Annotation- The Aim of the Investigation: Increasing the effectiveness of methods for morphological verification of tumours of the head of the pancreas (HP) and the distal common bile duct (DCBD) while reducing the risks of complications and reducing the time of diagnosis and treatment.
## I. EPIDEMIOLOGY
Malignant tumours of the hepatopancreatoduodenal zone (HPDZ) account for $15\%$ of tumours of the digestive tract. The pancreas accounts for up to $86\%$ of neoplasms, and more than $60\%$ are localized in the head of the pancreas [1]. Pancreatic cancer causes more than 331000 deaths in the world annually and accounts for $40\%$ of the total cancer mortality, ranking $7^{\text{th}}$ in the world, $5^{\text{th}}$ in Europe, $4^{\text{th}}$ in USA [2]. According to preliminary expert estimates, by 2030 pancreatic cancer will become the second most common cause of death from malignant neoplasms [3]. This determines the relevance of the problem of early diagnosis and treatment of this pathology at the present time. Ductal adenocarcinoma accounts for $95\%$ of HP malignancies and is the most common cause of OJ of tumour origin [4,5]. In second place is cholangiocarcinoma (CC), with an incidence ranging from 1 to 113 per 100000 population. The increase in the incidence of CC in Russia over the past ten years has increased and amounted to $26,64\%$ [6,7].
OJ in malignant lesions of the HPDZ organs occurs in 36,6 - 47,0% of patients, in HP and DCBD cancer it reaches 95% [8,9,10]. A blurred clinical picture and lack of clinical follow-up lead to the neglect of the pathology. Accordingly, resectability for pancreatic head cancer is no more than 15-20%. [11].
Purulent cholangitis (PH) is one of the most common and severe complications of benign and malignant diseases of the biliary tract caused by a violation of their patency. The main etiological factor of cholangitis are cholestasis, infection and the phenomenon of damage to the common bile duct mucosa [12]. It is believed, that without surgical resolution, acute purulent cholangitis leads to death in $100\%$ cases. Postoperative mortality, according to various authors, varies widely and ranges from 13 to $60\%$ [13].
A wide range of studies are used in the diagnosis of tumour diseases of the HPDZ organs. Ultrasound examination is routine and quite informative [14]. However, a more detailed examination of the biliary tract allows contrast-enhanced MRI of the abdominal cavity with MRCP, including in the version of the hydropressive MRSP [15]. Another highly informative research method is contrast-enhanced multislice computer tomography (MSCT). It was found that an average in $70 - 85\%$ of patients, the resectability of the tumourdetected using CT imaging was confirmed intraoperatively and is considered the gold standard for diagnosing cancer of the HP [16].
Endoscopic methods for examining the upper gastrointestinal tract are mandatory for diseases of the HPDZ organs [17]. The role of endoscopic retrograde cholangiopancreatography (ERCPG) is mainly therapeutic and is used to resolve jaundice [18]. ERCPG with transpapillary excisional biopsy showed a low detection rate of pancreatic head cancer (33-71%). According to some reports, the use of a method with accelerated cytological examination made (ROSE) it possible to increase sensitivity to 76-97% [19].
Confocal laser endomicroscopy (CLEM) is characterized by its effectiveness in treating tumour strictures at the initial stage of the process. But with its high efficiency $(93,3\%)$, high sensitivity $-91,7\%$, specificity $-93,8\%$ and overall accuracy $-92,8\%$, when combined with choledochoscopy and biopsy, demonstrated labour intensity and high operator-dependence method [20]. Puncture fine-needle CLEM has shown high diagnostic value in the diagnosis of cystic diseases of the pancreas [21].
The use of SpyGlass increased sensitivity and specificity to 71 and $100\%$, respectively. However, the technique is very expensive, which makes it impossible to use in most public sector medical institutions [22].
The probability of developing pancreatitis and peritoneal dissemination of tumour cells limits the use of percutaneous pancreatic biopsy. Fine-needle aspiration biopsy (FNAB) under EndoUS control has the lowest complication rate of $1 - 2\%$. The method is especially valuable for tumours less than $2\mathrm{~sm}$. Moreover, the sensitivity and specificity of the method, according to the meta-analysis, are $85$ and $98\%$, respectively. When atypical or suspicious cancer cytology was included, sensitivity increased to $91\%$, but specificity decreased to $94\%$ [23]. However, this method also has high operator dependence, complex and lengthy specialist training, and high cost of equipment.
Despite the possibilities of modern high-tech diagnostic and treatment methods, postoperative mortality after one-stage operations against the background of OJ remains relatively high and it amounts to $15 - 40\%$ for tumour jaundice [17]. Complications after percutaneous endobiliary drainage for OJ in 2,4-32,7% of patients, mortality – from 0,4 to $13,8\%$ [24].
Endoscopic transpapillary interventions can resolve OJ in cancer of organs of HPDZ in more than $80\%$ of cases. They make it possible to prepare patients with OJ for surgical interventions, including radical ones, or become the final methods of palliative treatment of HPDZ tumours complicated by OJ in inoperable patients [25]. In young patients with unresectable HPDZ tumours and OJ syndrome, with a life expectancy of more than six months, the choice of palliative internal bile diversion for distal biliary block is hepaticoenterostomy. In other clinical situations, using one of the methods of stenting the bile ducts is justified [26].
A combination of antegrade and transpapillary methods of decompression of the biliary tract, objective phasing of surgical treatment while observing the principle of an individual approach increases the effectiveness of treatment and reduces the percentage of complications and unfavorable outcomes.
The unsatisfactory results of chemotherapy for pancreatic cancer are due to the chemoresistance of this type of tumor for two reasons: the presence of very dense, poorly vascularized, fibrous tumor envelope and high expression of R-glycoprotein. This first makes it difficult to deliver the chemotherapy to the tumour, and then accelerates its elimination [3].
Regional methods of chemotherapy have shown their effectiveness. A combination of cycles of chemoembolization (CE) and chemoinfusion (CI) was the most effective method of intra-arterial therapy in patients with unresectable pancreatic cancer: the median was 15, 6 months compared with 11,7 and 10,8 months for CE and CI [27].
The probability of using targeted therapy in patients with pancreatic cancer based on molecular testing was $26\%$. Using it in this group of patients increased the median survival from the moment of disease progression to 31 months, compared to 18 months with standard chemotherapy [28].
Therefore, the current state of the problem of early diagnosis of pancreatic cancer, the lack of a simple method of histological verification with a low cost, dictates the search for new organizational solutions and scientific developments in this direction. Progress in drug therapy for pancreatic cancer makes the problem even more urgent.
## II. MATERIALS AND METHODS
An analysis of the treatment results of 441 patients with obstructive jaundice of tumour origin over 19 years was carried out.
Study Periods:
- 2000 - 2007the X-ray surgery department of the Republican Clinical Hospital, Vladikavkaz;
- 2014 - 2021 based on the surgical department of the Clinical Hospital of the NorthOssetian State Medical Academy;
- 2022-2024 surgical department of the State Budgetary Healthcare Institution "Republican Clinical Hospital of Emergency Medical Care" Vladikavkaz;
- 2023-2024 surgical department of the Suburban Central District Hospital in the v. Oktyabrskoye.
Innovative methods are approved for use in clinical practice after approval by the Ethical Committee at the North Ossetian State Medical Academy in 2018. All patients signed informed consent for the proposed therapeutic and diagnostic measures.
Of the treated patients, 223 (50,6%) were men, 218 (49,4%) were women. The age of patients was from 20 to 91 years. Average age - 69,5±0,97years, men - 69,4±1,3, women - 69,5±1,4 years (Fig. 1)
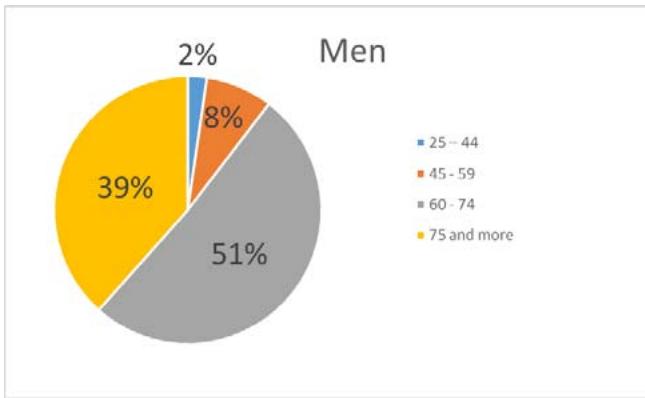
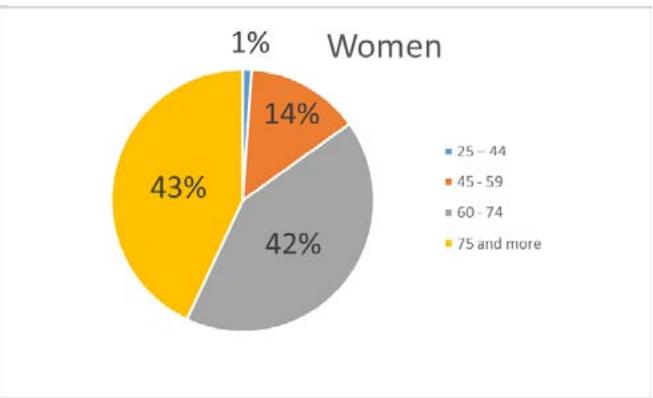 Fig. 2: The structure of biliopancreatoduodenal cancer Fig. 1: The analysis of the treatment results of 441 patients with tumour genesis for 19 years
In the morbidity structure, the most common types were cancer of the head of the pancreas and Klatskintumour (Fig.2).
<table><tr><td>Localization</td><td>Number of patients (%)</td></tr><tr><td>The Head of the Pancreas</td><td>236 (53,3%)</td></tr><tr><td>Cancer of the GDP (great duodenal papilla) and DCBD (distal common bile duct)</td><td>58 (13,1%)</td></tr><tr><td>Klatskintumour</td><td>86 (19,5%)</td></tr><tr><td>Liver cancer</td><td>8 (1,9%)</td></tr><tr><td>Gallbladder</td><td>29 (6,6%)</td></tr><tr><td>Metastases at the gates of the liver</td><td>24 (5,6%)</td></tr></table>
The duration of the pre-hospital period from the moment the complaints appeared was on average $24,3 \pm 4,7$ days.
The patients underwent clinical, labouratory and instrumental research methods: electrocardiogram, chest radiography, ultrasound of the abdominal organs, esophagogastroduodenoscopy, MRI of the abdominal cavity with MR cholangiography, MSCT of the abdominal cavity with/without contrast enhancement, ERCP, percutaneous transhepatic cholangiography, fistulocholangiography.
Distribution of patients according to the level of bilirubinemia: mild (22 - 100 $\mu$ mol/l) - 48 (10,9%), moderate (100 - 200 $\mu$ mol/l) - 114 (25,8%), severe (200 or more $\mu$ mol/l) - 279 (63,3%) patients.
Severe forms of acute cholangitis according to TG18 were detected in 169 (38,3%) patients: Grade II - 138 (32,4%), Grade III - 31 (7,3%).
Concomitant pathology was detected in 339 $(76.9\%)$ patients. The main share was made up of patients with cardiovascular diseases. Cardiac ischemia was diagnosed in 274 $(62.1\%)$ patients, and 93 $(21.1\%)$ patients suffered from type 2 diabetes mellitus.
We conditionally divided the patient into two groups: primary endoscopic (group I) and primary percutaneous (group II) interventions (Fig. 2).
The comparison of the groups was carried out for the period 2014-2019 when the possibility of both types of interventions became available. During the analysis, the average values M+mwere determined, where M – is the average value and m – is the standard error of the mean. The groups were representative according to Students t-test (p>0.05) in terms of hepatorenal, hematosis function and the degree of cholangiectasia according to radiological research methods. Moreover, in the group with primary endoscopic interventions, patients with severe cardiovascular pathology prevailed. (Fig.3)
<table><tr><td>NN</td><td>Criterion</td><td>Group I (primary endoscopic interventions)</td><td>Group II (primary percutaneous interventions)</td></tr><tr><td>1</td><td>Total bilirubin</td><td>304,3 ± 39,1</td><td>264,8 ± 21,4</td></tr><tr><td>2</td><td>Aspartate aminotransferase</td><td>145,1 ± 15,6</td><td>135,7 ± 15,3</td></tr><tr><td>3</td><td>Alanine aminotransferase</td><td>212,0 ± 25,7</td><td>154,2 ± 19,8</td></tr><tr><td>4</td><td>Alkaline phosphatase</td><td>1103,4 ± 278,1</td><td>1563,9 ± 395,2</td></tr><tr><td>5</td><td>Prothrombin index (%)</td><td>84,5 ± 2,3</td><td>82,8 ± 1,8</td></tr><tr><td>6</td><td>Blood urea (mmol/l)</td><td>5,7 ± 0,4</td><td>6,45 ± 0,6</td></tr><tr><td>7</td><td>Creatinine (μmol/l)</td><td>90,6 ± 6,5</td><td>89,0 ± 4,6</td></tr><tr><td>8</td><td>Leukocyte (x109)</td><td>8,0 ± 0.5</td><td>9,0 ± 0.5</td></tr><tr><td>9</td><td>Thrombocyte (x1000)</td><td>288,2 ± 15,8</td><td>253,7 ± 14,5</td></tr><tr><td>10</td><td>Hemoglobin (x1012)</td><td>124,1 ± 2,7</td><td>118,5 ± 2,9</td></tr><tr><td>11</td><td>Hepaticocholedochus diameter (mm)</td><td>12,8 ± 0,7</td><td>14,0 ± 0,6</td></tr><tr><td rowspan="5">12</td><td>Block level</td><td></td><td></td></tr><tr><td>High</td><td>9 (21,4%)</td><td>19 (36,5%)</td></tr><tr><td>Average</td><td>0</td><td>7 (13,5%)</td></tr><tr><td>Short</td><td>32 (76,2%)</td><td>25 (48,1%)</td></tr><tr><td>Double</td><td>1 (2,4%)</td><td>1 (1,9%)</td></tr><tr><td>13</td><td>Erosive and ulcerative lesions of the stomach and duodenum</td><td>6 (14,3%)</td><td>5 (9,6%)</td></tr><tr><td>14</td><td>Essential hypertension 2-3 stages</td><td>31 (73,8%)</td><td>33 (63,5%)</td></tr><tr><td>15</td><td>2A-B degree circulatory failure</td><td>18 (42,9%)</td><td>15 (28,8%)</td></tr><tr><td>16</td><td>Diabetes mellitus 2 type</td><td>6 (14,3%)</td><td>15 (28,8%)</td></tr></table>
Fig.3: Comparative characteristics of patients with primary endoscopic and percutaneous interventions [29]
During surgical treatment, preference was given to low-traumatic decompression surgical interventions. At a low and moderate level of block of extrahepatic bile ducts of tumour origin, in technically possible, we began with transpapillary interventions. When using antegrade decompression methods in isolation from 2000 to 2007, we adhered to the tactics of staged interventions (external, then external-internal drainage with an interval of 7-8 days). Subsequently, we tried to perform one-stage external-internal drainage with endoscopic papillosphincterotomy (EPST). The tactic of phasing is preserved only in patients with purulent cholangitis. Symptomatic therapy was prescribed to patients with Klatskintumour, Bismuth 4. Conservative treatment included detoxication, hepatoprotective therapy, correction of water and electrolyte disorders, and treatment of concomitant diseases.
For the purpose of cytological verification, antegrade and retrograde methods of brush-biopsy of the area of tumor strictures of the bile ducts and fine-needle aspiration-cutting biopsy of tumor formations were used. To obtain histological material, endoscopic excisional biopsy, percutaneous biopsy using semi automatic needles or an automatic biopsy system were used.
## III. THE RESULTS
439 (99,5%) patients received surgical treatment and 2 (0,5%) patients could not be prepared for surgery due to the severity of the condition. In total,
719 operations were performed, which amount to 1,64 operations per patient (Fig. 4). The choice of method depended on the availability of technical capabilities at the current time. In incurable patients, we tried to solve the OJ problem endoscopically.
<table><tr><td>Type of operation</td><td>Quantity</td></tr><tr><td colspan="2">Endoscopic</td></tr><tr><td>Atypical (incisional) papillotomy</td><td>5 (7,7%)</td></tr><tr><td>EPST</td><td>9 (13,8%)</td></tr><tr><td>Bilioduodenal stenting</td><td>43 (66,1%)</td></tr><tr><td>Nasobiliary drainage</td><td>1 (1,5%)</td></tr><tr><td>"Rendezvous" technique</td><td>12 (18,5%)</td></tr><tr><td colspan="2">Percutaneous</td></tr><tr><td>Primary percutaneous transhepatic external drainage of the bile ducts (PPTEBD)</td><td>303 (83,6%)</td></tr><tr><td>Separate external drainage</td><td>39 (12,9%)</td></tr><tr><td>Of which: Drainage of one lobe of the liver</td><td>15 (38,5%)</td></tr><tr><td>Separate sub-sectional drainage</td><td>21 (53,8%)</td></tr><tr><td>Separate segmental drainage</td><td>3 (7,7%)</td></tr><tr><td>Percutaneous transhepatic external-internal drainage of the bile ducts (PTEIDBD)</td><td>266 (70,0%)</td></tr><tr><td>Of them: Primary PTEIDBD</td><td>57 (21,4%)</td></tr><tr><td>The second stage after PPTEBD</td><td>205 (77,1%)</td></tr><tr><td>Failed PTEIDBD</td><td>4 (1,5%)</td></tr><tr><td>PTEIDBD was simultaneously supplemented with EPST</td><td>14 (5,3%)</td></tr></table>
Fig. 4: Decompressive surgical interventions for OJ of tumour genesis
Primary interventions on the Major Duodenal Papilla (MDP) were performed in 65 $(14,8\%)$ patients. In 2 $(3,1\%)$ of them it was unsuccessful, in one due to a previous gastric resection for Billroth - 2 peptic ulcer, in the other due to tumour deformation of the duodenum due to a tumour of the head of the pancreas. In 5 $(7,7\%)$ patients an atypical (incisional) papillotomy was performed, in 9 $(13,8\%)$ they were limited to endoscopic papillosphincterotomy (EPST). Both types of operations were used for cancer of the MDP (which was diagnosed as a result of histological examination). This turned out to be enough to relieve OJ and prepare patients for the second stage of the operation. In 43 $(66,1\%)$ patients, bilioduodenal stenting was required for adequate decompression of the bile ducts. In all cases, a plastic stent with a diameter of 8 to 12 Fr, more often 10 Fr, was used. In 1 $(1,5\%)$ patient, EPST was supplemented with nasobiliary drainage. The "Rendezvous" technique was used in 12 $(18,5\%)$ patients. The need arose due to the impossibility of cannulation of the MDP in patients with the presence of transhepatic drainage.
With a high block of the bile ducts, the presence of cholangitis, as well as with a medium and low block in the case of impossibility of endoscopic access or if there were indications for radical surgical interventions at the second stage, percutaneous decompression interventions were used. The presence of transhepatic drainage facilitated the formation and ensured decompression of biliodigestive anastomoses.
In total, 374 (85,2%) patients underwent percutaneous puncture interventions. It was unsuccessful in 4 (0,9%) cases in patients with Klatskintumour, Bismuth IV.
Primary percutaneous transhepatic external drainage of the bile ducts (PTEDBD) was performed in 303 $(83,6\%)$ patients. Of these, 264 $(87,1\%)$ with medium and low blocks achieved immediate complete decompression of the biliary tract. In 39 $(12,9\%)$ – with a high block involving the lobar bile ducts in the process of confluence, separate external drainage was used. Of these, 15 $(38,5\%)$ patients underwent drainage of one of the liver lobes. This was considered sufficient to reduce total blood bilirubin below $50\mu \mathrm{mol} / \mathrm{l}$. If higher levels of bilirubinemia persisted, the second lobe of the liver was drainage. Separate sublobar drainage was performed in 21 $(53,8\%)$ patients, separate segmental drainage was performed in 3 $(7,7\%)$ patients.
Percutaneous transhepatic external-internal drainage of the bile ducts was performed in 266 (70,0%) patients, of which primary PTEIDBD in 57 (21,4%), the second stage after PTEDBD - in 205 (77,1%). In 4 (1,5%), it was impossible to pass the stricture area. In 14 (5,3%) PTEIDBD added with EPST. Analysis of literature data and experience in treating patients after transpapillary methods of bilioduodenal drainage allowed us to switch to primary PTEIDBD. In order to prevent postoperative pancreatitis, especially in patients with absent or mild pancreatic hypertension, external-internal drainage was supplemented with EPST. This
tactic made it possible to reduce water and electrolyte losses and shorten the length of hospital treatment.
<table><tr><td>Complication</td><td>Treatment</td></tr><tr><td colspan="2">Endoscopic</td></tr><tr><td>Dislocation of a plastic stent – 14 (21,5%)</td><td>Stent replacement – 12.
Removal of the stent (cancer of the MDP) – 1.
Conservative – 1.</td></tr><tr><td>Bleeding from a papillotomy wound – 6 (9,2%)</td><td>Electrocoagulation – 1.
Conservative – 5.</td></tr><tr><td colspan="2">Percutaneous</td></tr><tr><td>Migration of bile drainage – 37 (9,9%)</td><td>Repeated drainage – 36.
Laparoscopic drainage of the abdominal cavity with repeated PTEBD – 1.</td></tr><tr><td>Hemobilia – 5 (1,3%)</td><td>Correction of the position of bile drainage – 5.</td></tr><tr><td>Bile leakage into abdominal cavity – 6 (1,6%)</td><td>Puncture drainage of the abdominal cavity with correction of the position of bile drainage – 2.
Translation PTEIDBD to external – 1.
Laparotomy, sanitation and drainage of the abdominal cavity – 3.</td></tr><tr><td>Liver biloma – 1 (0,3%)</td><td>Puncture drainage – 1.</td></tr><tr><td>Compression of the left lobar duct by self-expanding stents – 1 (0,3%)</td><td>Puncture drainage of the left lobar duct – 1.</td></tr><tr><td>Pancreatitis – 2 (0,6%)</td><td>Conservative – 1.</td></tr></table>
Fig. 5: Complications after decompressive surgical interventions for bile ducts
Complications after endoscopic interventions (Fig.5) on MDP developed in 20 $(30,7\%)$ of 65 patients, in 3 $(4,6\%)$ two complications were noted. In 14 $(21,5\%)$ cases, there was stent dislocation due to an anatomical feature (angulation of the common bile duct) and incorrect choice of stent light. In 12 $(18,5\%)$ cases repeated stenting was required; in 1 $(1,5\%)$, the stent had to be removed due to the unstable position of the stent (angulation of the hepaticocholedochus, short distal stenosis); in 1 $(1,5\%)$ patient with an MDP tumour, at the time of complete dislocation into the intestine, the clinical manifestations of cholangitis were relieved and did not require additional interventions. Bleeding from the papillotomy site was observed in 6 $(9,2\%)$ patients; in 4 $(6,1\%)$, it was of moderate severity and was stopped conservatively; in 2 $(3,1\%)$ - severe bleeding was stopped by electrocoagulation of the bleeding vessel in 1 $(1,5\%)$ patient and conservatively - in 1 $(1,5\%)$. In 10 of 43 patients with bilioduodenal stenting, it was necessary to change the plastic stent due to its obstruction. The cause of obstruction was increased lithogenicity of bile and chronic cholangitis. The stent replacement period was varied from 5 to 90 days, averaging of $52,7 \pm 9,63$ in two patients, two stents were installed during replacement.
The most common complication of percutaneous interventions was drainage dislocation in 37 (9,9%) patients, it was observed before the use of "Pigtail" drains with a thread fixation of the internal ring and in case of Klatskintumour, Bismuth 4. In all cases, repeated drainage was required, in 1 (0,3%) case, which was laparoscopic drainage of the abdominal cavity. Hemobilia developed in 5 (1,3%) cases, which was relieved by correcting the position of the bile drainage. All cases were at the stage of gaining experience in X-ray surgical interventions and were associated with an intrahepatic vessel falling into the trajectory of the bile duct puncture. In the future we tried to avoid such moments. Bile leakage into the abdominal cavity was observed in 6 (1,6%) patients, which required drainage of the abdominal cavity with correction of the drainage position in 2 cases, replacement of external-internal drainage with an external one in 1 patient, and laparotomy with sanitation and drainage of the abdominal cavity in 3 patients. In all cases, the cause was loss of adequate drainage due to drainage dislocation or obstruction. A liver biloma formed in 1 (0,3%) patient, which was drainage by puncture. In 1 (0,3%) case of a Klatskintumour after antegrade stenting of the bile ducts with self-expanding stent, compression of the left lobar duct occurred, which require additional its drainage. 2 (0,6%) patients developed mild pancreatitis, which was treated conservatively.
Out of 439 operated patients, 33 $(7.5\%)$ died, of which 23 $(5.7\%)$ died during the formation of X-ray surgical service. In 4 $(0.9\%)$ cases the cause of death was biliary peritonitis, all of them with a Klatskintumour, Bismuth IV. Progression of hepatocellular failure against the background of adequate decompression of the bile ducts was in 20 $(4.5\%)$ cases, acute cerebrovascular accident in 1 $(0.2\%)$ patient, progression of the underlying disease in 4 $(0.9\%)$ patients, pulmonary embolism (PE) in 2 $(0,4\%)$ patients, bleeding from a growing tumour of the head of the pancreas in the duodenum – in 2 $(0,4\%)$ cases.
For the purpose of morphological verification oftumours of the head of the pancreas, the following were used: puncture trephine biopsy of a liver tumour - 6, transgastric trephine biopsy of a tumour of the head of the pancreas - 3, brush biopsy of the distal common bile duct for cancer of the distal common bile duct and head of the pancreas - 7 (3 - antegrade, 4 - retrograde), antegrade brush biopsy for Klatskintumour - 2, pinch biopsy for locally advanced cancer of the head of the pancreas and distal common bile duct and for cancer of the MDP in 19 patients. A brush biopsy of the distal common bile duct for cancer of the head of the pancreas and the DCBD gave three cheerful, two doubtful and two false-negative cytological conclusions. Transgastric trephine biopsy of a tumour of the head of the pancreas gave one false-negative result, in 1 case it was complicated by pancreatorhea.
## IV. THE DISCUSSION
The lack of technical ability to biopsy tumours of the head of the pancreas and the distal common bile duct under EndoUS, performing CLEM and other expensive methods, the complexity and negative results of using transgastric trephine biopsy, the impossibility of obtaining histological material from brush biopsy led us to search for a safer, more effective and cost-effective an advantageous method for obtaining histological material from tumors of the HP and the DCBD.
We have developed and introduced into practical medicine method for trepan biopsy of tumours of the head of the pancreas and the distal common bile duct and device (Fig. 6)for their implementation by E.B.Revazov-Ts.S.Khutiev(patent NoNo 2722655, 2747591, 203409, 2768480). The essence of the invention is simultaneous percutaneous transhepatic external-internal drainage of the bile ducts with transcholangiostomy trephine biopsy of tumours of the head of the pancreas and the distal common bile duct and EPST (endoscopic papilla sphincterotomy) in patients with obstructive jaundice. We modified the antegrade method of core biopsy method[30] and used the principle of transjugular liver biopsy (percutaneous remote puncture biopsy with a flexible needle along a non-rectilinear trajectory with access through a tubular structure). The original deviceand method made it possible to provide trephine biopsy only under X-ray control. X-ray landmarks were the contours of the biopsy needle and metal guide (Fig.13).
The direction and depth of the puncture were chosen based on the preoperative analysis of CT and/or MRI, where the size of the tumour, its relationship to nearly structures, and the extent of occlusion were determined. The direction of the puncture was established by rotating the device around the axis of the guide and controlled by the polyposition of fluoroscopy.
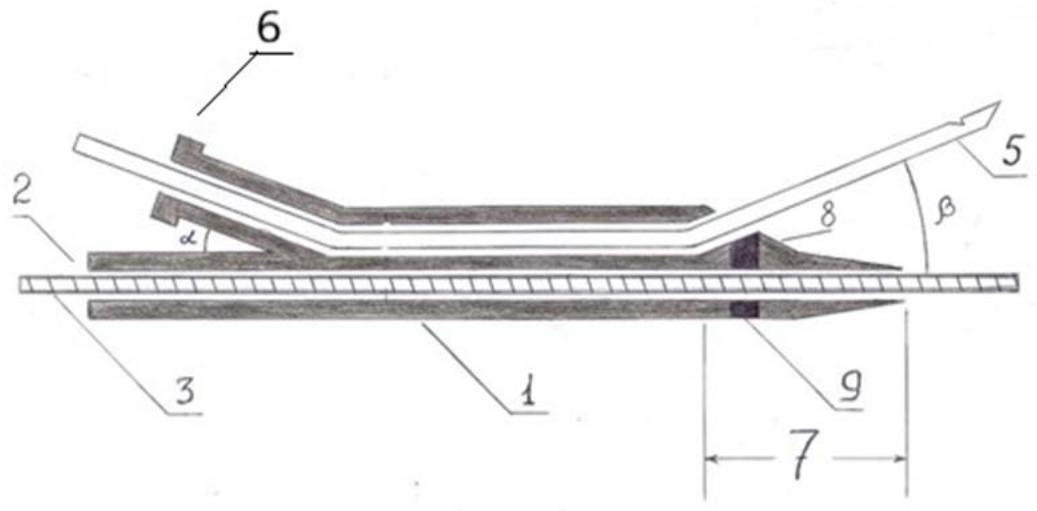
 Fig. 7: MRI-characteristics of a tumour of the head of the pancreas Fig. 6: Device for trephine biopsy of tumours of the head of the pancreas and the distal common bile ductbyE.B.Revazov-Ts.S.Khutiev
1 - device for trephine biopsy (frame); 2 - conductive channel; 3 - metal super-rigid guide type Lunderquist; 4 - internal opening of the puncture channel; 5 - biopsy needle; 6 - connector; 7 - distal end; 8 - projection; 9 - radio-opaque mark; a - angle to ensure operation of the device; b - angle of axial deviation of the biopsy needle.
To determine the technical aspects of performing trephine biopsy, we performed a continuous retrospective analysis from a database of 100 contrast-enhanced MRIs of the abdominal cavity in patients with a tumour of the head of the pancreas and biliary hypertension (Fig. 7). Studied the size and preferential localization of the tumour in relation to the hepaticocholedochus (HC), its relationship to the main pancreatic duct (MPD) and duodenum (DU). The age of the patients was 35-90 years, with average of $65 \pm 1,13$. Men - 54 (54%), women - 46 (46%).
<table><tr><td>Sign</td><td>Minimum</td><td>Maximum</td><td>Average</td></tr><tr><td>Tumour size (mm)</td><td>11</td><td>89</td><td>35.4±1,53</td></tr><tr><td>Diameter of hepaticocholedochus (HC)(mm)</td><td>7.5</td><td>35</td><td>15,6±0,51</td></tr><tr><td>The length of the tumour narrowing of the HC(mm)</td><td>4</td><td>50</td><td>27±1,1</td></tr><tr><td>Hepaticocholedochus angle (degrees)</td><td>80</td><td>162</td><td>115,3±1,69</td></tr><tr><td>Diameter of the main pancreatic duct (MPD)(mm)</td><td>2</td><td>18</td><td>7,3±0,37</td></tr><tr><td>Distance from the level of the HC block to the level of the MPD block (mm)</td><td>1</td><td>63</td><td>11,9±1,02</td></tr><tr><td>Length from the proximal border of the HC block to the duodenum (mm)</td><td>14</td><td>52</td><td>30,8±0,91</td></tr></table>
Tumour size varied from 11 to $89\mathrm{mm}$, averaging $35,4 \pm 1,53\mathrm{mm}$. According to the TNM system in stages $T_{1} - 4$, $T_{2} - 60$, $T_{3} - 32$, $T_{4} - 4$ patients. Signs of germination in the duodenum and MDP were noted in 18 (18%) patients; limited to invasion of parapancreatic tissue in 7 (7%) patients. Enlarged regional lymph nodes in 14 (14%) patients. Distant metastases were detected in 28 (28%) patients. Of them: $T_{1} - 2$, $T_{2} - 10$, $T_{3} - 15$, $T_{4} - 1$.
The diameter of the hepaticocholedochus (HC) ranged from 7,5 to $35\mathrm{mm}$, on average $-15,6\pm 0,51\mathrm{mm}$; the length of the tumour narrowing of the HC is from 4 to $50\mathrm{mm}$, the average is $27\pm 1,1\mathrm{mm}$. In 19 (19%) cases, an intact distal part of the common bile duct was identified extending from 5 to $36\mathrm{mm}$. The length from the proximal border of the HC block to the duodenum along the HC axis is $14 - 52\mathrm{mm}$, on average $-30,8\pm 0,91\mathrm{mm}$.
The HC angle in the distal third was $80 - 160^{\circ}$ with an average of $115,3\pm 1,69^{\circ}$. The magnitude of the angle depended on the predominant localization of the tumour, the degree of biliary hypertension and the associated elongation of the HC and its tortuosity. Currently, there is no clear understanding of the dependence of technical actions on a simple statement of the value of the angle HC. The opinion of an individual approach to this parameter and its further study remains.
The diameter of the main pancreatic duct (MPD) is from 2 to $18\mathrm{mm}$, on average $-7,3 \pm 0,37\mathrm{mm}$. The absence of pancreatic hypertension was noted in 14 $(14\%)$ patients, including 4 $(4\%)$ with a tumour size more than $40\mathrm{mm}$. 27 $(27\%)$ had MPD from 3,5 to $6\mathrm{mm}$ and 57 $(57\%)$ had $6\mathrm{mm}$ or more. In 2 $(2\%)$ MPD was not possible to visualize due to the extensive of the tumour process in the pancreas.
The distance from the HC block level to the MPD block level ranged from 1 to $63 \mathrm{~mm}$, with an average of $11,9 \pm 1,02 \mathrm{~mm}$. In 26 (26%) patients this distance is less than $7 \mathrm{~mm}$ and in 13 (13%) of them the
MPD diameter is 6mm or more. Erosion of the internal contour HC in the area of the block was detected in 35 (35%), of which in 17 (17%) – like a "writing pen", in 9 (9%) – on the lateral side, in 9 (9%) – on the medial side; in 65 (65%) a "transverse block" is determined. There is no unambiguous connection between the predominant localization of the tumour and the size of HC contour usuration. Preferential location of the tumourabout HC: antemedial – 49 (49%), antegrade – 18 (18%), circular – 20 (20%), medial – 6 (6%), antelateral – 5 (5%), latero-antemedial – 2 (2%). In 67 (67%), the tumour thickness anterior to the HC lumen was more than 10 mm (Fig.8, 9, 10).
Analysis of the preferential localization of the tumour in relation to HC was one of the main ones for determining the technical feasibility and safety of performing transcholangiostomy biopsy. The thickness of the tumour anterior to the lumen of the HC of more than $10\mathrm{mm}$ was examined in order to select the angle of $\beta$ axial deviation of the biopsy needle in the original device for trephine biopsy. It was important that at the time of biopsy the needle penetrated the tumour tissue, but did not have the opportunity to exit into the free abdominal cavity.
<table><tr><td>Localization</td><td>Quantity (percentage)</td></tr><tr><td>Ante-medially</td><td>49 (49%)</td></tr><tr><td>Antegrade</td><td>18 (18%)</td></tr><tr><td>Circulary</td><td>20 (20%)</td></tr><tr><td>Medially</td><td>6 (6%)</td></tr><tr><td>Ante-lateral</td><td>5 (5%)</td></tr><tr><td>Latero-ante-medial</td><td>2 (2%)</td></tr><tr><td>The thickness of the tumour anterior to the lumen of the HC is more than 10mm</td><td>67 (67%)</td></tr></table>
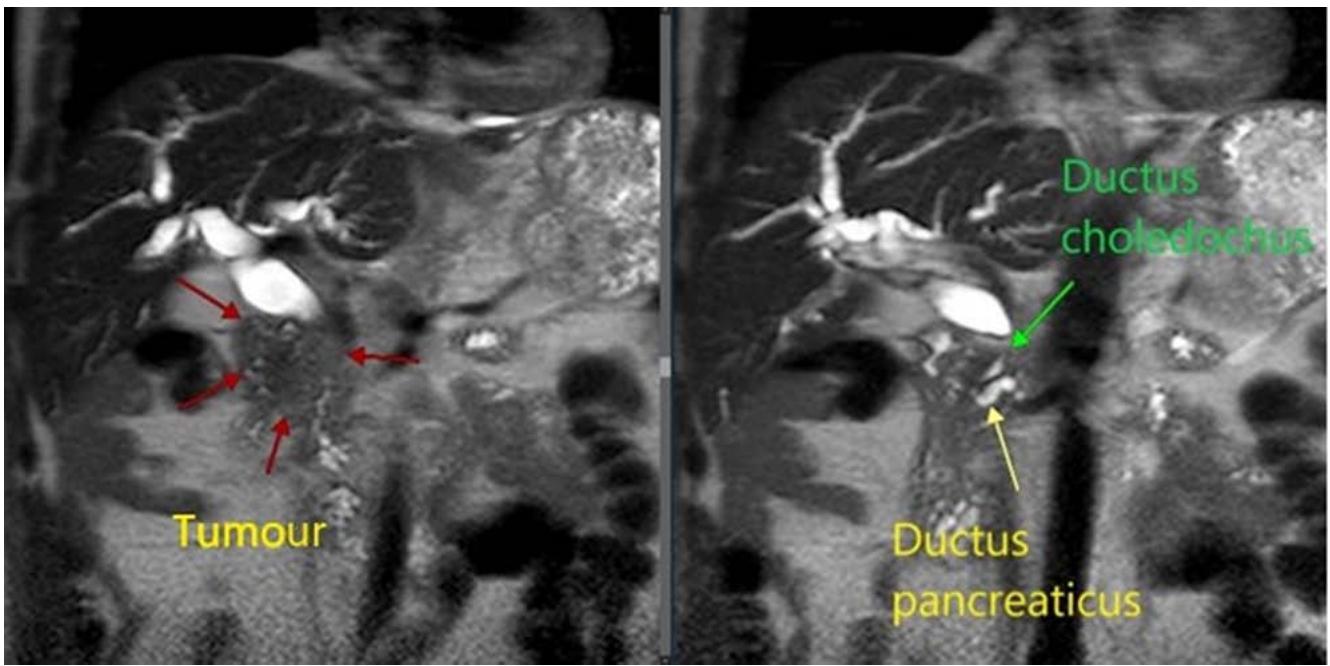 Fig. 8: Preferential localization of the tumour in relation to hepaticocholedochus
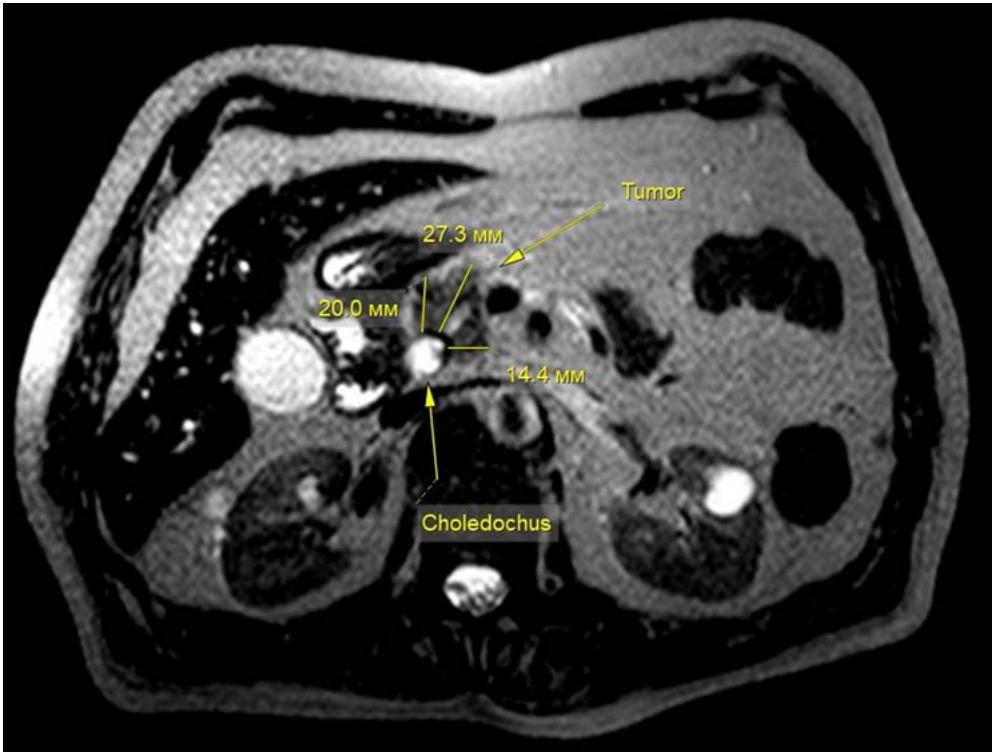 Fig. 10: Ante - medial localization of the tumour
As the result, we have identified a successful coincidence of the most common localization of the tumour of the head of the pancreas with the safest direction of transcholangiostomy biopsy - ante-medial and medial, which is effectively applicable in $77\%$ of cases. Our chosen angle $\beta$ of the axial deviation of the biopsy needle allows us to safely perform a biopsy in another $18\%$ of cases (with antegradetumour growth), which is a total to $95\%$. The average distance from the proximal border of the tumour to the duodenum is up to $30,8 \pm 0,91 \mathrm{~mm}$. Allows the use of an automatic biopsy system in most cases, including with a biopsy depth of $22 \mathrm{~mm}$, avoiding the needle entering the duodenum with the risk of bleeding. If the tumour is small and there is a risk of damage to the duodenum or exit into the abdominal cavity, it is necessary to consider the possibility of aspiration-cutting biopsy in manual mode. Damage to the MPD can be reliable avoided in 87 (87%) of patients in any direction of puncture, because, taking into account the course of the MPD with HC, in 74 (74%)the MPD is located outside the puncture trajectory. In 13 (13%) patients, due to severe pancreatic hypertension, the probability of pancreatitis during MPD puncture is low. Cases of leakage of pancreatic juice through the puncture channel into the common bile duct will not have clinical significance.
The effectiveness of the method is explained by the fact that after passing the distal end of the original device (Fig. 6) along a metal guide through the tumour stenosis along the true lumen of the HC, we achieve tight contact of the internal opening of the puncture channel with the proximal border of the occlusion (Fig.
11). The correct choice of puncture direction necessarily ensures penetration into the tumour tissue with the collection of histological material. The combination of the biopsy needle and the metal guide ensures the safety of the intervention. The device is made of a double-lumen plastic tube, which determines its low cost. Ease of implementation makes the method accessible to most interventional radiologists.
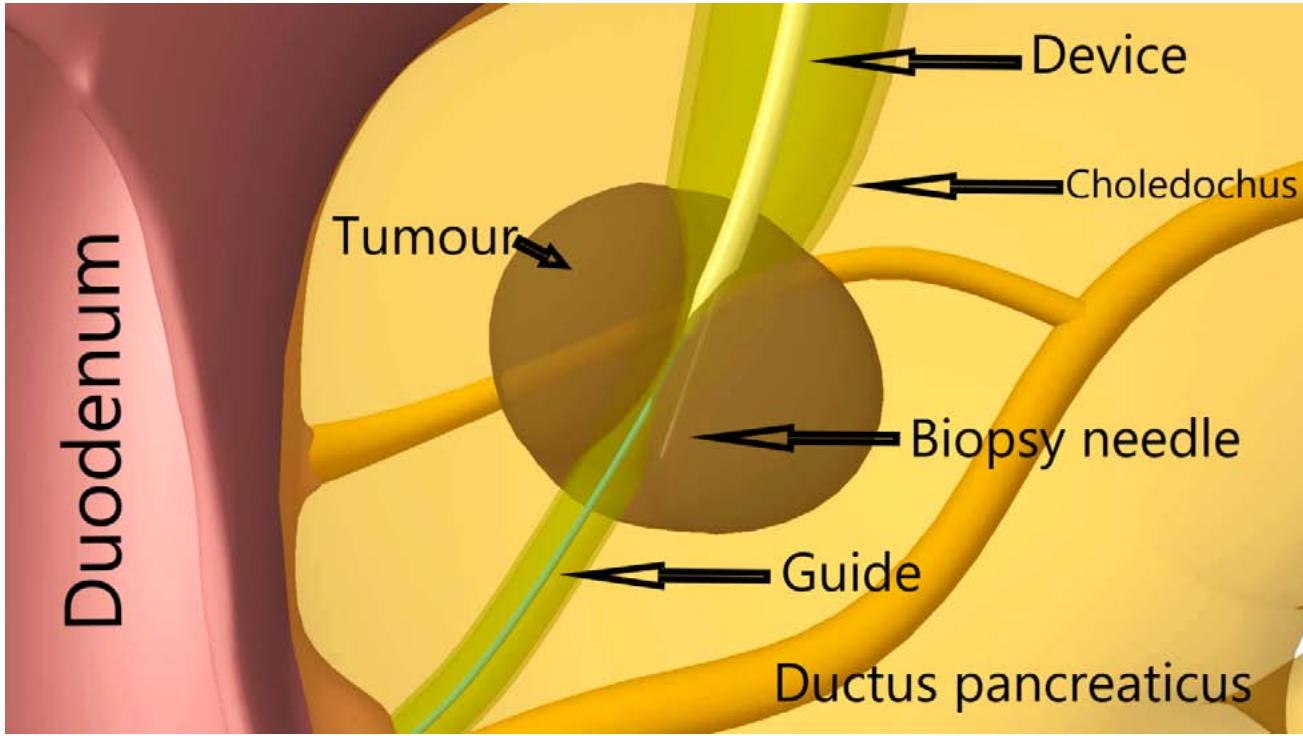 Fig. 11: Transcholangiostomy trephine biopsy of a tumour of the head of the pancreas by E.B.Revazov - Ts.S.Khutiev
As a result, the method we developed was used in 8 patients. Sufficient cytological and histological material was obtained in all cases. To take material, in two instances a Franzen 21G biopsy needle with a vacuum syringe was used, in 6 cases – a Bard Magnum 20G was used on the Bard Magnum automatic biopsy system. Cancer of the head of the pancreas was confirmed in 6 cases. In 4 hystologically and cytologically, in two instances – only cytologically. The absence of tumour tissue in the histological material in these patients is due to a biopsy along the edge of the tumour. Chronic pancreatitis was detected in 2 patients, confirmed in 1 case at the second stage of treatment. No complications were observed after using the trephine biopsy method we developed.
The issue of partial success in two cases, where we received only cytological confirmation of cancer in the absence of it in the histological material, we believe is due to insufficient development of the technique.
Figures 12-15 show the stages of transcholangiostomy trephine biopsy with external-internal drainage of the bile ducts and endoscopic papillsphincterotomy.
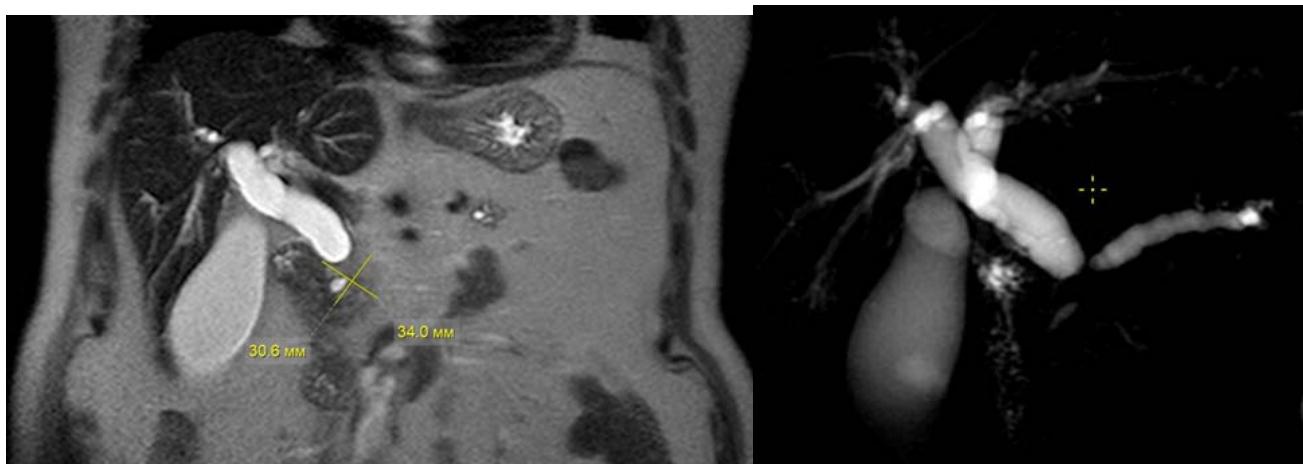 Fig. 12: Preoperative analysis of MRI of the abdominal cavity with MR-cholangiography
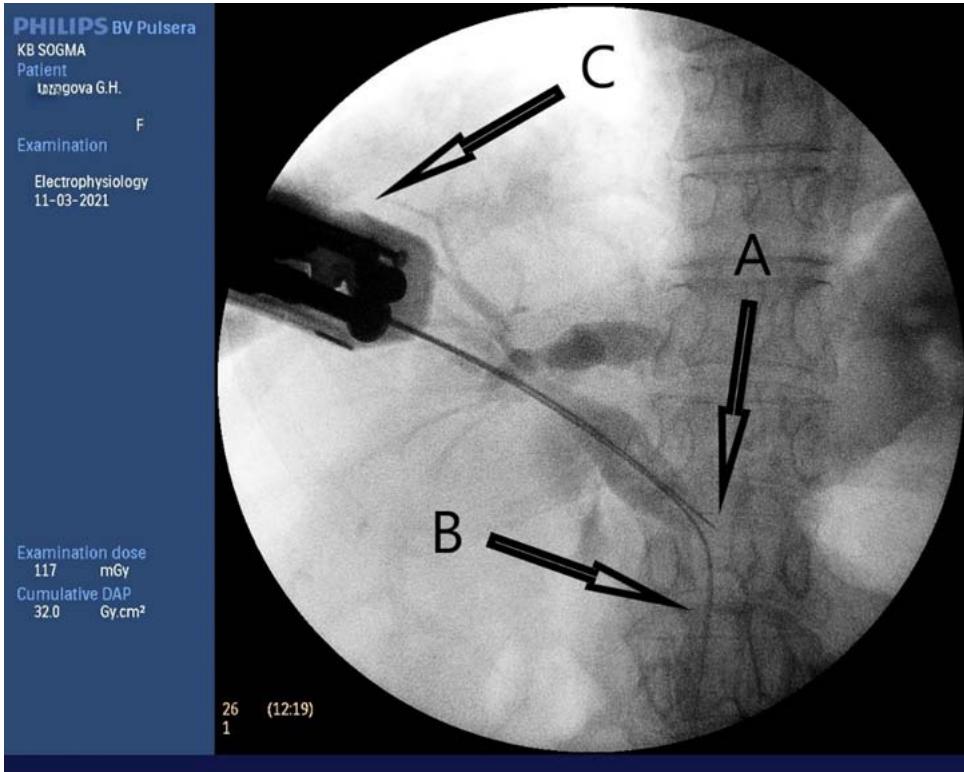 Fig. 13: Transcholangiostomy trephine biopsy of a tumour of the head of the pancreas (A - Biopsy needle; B - Super-rigid guide; C - biopsy gun.)
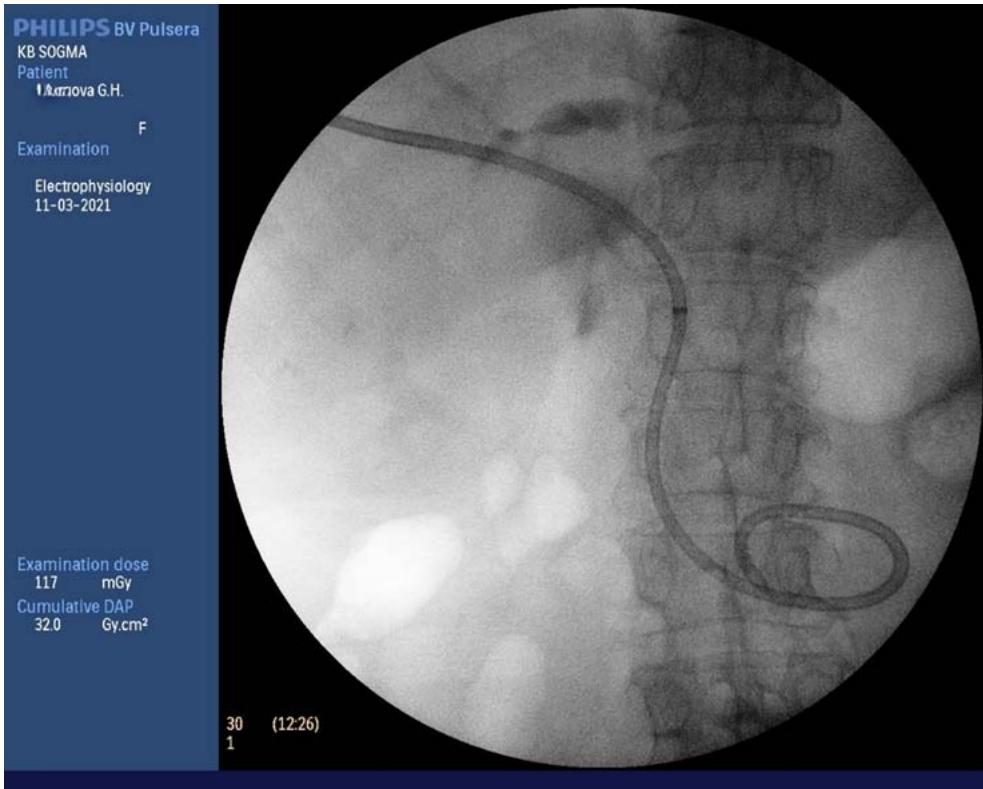 Fig. 14: External-internal drainage of the bile duct
 Fig. 9: Lateral localization of the tumour
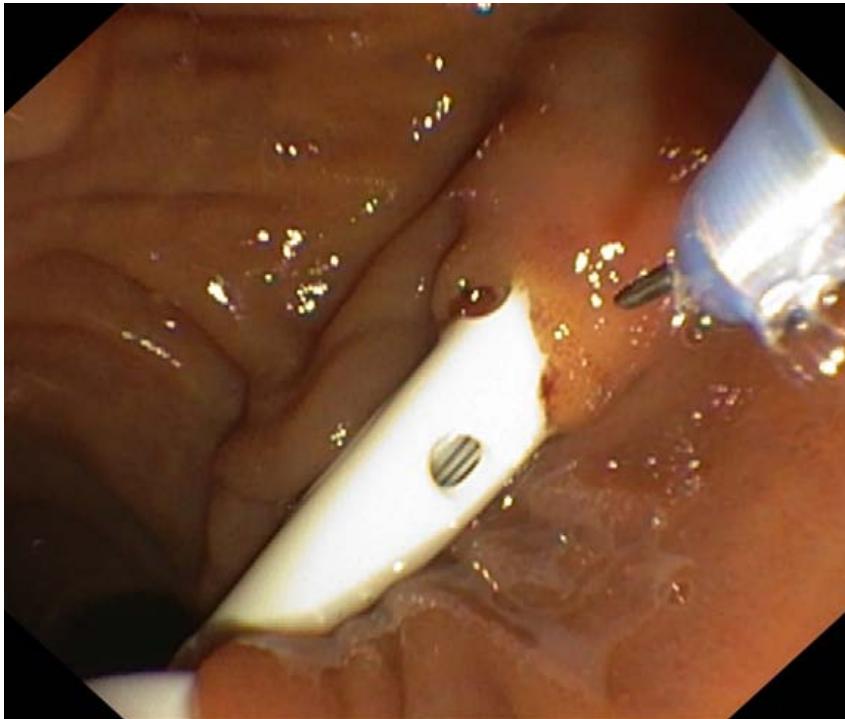 Fig.15: Endoscopic papillosphincterotomy.
## V. CONCLUSION
The method of trephine biopsy of tumours of the head of the pancreas and the distal common bile duct (by E.B. Revazov – Ts.S. Khutiev) made it possible, within the framework of one combined minimally invasive surgical intervention, to perform internal drainage of the biliary tract with obtaining morphological verification in all 8 cases, in 6 of them – histological confirmation. No complications were observed. This distinguishes the method from fine-needle aspiration biopsy under EndoUS with a complication rate of 0,88-2% and obtaining predominantly cytological material. Transgastric trephine biopsy is currently not recommended due to the frequency of complications [31,32].
Preoperative MRI/CT analysis of the abdominal organs with contrast enhancement is necessary for safe and effective transcholangiostomy trephine biopsy of a tumour of the head of the pancreas and the distal common bile duct. These research methods provide sufficient information about the location, size of the tumour and its relationship to nearby anatomical structures. The main aspects are: the preferential localization of the tumour in relation to the HC, the distance from the proximal border of the tumour stricture to the duodenum along the true lumen of the hepaticocholedochus, the thickness of the tumour anterior to the HC.
The possibility of performingbiopsy with an automatic system (we consider it a priority due to the powerful mechanism) or semi-automatic needle is assessed – if the thickness of the tumour tissue along the puncture trajectory exceeds the length of the needle extension. If the thickness is insufficient, it is advisable to perform aspiration-cutting biopsy manually.
The first results of using the proposed method and device for trephine biopsy of tumours of the head of the pancreas and the distal common bile duct are scientifically substantiated and have proven effective. In our opinion, this makes our method a promising, reliable direction in solving the problem of morphological diagnosis of tumours of a given localization, complicated by obstructive jaundice. Further study on a large number of patients will reveal the true picture of the effectiveness and safety of the method. A priority method for preoperative testing will likely be identified. Criteria should be defined according to which preference will be given to aspiration and trephine biopsy.
Conflict of interest The authors declare no conflict of interest.
Compliance with ethical principles The authors confirm that they respect the rights of the people participated in the study, including obtaining informed consent when it is necessary, and the rules of treatment of animals when they are used in the study. Author Guidelines contains the detailed information.
J Ferlay,I Soerjomataram,M Ervik,R Dikshit,S Eser,C Mathers,M Rebelo,D Parkin,D Forman,F Bray,Globocan (2012). V 1.0, Cancer Incidence And Mortality Worldwide: Iarc CancerBase.
D Kaprin (2022). Intra-Arterial Regional Chemotherapy for Pancreatic Cancer (Review).
M Ducreux,A Cuhna,C Caramella,A Hollebecque,P Burtin,D Goéré,T Seufferlein,K Haustermans,J Van Laethem,T Conroy,D Arnold (2015). Cancer of the pancreas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up.
D' Benson Ab 3rd,M Angelica,D Abbott,T Abrams,S Alberts,D Saenz,C Are (2017). Nccn Guidelines Insights: Hepatobiliary Cancers, Version 1.
D Frantsev,O Sergeeva,B Dolgushin (2019). Therapy for hilar cholangiocarcinoma.
A Kovalenkoyu,O Zharikovyu (2017). Portal Cholangiocarcinoma: Epidemiology, Staging Principles and Aspects of Tumour Biology // Hirurgia.
V Ivshin,A Yakunin,Yu,O Lukichev (2000). Percutaneus Diagnostic and Bile-Remuving Operations In Patients With Obstructive Jaundice.
I Patyutkoyu,A Kotelnikov (2007). Surgery Of Bilio-Pancreato-Duodenal Cancer (Manual for Medical Doctors) // M. "Meditsina.
V Kubyshkin,V Vishnevskii (2003). Cancer of Pancreas. // M. Medpraktika.
A Kashintsev,Kokhanenkon,Yu (2013). Relationship Between Diabetes Mellitus And Pancreatic Cancer.
B Datsenko,V Borisenko (2013). Mechanical jaundice, acute cholangitis, biliary sepsis: pathogenic relationship and principles of differential diagnostics.
S Bagnenko,S Shlyapnikov,A Korolkov (2007). Modern approaches to etiology, pathogenesis and treatment of cholangitis andbiliary sepsis.
Z Agaeva (2009). Possibilities of Ultra-Sonic Investigation Methods in Diagnostic of Porto Choliiigiocarcinoma And After Complications // Kubanskiinauchnyimeditsynskiivestnik.
Y Parkhisenko,A Gorokhov (2011). Hydropressive Magnetic Resonans Cholangiopancreatography In the Differential Diagnosis of Benign Bile Duct Narrowing Genesis and Tumour Strictures // Vestniknovykhmeditsinskikhtekhnologii.
Mark Callery,Kenneth Chang,Elliot Fishman,Mark Talamonti,L William Traverso,David Linehan (2009). Pretreatment Assessment of Resectable and Borderline Resectable Pancreatic Cancer: Expert Consensus Statement.
D Krasilnikov,S Salimzyanovsh,A Abdulyanov,M Minnullin,A Zakharova (2011). Diagnostics and Surgical Treatment of Patients with the Syndrome of Obstructive Jaundice. // Kreativnayahirurgiyaionkologiya.
J Kostina,K Pavelets (2018). Diagnosis and Treatment of Pancreatic Cancer the Current State of the Problem.
E Solodinina,N Fomicheva,D Ulyanov (2019). Confocal Laser Endomicroscopy in the Diagnosis of Extrahepatic Bile Duct Diseases.
Yung Ka,Chin,Clement Chun Ho,Damien Wu,Yew (2021). Tan the Role of Needle-Based Confocal Laser Endomicroscopy in the Evaluation of Pancreatic Cystic Lesions: a Systematic Review.
D Savin,M Kuzmin-Krutetskiy,M Safoev,S Khamid,Topilin Dk (2019). Relevant Techniques of the Peroral Transpapillary Cholangioscopy (in Russian only).
I Mikhailov,V Beliakouski,V Kudrashou,A Dyatlov,Ch. Weerakoon (2021). Biopsy of pancreatic tumors prior to resection: for and against.
Sh. Karimov,M Khakimov,A Adylkhodzhaev,S Rakhmanov,V Khasanov (2015). Correction of Complications of the Transhepatic Endobiliary Interventions for Obstructive Jaundice Caused by Periampullar Tumors.
S Budzinsky,S Shapovalyants,S Fedorov,E Mylnikov,A Pankov,A Chernyakevich,P (2013). Opportunities of Endoscopic Retrograde Stenting of the Bile Ducts in Malignant Tumours of the Pancreatobiliary Zone, Complicated by Obstructive Jaundice.
A Beburishvili,E Zyubina,Yu. Vedenin,V Mandrikov,S Panin (2020). Minimally invasive bile internal drainage in tumor obstructive jaundice.
A Kozlov,D Granov,P Tarazov,A Pavlovskiy,L Korytova,A Polikarpov,S Popov,E Rozengauz (2019). Intra-arterial chemotherapy for advanced pancreatic cancer.
Michael Pishvaian,Edik Blais,Jonathan Brody,Emily Lyons,Patricia Dearbeloa,Andrew Hendifar,Sam Mikhail,Vincent Chung,Vaibhav Sahai,Davendra Sohal,Sara Bellakbira,Dzung Thach,Lola Rahib,Subha Madhavan,Lynn Matrisian,Emanuel Petricoin (2020). Overall survival in patients with pancreatic cancer receiving matched therapies following molecular profiling: a retrospective analysis of the Know Your Tumor registry trial.
E Revazov,T Khutiev,M Revazova,A Chetiev,U Beslekoev,T Ardasenov (2023). Improvement of methods of diagnosis and treatment of patients with obstructive jaundice of tumor genesis.
U Kulezneva,M (2016). Percutaneoues Interventions in Abdominal Surgery.
Andrey Mikhetko,Anna Artemieva,Olga Ivko,Oleg Tkachenko,Marina Grinkevich,Aleksandra Sidorova,Iurii Petrik (2021). Endoscopic endosonography with fine-needle aspiration biopsy in the diagnosis of pancreatic tumors.
I Mikhailov,V Beliakouski,V Kudrashou,A Dyatlov,Ch. Weerakoon (2021). Biopsy of pancreatic tumors prior to resection: for and against.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
E.B.Revazov. 2026. \u201cAdvancements in Morphological Verification: Evaluating a Novel Method for Pancreatic and Distal Bile Duct Tumour\u201d. Global Journal of Medical Research - F: Diseases GJMR-F Volume 24 (GJMR Volume 24 Issue F2).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.