The present study investigated cardiac oxidative status in carbon tetrachloride (CCl 4 )exposed rats treated with aqueous and ethanol extracts of Dialium guineense stem bark. Adult male Wistar rats (n = 25) weighing 170 -190 g (mean weight = 180 ± 10 g) were randomly assigned to five groups (5 rats per group): normal control, CCl 4 control, silymarin, aqueous extract and ethanol extract groups. With the exception of normal control, the rats were exposed to CCl 4 at a single oral dose of 1.0 mL/kg body weight, bwt. Rats in the silymarin group were administered silymarin (standard cardioprotective drug) at a dose of 100 mg/kg bwt, while those in the two treatment groups received 1000 mg/kg bwt of aqueous or ethanol extract orally for 28 days. Activities of antioxidant enzymes such as catalase, superoxide dismutase (SOD), glutathione peroxidase (GPx) and glutathione reductase (GR) were evaluated in heart homogenate.
## I. INTRODUCTION
Carbon tetrachloride $\left(\mathrm{CCl}_{4}\right)$ is a colorless liquid with a "sweet" smell that can be detected at low levels [1]. Its production has steeply declined since the 1980s due to environmental concerns and the decreased demand for chlorofluorocarbons (CFCs), such as the Freons dichlorodifluoromethane (F-12) and trichlorofluoro-methane (F-11), which are used primarily as refrigerants [2]. It is also used in petroleum refining, pharmaceutical manufacturing, as an industrial solvent, in the processing of fats, oils, and rubber, and in laboratory applications [3]. Currently, $\mathrm{CCl}_{4}$ is not permitted in products intended for home use. The primary routes of potential human exposure to $\mathrm{CCl}_4$ are inhalation, ingestion, and dermal contact. The general population is most likely to be exposed to $\mathrm{CCl}_4$ through air and drinking water[4 - 6]. In humans and animals, $\mathrm{CCl}_4$ is rapidly absorbed by any route of exposure. Once absorbed, it is widely distributed among tissues, especially those with high lipid content, reaching peak concentrations in $<1-6\mathrm{~h}$, depending on exposure concentration or dose. The compound is metabolized in the body, primarily by the liver, but also in the kidney, lung, and other tissues containing cytochrome P450 (CYP450). The fraction of the compound that is metabolized varies with dose [7, 8].
The heart is a muscular organ which pumps blood through the blood vessels of the circulatory system [9]. Blood provides the animal's body with oxygen and nutrients as well as assist in the removal of metabolic wastes. In humans, the heart is located between the lungs in the middle compartment of the chest [10 - 12]. The heart is effectively a syncytium, a meshwork of cardiac muscle cells interconnected by contiguous cytoplasmic bridges [13 - 15].
Plants are at the center of Traditional Medicine. Their use in disease management is as old as man [16, 17]. Medicinal plants serve as cheap alternative to orthodox medicine since they are readily available [18 - 20]. Dialium guineense is a medicinal plant used in folklore medicine for the treatment of infections such as diarrhea, severe cough, bronchitis, wound, stomachaches, malaria, jaundice, ulcer and hemorrhoids [21, 22]. At present not much is known about the potential of extracts of D. guineense stem bark to protect against $\mathrm{CCl}_4$ -induced cardio-toxicity in rats. The aim of this study was to investigate cardiac oxidative status in $\mathrm{CCl}_4$ -exposed rats treated with aqueous and ethanol extracts of D. guineense stem bark.
## II. MATERIALS AND METHODS
### a) Chemicals
All chemicals and reagents used in this study were of analytical grade and they were products of Sigma-Aldrich Ltd. (USA).
b) Collection of Plant Material
- The stem barks of $D$. guineense were obtained from Auchi, Edo State, Nigeria and authenticated at the herbarium of the Department of Plant Biology and Biotechnology, University of Benin, Benin City, Nigeria (No. UBH<sub>D</sub>330).
c) Plant Preparation and Extraction
The stem bark was washed and shade-dried at room temperature for a period of two weeks and crushed into small pieces using clean mortar and pestle. Aqueous and ethanol extracts of the stem bark were obtained using cold maceration method as described previously [23].
d) Experimental Rats
- Adult male Wistar rats $(n = 25)$ weighing $170 - 190\mathrm{g}$ (mean weight $= 180\pm 10\mathrm{g}$ ) were obtained from the Department of Anatomy, University of Benin, Benin City, Nigeria. The rats were housed in metal cages under standard laboratory conditions: room temperature, $55 - 65\%$ humidity and 12-h light/12-h dark cycle. They were allowed free access to rat feed (pelletized growers mash) and clean drinking water. Prior to commencement of the study, the rats were acclimatized to the laboratory environment for one week. The study protocol was approved by the University of Benin Faculty of Life Sciences Ethical Committee on Animal Use.
e) Experimental Design
- The rats were randomly assigned to five groups (5 rats per group): normal control, $\mathrm{CCl_4}$ control, silymarin, aqueous extract and ethanol extract groups. With the exception of normal control, the rats were exposed to $\mathrm{CCl_4}$ at a single oral dose of $1.0~\mathrm{mL / kg}$ bwt [23]. Rats in the silymarin group were administered silymarin (standard cardioprotective drug) at a dose of $100~\mathrm{mg / kg}$ bwt, while those in the two treatment groups received $1000~\mathrm{mg / kg}$ bwt of aqueous or ethanol extract orally for 28 days.
f) Tissue Sample Collection and Preparation
- At the end of the treatment period, the rats were euthanized and their hearts excised, and used to prepare $20\%$ tissue homogenate. The homogenate was centrifuged at 2000 rpm for 10 min to obtain supernatant which was used for biochemical analysis.
g) Biochemical Analyses
- The activities of catalase, SOD and GPx were determined [24 - 26]. Levels of total protein, MDA and GSH were also measured [27 - 29]. The activity of GR was determined using a previously described method [30].
## III. RESULTS
### a) Effect of Extracts of D. guineense Stem Bark on Relative Organ Weight
As shown in Table 1, there were no significant differences in relative organ weight among the groups $(p > 0.05)$.
Table 1: Relative Organ Weights of Rats
<table><tr><td>Group</td><td>Relative organ weight x 10-2</td></tr><tr><td>Normal Control</td><td>3.34± 0.54</td></tr><tr><td>CCl4Control</td><td>3.02± 0.10</td></tr><tr><td>Silymarin</td><td>3.17± 0.16</td></tr><tr><td>Aqueous Extract</td><td>3.52± 0.24</td></tr><tr><td>Ethanol Extract</td><td>2.96± 0.14</td></tr></table>
- Data are relative organ weights and are expressed as mean $\pm$ SEM $(n = 5)$ b) Effect of Extracts of D. guineense Stem Bark on Oxidative Status in Rat Heart
There were no significant differences in the concentrations of cardiac TP among the groups $(p > 0.05)$. The activities of the antioxidant enzymes and level of GSH were significantly lower in $\mathrm{CCl_4}$ control group than in normal control group, but they were increased by extract treatment $(p < 0.05)$. However, the level of cardiac MDA increased by $\mathrm{CCl_4}$ intoxication was significantly reduced after treatment $(p < 0.05)$. These results are shown in Figures 1 to 3.
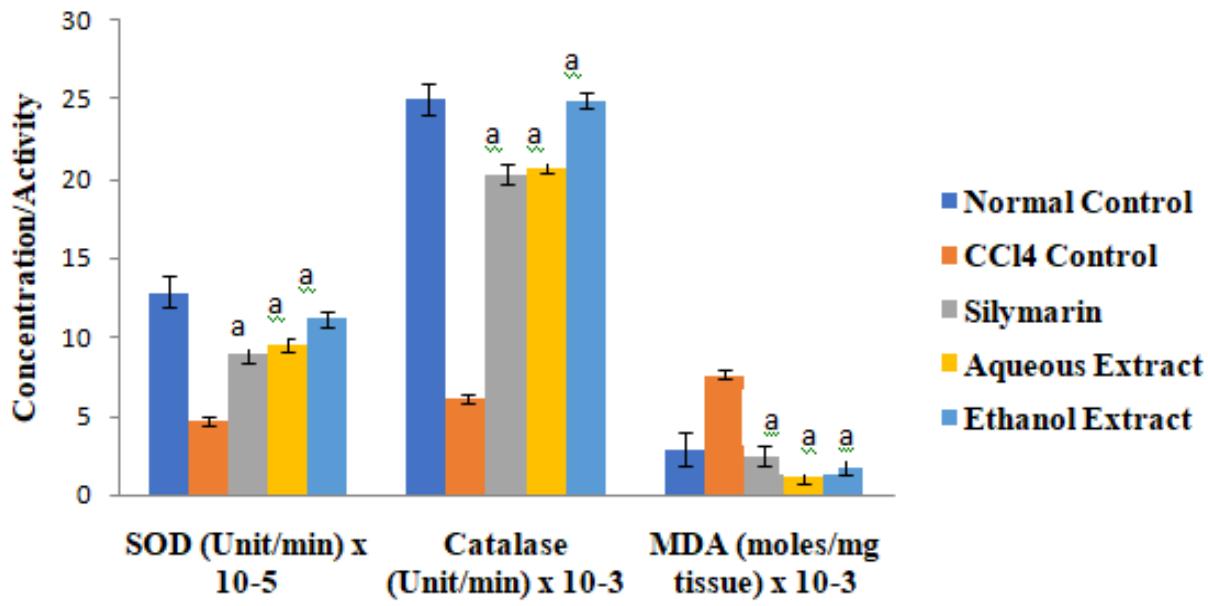 Figure 1: Effect of Extracts of D. guineense Stem Bark on Markers of Oxidative Stress in Rat Heart
Data are oxidative stress markers, and are expressed as mean $\pm$ SEM. $^\mathrm{a}p<0.05$, when compared with $\mathrm{CCl}_4$ control group.
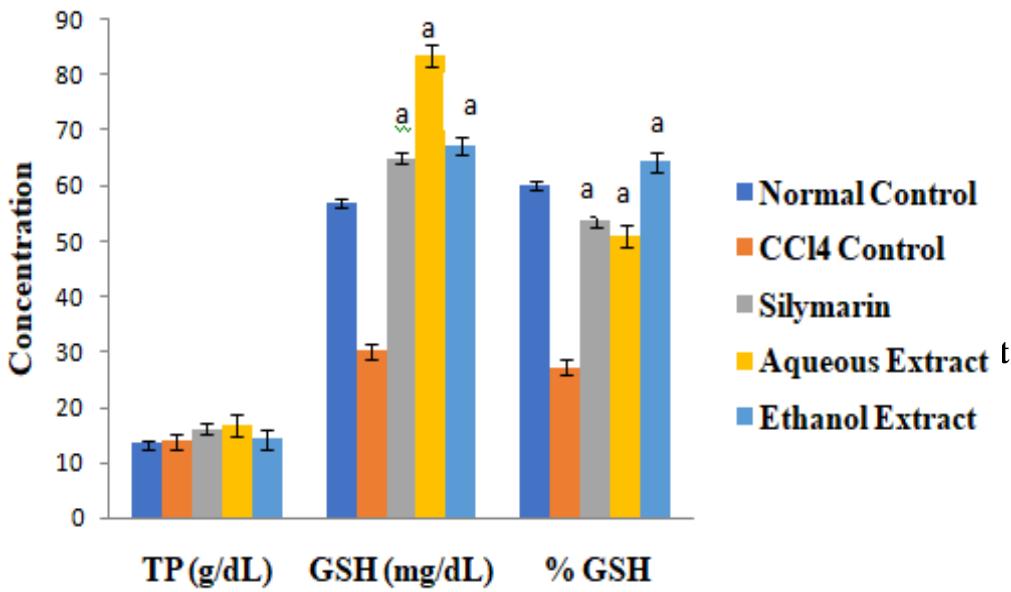 Figure 2: Effect of Extracts of D. guineense Stem Bark on Some Oxidative Stress Parameters
Data are oxidative stress markers, and are expressed as mean $\pm$ SEM. $^\mathrm{a}p<0.05$, when compared with $\mathrm{CCl}_4$ control group.
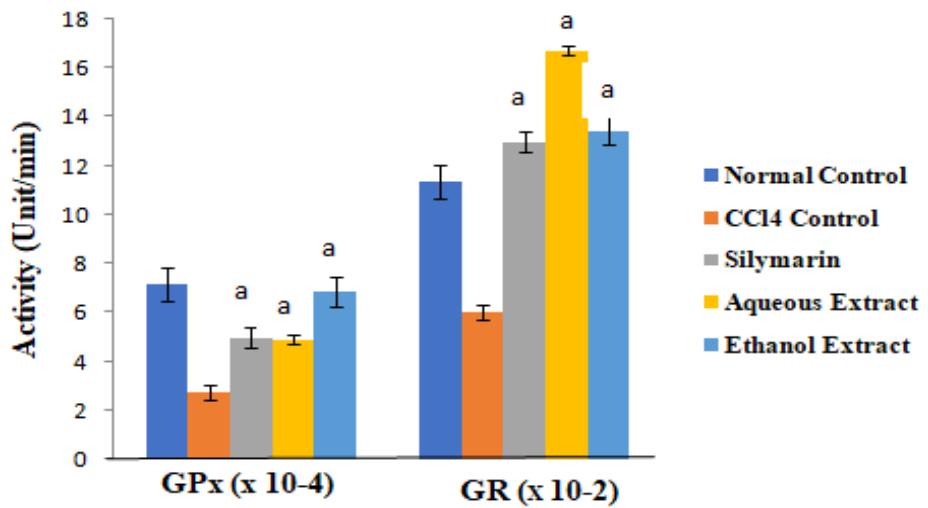 Figure 3: Effect of Extracts of D. guineense Stem Bark on Rat Oxidative Status
Data are oxidative stress parameters, and are expressed as mean $\pm$ SEM. $^a p < 0.05$, when compared with $\mathrm{CCl}_4$ control group.
## IV. DISCUSSION
In animals, $\mathrm{CCl}_4$ is rapidly absorbed via any route of exposure. Once absorbed, it is widely distributed among tissues, especially those with high lipid content. It is metabolized in the body, primarily by the liver, but also in the kidney, lung, and other tissues containing CYP450. The poison reaches its maximum concentration in the liver within 3 h of administration, thereafter it falls and by 24 h it is completely cleared from the organ [7, 8].
Tissue injury produced by $\mathrm{CCl}_4$ is mediated by two major processes resulting from bioactivation in the endoplasmic reticulum (ER) and mitochondria of centrilobular hepatocytes [31]; haloalkylation of cellular macromolecules by reactive metabolites such as trichloromethyl free radical or trichloromethyl peroxyl free radical [32-34]; and lipid peroxidation [35].
Reactive oxygen species (ROS) and oxidative stress have been shown to play an important role in the etiopathogenesis of tissue injury. The role of oxidative stress in cardiac hypertrophy and remodeling has been demonstrated. An increased generation of ROS in the vascular wall and a reduction of nitric oxide (NO) bioavailability lead to endothelial dysfunction in atherogenesis [36, 37]. The ROS cause damage to cellular structures within the vascular wall, thereby triggering several redox-sensitive transcriptional pathways, shifting the cell towards a proatherogenic transcriptomic profile. Animal models of atherosclerosis demonstrate the involvement of ROS in atherosclerosis by the accumulation of lipid peroxidation products and induction of inflammatory genes and activation of matrix metalloproteinases [38, 39]. The ROS and reactive nitrogen species (RNS) produced by the endothelium promote oxidative modification of low-density lipoprotein-cholesterol (LDL-C) in the phase that precedes the transfer into the subendothelial space of the arterial wall, where they initiate atherosclerosis [40].
This study investigated cardiac oxidative status in $\mathrm{CCl}_4$ -exposed rats treated with extracts of Dialium guineense stem bark. The results showed that the activities of the antioxidant enzymes measured as well as level of GSH were significantly lower in $\mathrm{CCl}_4$ control group than in normal control group, but these parameters were increased by extract treatment. However, the level of cardiac MDA increased by $\mathrm{CCl}_4$ was significantly reduced after treatment. These results suggest that extracts of $D$. guineense stem bark may enhance antioxidant defense in rat heart exposed to $\mathrm{CCl}_4$. The capacity of extracts of the medicinal plant to potentiate natural antioxidant defense system has been reported [41-43]. Plants rich in polyphenols are reported to possess good antioxidant capacity [44-46]. Plants with cardioprotective potential have been shown to contain a variety of bioactive compounds, such as diosgenin, isoflavones, sulfuraphane, carotenoids, catechins, quercetin, allicin, cardiac glycosides, saponin-shatavarins 1-1V, cyclovirobuxine D and triterpenes/triterpenoids [47-49].
The cardioprotective effect of medicinal plants may involve attenuation of the damage in cardiac muscle cells, vascular smooth muscle cells (VSMCs), endothelial cells (ECs), and macrophages and monocytes. In cardiomyocytes, cardioprotective agents may promote the opening of $\mathsf{K}_{\mathsf{ATP}}$ channel, increased secretion of atrial natriuretic peptide, as well as the regulation of cardiac hypertrophy, oxidative stress, and apoptosis [50, 51].
## V. CONCLUSION
The results of this study suggest that aqueous and ethanol stem bark extracts of $D$. guineense enhance antioxidant defense in rat heart exposed to $\mathrm{CCl}_4$. Their bioactive molecules may exert cardioprotective function via suppression of specific factors, regulation of key enzymes, and scavenging of oxygen-free radicals.
Generating HTML Viewer...
References
52 Cites in Article
Richard Doherty (2000). A History of the Production and Use of Carbon Tetrachloride, Tetrachloroethylene, Trichloroethylene and 1,1,1-Trichloroethane in the United States: Part 1--Historical Background; Carbon Tetrachloride and Tetrachloroethylene.
Ann Doherty,Sian Ellard,Elizabeth Parry,J Parry (1996). An investigation into the activation and deactivation of chlorinated hydrocarbons to genotoxins in metabolically competent human cells.
Iarc (1999). Carbon tetrachloride. In Re-evaluation of Some Organic Chemicals, Hydrazine, and Hydrogen Peroxide. IARC Monographs on the Evaluation of Carcinogenic Risk of Chemicals to Humans.
M Rossberg,L Wilhelm,P Gerhard,T Adolf,D Eberhard-Ludwig,L Ernst,J Heinz,K Peter,S Heinz,C Richard,B Uwe,L Karl-August,R Theodore,L Eckhard,K Klaus (2006). Drei Fragen an die Komponisten: Klaus Ager, Martin Bjelik, Thomas Christian David, Heinz Karl Gruber, Ernst Kölz, Paul Kont, Wolfgang R. Kuhizek, Kurt Rapf, Marcel Rubin, Jenö Takäcs, Alfred Uhl, Gerhard Wimberger, Wilhelm Zofe.
W Reusch (2013). Introduction to Nuclear Magnetic Resonance Spectroscopy.
M Holbrook (1993). Carbon tetrachloride.
Lester Reinke,Edward Janzen (1991). Detection of spin adducts in blood after administration of carbon tetrachloride to rats.
U Sanzgiri,H Kim,S Muralidhara,C Dallas,J Bruckner (1995). Effect of Route and Pattern of Exposure on the Pharmacokinetics and Acute Hepatotoxicity of Carbon Tetrachloride.
J Benson,B Tibbetts,K Thrall (2001). Uptake, tissue distribution, and fate of inhaled carbon tetrachloride: comparison of rat, mouse, and hamster.
C Taber,D Venes (2009). Taber's cyclopedic medical dictionary.
F Davies,Co (2009). U.
K Moore,A Dalley,A Agur (2009). Clinically oriented anatomy.
A Guyton,J Hall (2006). Sports Physiology.
M Macdonald (2009). Your Body: The Missing Manual.
A Maton,H Jean,W Charles,J Susan,Q Maryanna,L David,Jill,D Wright (1993). Human Biology and Health.
T Dubose (1996). Fennema's Food Chemistry.
William Brattin,Eric Glende,Richard Recknagel (1985). Pathological mechanisms in carbon tetrachloride hepatotoxicity.
T Grabley,C Akuodor (2010). Medicinal Plants and Traditional Medicine.
Osahon Abu,Iyere Onoagbe (2019). Biochemical effect of aqueous extract of Dialium Guineense stem bark on oxidative status of normal wistar rats.
A Sofowora (1993). Medicinal Plants and Traditional Medicine in Africa. 2nd Edition.
Peter Akah,Alphonsus Nwambie (1994). Evaluation of Nigerian traditional medicines: 1. Plants used for rheumatic (inflammatory) disorders.
J Dalziel,J Hutchison (1973). Flora of West Tropical Africa.
Joanne Bero,Habib Ganfon,Marie-Caroline Jonville,Michel Frédérich,Fernand Gbaguidi,Patrick Demol,Mansourou Moudachirou,Joëlle Quetin-Leclercq (2009). In vitro antiplasmodial activity of plants used in Benin in traditional medicine to treat malaria.
O Abu,K Imafidon,M Iribhogbe (2017). Aqueous leaf extract of Icacina trichanta Oliv. ameliorates CCl 4 -induced liver toxicity in Wistar rats.
Gerald Cohen,Dorothy Dembiec,Judith Marcus (1970). Measurement of catalase activity in tissue extracts.
Hara Misra,Irwin Fridovich (1972). The Role of Superoxide Anion in the Autoxidation of Epinephrine and a Simple Assay for Superoxide Dismutase.
J Rotruck,A Pope,H Ganther,A Swanson,D Hafeman,W Hockstra (1973). Selenium biochemical role as a component of glutathione peroxidase.
R Henry,Charles. Sobel,Sam. Berkman (1957). Interferences with Biuret Methods for Serum Proteins Use of Benedict's Qualitative Glucose Reagent as a Biuret Reagent.
George Ellman (1959). Tissue sulfhydryl groups.
John Gutteridge,Stephanie Wilkins (1982). Copper‐dependent hydroxyl radical damage to ascorbic acid.
O Abu,O Ikponmwosa-Eweka (2022). Evaluation of the Potential of Total saponins and Tannins of Dialium guineense Stem Bark in the Amelioration of Carbon Tetrachloride-Induced Renal Oxidative Stress.
J Raucy,J Kraner,J Lasker (1993). Bioactivation of halogenated hydrocarbons by cytochrome P4502E1.
Bruce Mico,Lance Pohl (1983). Reductive oxygenation of carbon tetrachloride: Trichloromethylperoxyl radical as a possible intermediate in the conversion of carbon tetrachloride to electrophilic chlorine.
J Poyer,P Mccay,E Lai (1980). Confirmation of assignment of the trichloromethyl radical spin adduct detected by spin trapping during 13C-carbon tetrachloride metabolism in vitro and in vivo.
T Slater (1981). Free Radicals as Reactive Intermediates in Tissue Injury.
Lutz Weber,Meinrad Boll,Andreas Stampfl (2003). Hepatotoxicity and Mechanism of Action of Haloalkanes: Carbon Tetrachloride as a Toxicological Model.
R Lee,M Margaritis,K M. Channon,C Antoniades (2012). Evaluating Oxidative Stress in Human Cardiovascular Disease: Methodological Aspects and Considerations.
K Channon,T Guzik (2002). Mechanisms of superoxide production in human blood vessels: relationship to endothelial dysfunction, clinical and genetic risk factors.
F Liao,A Andalibi,J Qiao,H Allayee,A Fogelman,A Lusis (1994). Genetic evidence for a common pathway mediating oxidative stress, inflammatory gene induction, and aortic fatty streak formation in mice..
S Rajagopalan,X Meng,S Ramasamy,D Harrison,Z Galis (1996). Reactive oxygen species produced by macrophage-derived foam cells regulate the activity of vascular matrix metalloproteinases in vitro. Implications for atherosclerotic plaque stability..
Roland Stocker,John Keaney (2004). Role of Oxidative Modifications in Atherosclerosis.
O Abu,K Imafidon,H Obayuwana (2020). Evaluation of in vitro antioxidant activities of extracts of Citrullus lanatus seed.
Abu O.D (2020). Quantitative Phytochemical Evaluation and Phenolic Contents Of Extracts Of Citrullus Lanatus seed.
O Abu,K Imafidon,H Obayuwana (2021). Nephrotoxic and in vivo antioxidant effects of citrulluslanatus seed extract.
O Abu,I Onoagbe,I Ojo (2020). Dose Response of Total Saponins Isolated from the Stem Bark of Dialium Guineense.
O Abu,I Onoagbe (2020). ACUTE TOXICITY OF AQUEOUS AND ETHANOL EXTRACTS OF DIALIUM GUINEENSE STEM BARK.
N Bopana,S Saxena (2007). Asparagus racemosus-ethnopharmacological evaluation and conservation needs.
N Sheena,B Lakshmi,K Janardhanan (2005). Therapeutic potential of Ganoderma lucidum (Fr.) P. Karst.
G Jain,S Jhalani,S Agarwal,K Jain (2007). Hypolipidemic and antiatherosclerotic effect of Leptadenia pyrotechnica extract in cholesterol fed rabbits.
Anand Zanwar,Mahabaleshwar Hegde,Subhash Bodhankar (2011). Cardioprotective activity of flax lignan concentrate extracted from seeds of Linum usitatissimum in isoprenalin induced myocardial necrosis in rats.
C Huang (2016). Chinese herbal medicine on cardiovascular diseases and the mechanisms of action.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Abu O.D.. 2026. \u201cCardiac Oxidative Status in CCl 4 -Exposed Rats Treated with Extracts of Dialium guineense Stem Bark\u201d. Global Journal of Science Frontier Research - B: Chemistry GJSFR-B Volume 22 (GJSFR Volume 22 Issue B1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.