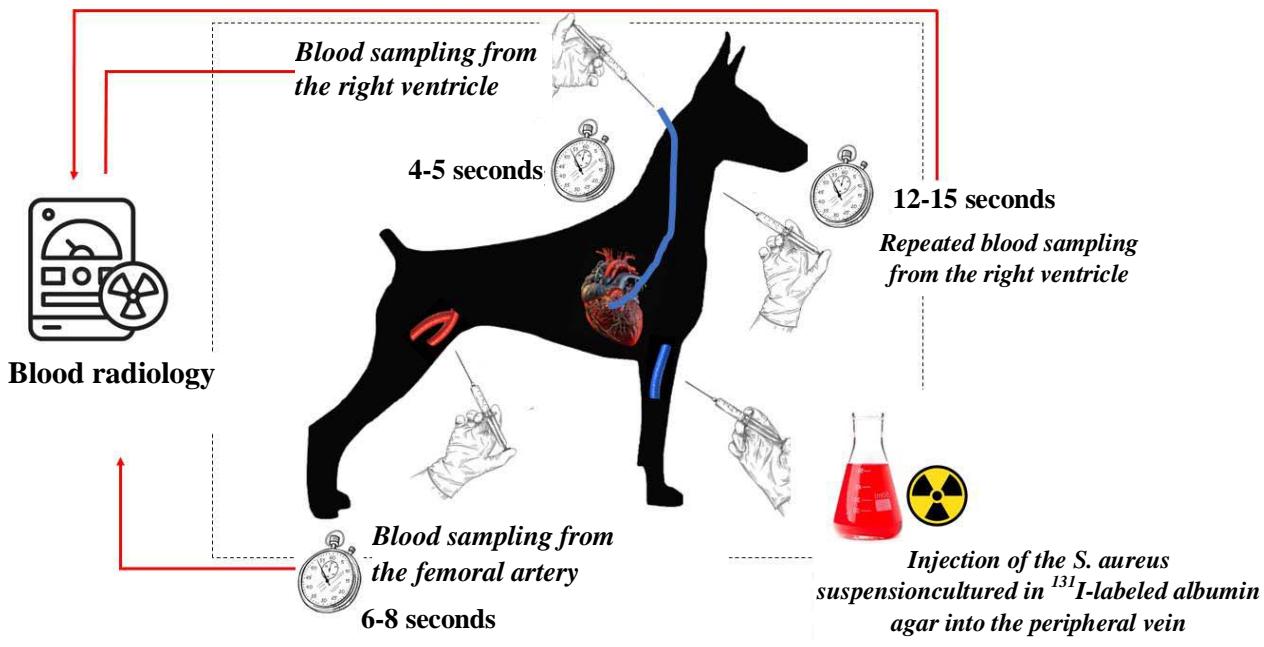 Figure 6: Graphical presentation of the fourth series of experiments (refer to the test for further clarification)

To further substantiate the proposed hypothesis, the authors conducted examinations on 141 patients presenting with signs of intracardiac infection.
Arterial and venous blood samples were obtained from all surgical patients for blood culture testing. Blood samples were collected in accordance with standard blood culture collection practices, with an amount of $10\mathrm{mL}$ of blood per $100\mathrm{mL}$ of the suitable medium. Venous blood was collected via puncture of the ulnar vein or through a catheter inserted into the subclavian or superior vena cava. Arterial blood was drawn from the femoral artery via puncture, in accordance with the established standard procedure.
Prior to blood collection, patients were instructed to perform physical exercises of moderate intensity to enhance the mechanical effect on the contaminated heart valves, thereby facilitating a more substantial elimination of bacteria into the bloodstream.
An increase in the functional load on the heart was achieved by changing from a lying to a sitting position five to seven times. It is noteworthy, however, that the physical exertion was not suitable for hyperthermic or tachycardic patients or those whose condition was considered severe.
## III. RESEARCH EVIDENCE
All 18 arterial blood samples and only eight venous blood samples (44.4%) were positive for microbial contamination in first experimental series. The results of second series of experiments are presented in Table I.
The calculated load difference in the arterial blood was 3.81:1. The bacterial load in the arterial blood is crucial for the development of IE localized in the left heart.
Table I: Radionuclide measurements of simulated arterial and venous bacteremia localized in the left heart
<table><tr><td rowspan="2">Number of experiments</td><td colspan="3">Counts per minute (M + t)</td></tr><tr><td>Reference value</td><td>Arterial blood</td><td>Venous blood</td></tr><tr><td>10</td><td>2650±54.5</td><td>1764.3±43.5P<0.05</td><td>462.5±14.4 0.03P<0.05</td></tr></table>
The study results with ganglionic blockers shown in Table II demonstrate the significantly decreased difference in the bacterial load between the arterial and venous blood.
Table II: Effect of ganglionic blockers on the difference in the bacterial load between the arterial and venous blood
<table><tr><td rowspan="2">Number of experiments</td><td colspan="3">Counts per minute (M + m)</td></tr><tr><td>Reference value</td><td>Arterial blood</td><td>Venous blood</td></tr><tr><td>10</td><td>2724±49.6</td><td>1638.9±34.5P<0.05</td><td>789.4±19.9P<0.05</td></tr></table>
The results of this series of experiments are presented in Table III.
The evaluation of the filtration capacity of the pulmonary circulation and the difference in the bacterial load for arterial and venous bacteremia localized in the right heart demonstrated a value of 2.62:1, which is responsible for the development of IE.
Table III: Measurements of the blood load for arterial and venous bacteremia localized in the right heart
<table><tr><td rowspan="2">Number of experiments</td><td colspan="4">Counts per minute (M + t)</td></tr><tr><td>Reference value</td><td>Blood flow from the right ventricle</td><td>Arterial blood</td><td>Blood flow from the right ventricle</td></tr><tr><td>10</td><td>2840±55.4</td><td>1984.5±21.6</td><td>1015±16.2 P=0.05</td><td>386.5±9.19 P<0.05</td></tr></table>
Experimental studies have demonstrated that the bacterial load in arterial blood is significantly higher than in venous blood for the cardiac localization of infection. The experimental model of severe transient bacteremia localized in the left heart demonstrated that microbial detection rates were higher for arterial blood (100%) compared to venous blood (44.4%). A quantitative assessment of the bacterial load in arterial and venous bacteremia simulated using $^{131}$ I-labeled microorganisms demonstrated that the bacterial load in the arterial blood was approximately 4.5-5 times higher than in the venous blood. Furthermore, the series of experiments conducted with the administration of a ganglionic blocker demonstrated the significance of direct mechanical blood filtration by tissues. The direct effect of the ganglionic blocker on filtration capacity (arteriole dilation and the opening of arterio-venous anastomoses) contributes to the easier penetration of bacterial conglomerates into the venules and veins. Consequently, the difference in the bacterial load between the arterial and venous blood is significantly decreased. The findings of the experimental study suggest that while the pulmonary circulation displays a considerable degree of filtration capacity, there is a higher bacterial load associated with arterial bacteremia than with venous bacteremia, even in the presence of a septic focus in the right heart.
Bacteriological Diagnosis of IE in Clinical Practice: The preoperative blood culture tests for each patient consisted of three to seven arterial and venous blood samples. A total of eight to ten blood samples were collected during the postoperative period. Bacterial cultures were identified in the blood, surgical specimens, such as excised valves, papillary muscles, vegetations (infected thrombotic masses), and tissue specimens (subcutaneous fat, muscles), i.e., so-called filtration material, from the surgical wound.
A total of 120 patients had between three and six positive arterial blood cultures prior to surgery. The microbial strains present in the surgical specimens of these patients were identified. A total of 45 patients exhibited single positive results in their venous blood cultures.
The pathogens were identified in 141 patients. The pathogens were detected in the arterial blood of 120 individuals, including 45 patients who had positive venous blood cultures. In 21 individuals with persistent negative arterial and venous blood cultures, the pathogens were isolated from surgical specimens. The patients were admitted to the hospital between three and five months after the onset of the disease. Prior to admission, they had received aggressive antibacterial therapy. Their recovery process was generally uncomplicated, with no symptoms of sepsis. However, they presented with significant valve changes, circulatory failure, and various complications in internal organs. Histopathology of heart valves in all patients demonstrated bacterial invasion in deep layers of detritus or calcium conglomerate covered with massive organized blood clots or a solid fibrin layer. It was unlikely that microorganisms could pass into the blood flow from this site. Fifteen patients had achieved remission prior to the surgery, so the growth of microorganisms was not detected either in the blood or in the heart valves.
Five patients who presented with a typical clinical picture of IE and respective surgical findings also had sterile blood and heart tissue cultures. However, the histological examination of the affected tissues revealed the presence of a considerable round-cell infiltration of histiocytes in the damaged and scarred valves. This indicates the occurrence of a long-term, chronic inflammatory process, which was most likely of an infectious origin.
The subsequent years of experience in the treatment of intracardiac infections have definitively demonstrated the prevalence of arterial bacteremia over venous blood load. The results of blood and surgical specimen culture tests for patients exhibiting varying degrees of clinical symptoms are presented in Table IV. Consequently, the blood culture values reported for IE patients, as compared to intracardiac microbiological findings indicate a high diagnostic significance of arterial blood cultures.
Table IV: Blood and surgical specimen culture tests in IE patients with various clinical symptoms
<table><tr><td rowspan="2">Clinical symptom severity</td><td colspan="2">Pre-operative positive blood cultures</td><td rowspan="2">Bacteria detection in heart specimen (surgical specimen)</td></tr><tr><td>Venous blood</td><td>Arterial blood</td></tr><tr><td>IE with full-scale clinical picture</td><td>40%</td><td>94.5%</td><td>95%</td></tr><tr><td>IE with no significant clinical symptoms</td><td>15%</td><td>75%</td><td>98%</td></tr><tr><td>Asymptomatic IE</td><td>—</td><td>—</td><td>25%</td></tr></table>
## IV. DISCUSSION
The identification of pathogens in the context of targeted antimicrobial therapy is of great significance, as it directly influences the treatment plan, particularly for cardiac surgical patients with purulent septic infections.[2,15,16] The IE epidemiology has proven to be highly variable over the past decades. On the one hand, the spectrum of identified IE pathogens is continuously expanding, and their ratio in different cohorts of patients is determined by both patient characteristics (age, underlying and concomitant diseases) $^{17}$ and medical care facility experience (cardiac surgery, arrhythmia and electrophysiology, nephrology). On the other hand, the false negative rates of blood culture tests tend to increase with the prevalence of fastidious, aggressive multidrug-resistant pathogens. $^{1,6,8,12}$
The diagnostic significance of bacterial blood cultures varies significantly, ranging from 20 to $65\%$ or more. $^{1,8,18,19}$ This may be attributed to the early and aggressive antibacterial therapy, pathogen character- ristics (including rare hard-to-cultivate microorganisms, fungi, and intracellular bacteria), and limited options for microbiology testing methods. $^{13}$
In practice, a positive blood culture may be obtained from only $45 - 50\%$ of hospitalized patients with acute IE and from $15 - 25\%$ with subacute or indolent IE. This is contingent upon the microbiological laboratory having the requisite experience and facilities.[1,6,13,14,20] Furthermore, the pathogen is most likely to be isolated - from the patient's blood at the clinically advanced stage of the disease, when the infectious process has generalized and gross morphological changes appear in internal organs (heart disease, diffuse glomerulonephritis, embolism, septic meningitis, etc.).
The IE diagnosis is still based on a comprehensive assessment of the patient, which includes an analysis of the medical history and clinical data. The Duke criteria may be of substantial utility in standardizing approaches to the detection of intracardiac infection and integrating the entire process of IE verification into a single program. However, these criteria are hardly utilized in practice. Their use is primarily limited to IE specialists or in the retrospective setting, where there is sufficient information available to make an accurate diagnosis. This also causes a low frequency of using bacteriological testing in patients with suspected IE. It is also an important reason for increasing the diagnostic significance of bacteriological diagnosis.[2,3,18]
At present, the range of microbial studies includes imaging (bacterioscopy) and culture-based methods and +/− mass spectrometry, molecular biological (MALDI-TOF MS, PCR, FISH), and serum immunochemistry (antinuclear antibodies, anti-phospholipid antibodies, anti-porcine protein antibodies, anti-xenogeneic antibody, etc.). In the event that the surgical procedure involves the site of an intracardiac infection, it is essential to consider histological findings using standard (Gram) and specific staining techniques.[2,18,21]
Microbiological (culture-based) blood testing offers a number of significant advantages. These include high sensitivity (a detection rate of $10^{2}\mathrm{mc/mL}$ or more), high specificity, the ability to determine antibiotic sensitivity and antibiotic resistance, and relatively low cost. Among the limitations are the significant influence of the media and inoculation conditions, the lengthy duration of the procedure (up to 120 hours or more), the stringent requirements for biological sample collection, and the high false negative rates.[18]
The use of blood PCR has been demonstrated to be highly effective in patients with negative blood cultures, particularly in the early stages of the etiological diagnosis of IE. Kotova et al. demonstrated that the use of PCR diagnostic testing resulted in a significantly higher probability of lifetime pathogen identification in the blood, $76.6\%$.[13,18]
Matrix-assisted laser desorption ionization time-of-flight mass spectrometry (MALDI-TOF MS) is being incorporated into current diagnostic approaches, enabling the identification of unique protein spectra associated with the pathogen. The substance of this method is the accurate identification of microorganisms based on the analysis of their protein spectra. MALDI-TOF MS offers a number of advantages, including high analytical specificity and sensitivity, the ability to identify more than 2,700 microbial species from the most up-to-date reference database, and low operating costs. The method's inherent limitations diminish its diagnostic value. The following limitations have been identified, i.e., potential spectral interference, inability to differentiate similar or closely related organisms (e.g., Escherichia coli and Shigella spp., various fungi), and significant limitations related to the identification of polymicrobial flora (no more than two microorganisms) and the determination of antibiotic sensitivity and antibiotic resistance. Obtaining positive blood cultures (either primary or those grown on solid media) represents a crucial initial step in the diagnostic process.[7,18]
Although novel microbial detection techniques have been implemented, blood culture testing remains the primary method for pathogen identification. The results of experimental and clinical studies have demonstrated a higher detection rate in arterial blood samples compared to venous blood samples. The obtained clinical data suggest that the diagnostic value of the arterial blood culture testing is significantly higher than that of similar venous blood cultures. The difference in the bacterial load between the arterial and venous blood is likely to be associated with the biological and mechanical blood filtration through body tissues. Moreover, the quality of microorganisms that have passed through a tissue filter and entered the venules and veins of the systemic circulation may change to a certain extent. The probability of their proper growth in artificial media thereby decreases.[2,6,14]
It should be noted that more significant arterial bacteremia can also occur in suppurative lung diseases, since the runoff blood collects in the left heart. The oxygenized, contaminated blood flows from the left heart into the arteries of the systemic circulation. Nevertheless, the clinical and instrumental modalities for the recognition of suppurative lung diseases are numerous, even in the absence of blood culture testing. Nevertheless, the difference (or no difference) in the bacterial load between the arterial and venous blood (or no difference) both in IE and suppurative lung diseases may be considered a criterion of the qualitative assessment of a macroorganism, including general biological reserves, body defenses, immunity, etc. In this context, the concurrent culture-based testing of venous and arterial blood is becoming increasingly important.
## V. CONCLUSION
It can be demonstrated that the phenomenon of the prevalence of arterial bacteremia over venous bacteremia with an infection focus localized in the heart is based on the microcirculatory tissue filtration of bacteria. This has a very important theoretical and practical significance. This phenomenon specifies the infection localization, significantly improves the pathogen detection rate and identification of its pathogenic properties, and enhances sensitivity to antibiotics. This may result in the development of the most efficient individual program of conservative and surgical strategy and significantly improve the treatment outcomes for this life-threatening disease.
Declaration
Local Ethics Committee of the Pirogov National Medical and Surgical Center of the Russian Ministry of Health; Stukolova Tl; No.2; February 15, 2022.
Conflicts of Interest
The authors certify that there is no conflict of interest with any financial organization regarding the material discussed in the manuscript.
Funding
The authors report no involvement in the research by the sponsor that could have influenced the outcome of this work.
Y Shevchenko (2020). Surgical treatment of infective endocarditis and basics of purulent septic cardiac surgery.
G Habib,P Erba,B Iung,Donal Cosyns,B Laroche,C (2019). Clinical presentation, aetiology and outcome of infective endocarditis. Results of the ESC-EORP EURO-ENDO (European infective endocarditis) registry: a prospective cohort study.
Paul Scully,Simon Woldman,Bernard Prendergast (2021). Infective endocarditis: we could (and should) do better.
F Loeffler (1884). Untersuchungen uber die Bedeutung der Mikroorganismen fur die Entstehung der Diptherie beim Menschen, bei der Taube und beim Kalbe.
Y Shevchenko (2020). Surgery of infective endocarditis (40 years of clinical experience).
H Abdelgawad,S Azab,Abdel Hay,M Almaghraby,A (2022). Clinical features and outcomes of infective endocarditis: a single-centre experience.
Alekseevaiv Danilovai,Asnertv (2013). Real-practice therapy of infective endocarditis in Russia: preliminary results of MAESTRO study.
Amany El-Kholy,Nevine Gamal El-Din El-Rachidi,Mervat El-Enany,Eiman Abdulrahman,Reem Mohamed,Hussien Rizk (2015). Impact of serology and molecular methods on improving the microbiologic diagnosis of infective endocarditis in Egypt.
P Fournier,F Gouriet,J Casalta,H Lepidi,H Chaudet,F Thuny (2017). Blood culture-negative endocarditis: Improving the diagnostic yield using new diagnostic tools.
L Li,N Mendis,H Trigui,J Oliver,S Faucher (2014). The importance of the viable but non-culturable state in human bacterial pathogens.
Salama Sadaka,Iman El-Ghazzawy,Mahmoud Hassanen,Ahmed Abu El Kasem,Amina Nour El Din,Marwa Meheissen (2013). Molecular and serological techniques for the diagnosis of culture negative infective endocarditis in Alexandria Main University Hospital.
J Kobalava,E Kotova (2023). Global and national trends in the evolution of infective endocarditis.
Y Shevchenko (1995). Surgical treatment of infective endocarditis.
Aleksandr Baranov,Andrei Mayanskii,Igor’ Chebotar’,Nikolai Mayanskii (2015). A new epoch in medical microbiology.
Mika Halavaara,Timi Martelius,Asko Järvinen,Jenni Antikainen,Pentti Kuusela,Ulla-Stina Salminen,Veli-Jukka Anttila (2019). Impact of pre-operative antimicrobial treatment on microbiological findings from endocardial specimens in infective endocarditis.
X Yang,H Chen,D Zhang,L Shen,G An,S Zhao (2022). Global magnitude and temporal trend of infective endocarditis, 1990-2019: results from the Global Burden of Disease Study.
Kotovaeo,A Pisaryuk,E Domonova,Karaulova Yul,Sil'veistrova Oyu,Shipulinaoyu (2016). Infective endocarditis: etiology and role of current methods in microbiological diagnosis.
P Bosshard,A Kronenberg,R Zbinden,C Ruef,E Böttger,M Altwegg (2003). Etiologic Diagnosis of Infective Endocarditis by Broad-Range Polymerase Chain Reaction: A 3-Year Experience.
A Danilov,T Osipenkova,R Kozlov (2018). Current aspects of infective endocarditis in Russia.
N Schizas,T Michailidis,I Samiotis,V Patris,K Papakonstantinou,M Argiriou (2020). Delayed diagnosis and treatment of a critically ill patient with infective endocarditis due to a false-positive molecular diagnostic test for SARS-CoV-2.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Yuriy. L. Shevchenko. 2026. \u201cSignificance of the Prevalence of Arterial Bacteremia over Venous Bacteremia in the Etiological Diagnosis of Intracardiac Infection\u201d. Global Journal of Medical Research - F: Diseases GJMR-F Volume 24 (GJMR Volume 24 Issue F2).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.