## I. INTRODUCTION
Though uncommon with incidence rate of 1 to 2 in 100,000 people globally per year (py) [1], CRAO is an ocular emergency with ability to cause rapid irreversible visual loss and deterioration [2], a manifestation which could be omen for mortality in those affected. In some Asian countries the incidence is 6 in 100,000 people py [3]. In the USA, it is 2 in 100,000 py [4]. The magnitude of the problem has not been well established in Africa due to paucity of research in that field. However, in the study hospital in Ghana it is 2 in 100,000 py.
The most common aetiology as determined by scientific studies is ipsilateral internal carotid artery stenosis [5]. The heart, aortic arch and great vessels may throw emboli to the central retinal artery and cause the disease [6]. Findings from the EAGLE study corroborate the fact that cardiovascular risk factors like obesity, hypertension, tobacco use, hypercholesterolemia and diabetes are the major risk factors associated with the disease [7]. There are three major events capable of leading to CRAO: embolism, in situ thrombosis and vascular spasm with the former being the most common. Emboli may be formed from cholesterol, calcium and platelet-fibrin [8]. Other overlooked, yet common embolus forming agents include fat from fracture of long bones, sickled cells, amniotic fluid, metastatic cells from tumours, gestational trophoblastic disease, septic cells from endocarditis, hepatic hydatid cyst, particulate material from intravenous injection, foreign material from catheter insertion, gas from central venous cannulation surgery and chemotherapy [9].
CRAO-associated thrombi may be caused by atherosclerosis, collagen-vascular disease, inflammatory, and/or hypercoagulable states. Diseases like polycythemia vera, sickle cell anemia, multiple myeloma, systemic lupus erythematosus, factor V Leiden, prothrombin mutation, hyperhomocysteinemia, polyarteritis nodosa, giant cell (temporal) arteritis, antiphospholipid syndrome, activated protein C resistance, Behcet disease, and syphilis are therefore causes of CRAO. Appropriate clinical judgment to differentiate thromboembolic from arteritic processes is crucial as rapid treatment with steroids for vasculitis is associated with improved outcomes in CRAO [8].
Pathophysologically, occlusion of the central retinal artery results in retinal ischemia, vision loss, and eventual necrosis. Additionally, there is retinal edema with pyknosis of the ganglion cell nuclei. Progression of the ischaemia makes the retina become opacified and yellow-white in appearance. Research has proved that incomplete CRAO, permanent retinal damage occurs in just over 90 minutes [8]. In the clinical setting where occlusion may be partial, the return of vision may be achieved after 8 to 24 hours. Approximately $15\%$ of the general population has significant macular collateral circulation from the cilioretinal artery. Patients with this anatomical variant typically may have less severe presentations and better long-term prognoses [8]. Typically, the clinical features of CRAO include sudden, painless, monocular vision loss that occurs over seconds characterized by relative afferent pupillary defect, box-carring, cherry red spot and pale retina (fig 2 and 3).
Once an individual develops CRAO, there is no standard therapeutic intervention which can be used to improve vision in the affected eye [10]. Non-invasive treatments used without success include ocular massage, hyperbaric oxygen (HBOT), carbogen inhalation, intraocular pressure reduction, anticoagulation, sublingual isosorbide dinitrate (SISDN) and systemic steroid. Invasive therapies, some of which made sight in the affected eye worse, include anterior chamber paracentesis [11], laser embolectomy [12], pars plana vitrectomy with direct central retinal artery massage [13] and intraarterial thrombolysis [14].
Many are the theories which have been propounded to emphasise that the retina forms part of central nervous system [15]. Characteristics of blood vessels within the brain and retina are therefore similar. As shown in fig 1, the first branch of internal carotid is ophthalmic whose first branch is central retinal artery. Similarly, other arteries branch off from the internal carotid to supply other parts of the brain. An occlusion of retinal artery may be a strong indication that other vessels could be equally occluded in almost all the vital organs in the body (heart, brain, kidney, liver, lung) to cause ischaemia and possible mortality. In fact, it has been scientifically proved that the same atherosclerotic risk factors for peripheral vascular, cerebrovascular and cardiovascular diseases are present in CRAO and these must be timely and properly assessed to prevent mortality, and morbidity in the better eye [16]. Ischaemic stroke and ischemia in general do not have well established treatment and CRAO is not an exception [17,18,19,20].
One of the branches of ophthalmic artery is the cilioretinal artery (CRA) found in 14 to $26\%$ of the general population [21]. It constitutes extra source of blood supply to the macula. The fortunate patients who have CRA maintain their central vision even when they get CRAO [22]. Their peripheral vision, however, remains seriously and irreversibly impaired [16].
To date a few publications have shown several types of unspecific treatment with unfavourable visual prognosis of affected eye in management of CRAO [10,11,12,13,14]. To the best of our knowledge, this is the first time a study is being published to ascertain visual prognosis of cross section of Africans who had CRAO.
The purpose of the study was to analyse visual prognosis of patients in Sub-Saharan Africa who underwent different types of treatment after being diagnosed with CRAO.
## II. MATERIALS AND METHODS
This is an analytical comparative retrospective study carried out from October 2020 to review medical records of 40 patients (40 eyes) who underwent various types of management from September 2013 to September 2021 after being diagnosed with CRAO (fig 2 and 3) in the study hospital.
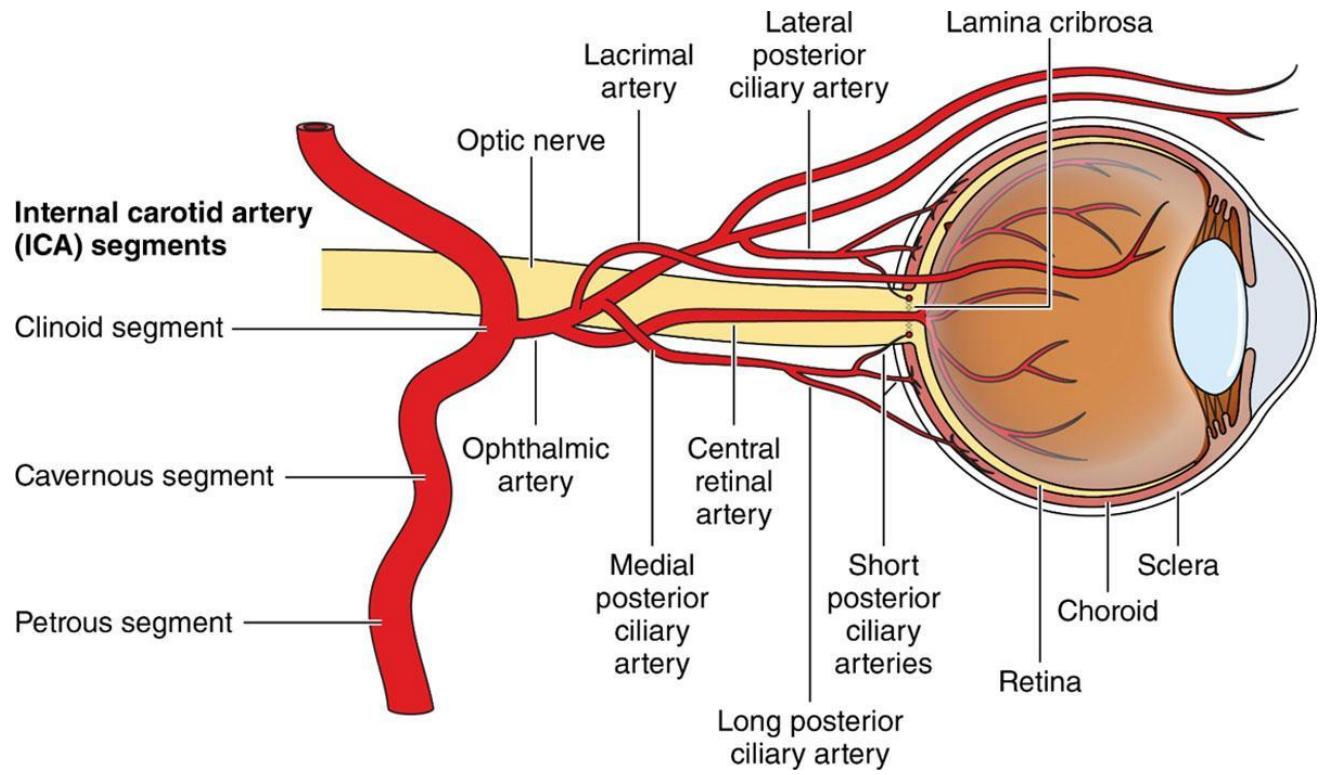 Fig. 1: Origin of central retinal artery from major blood vessel
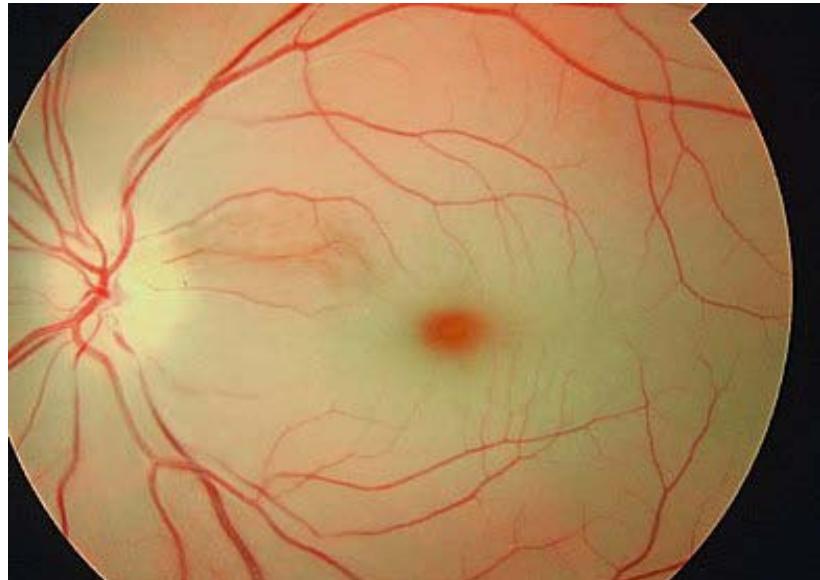
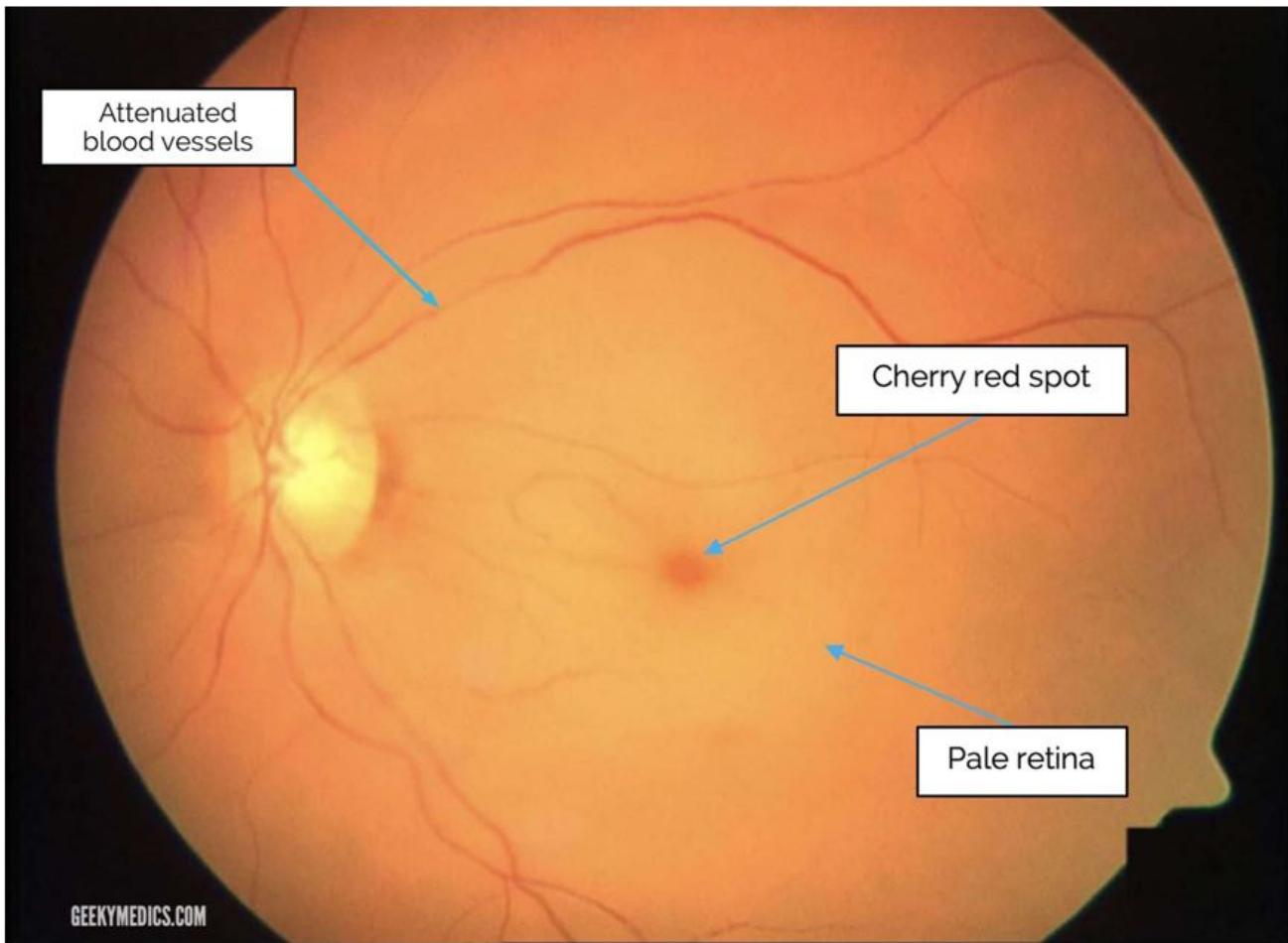 Fig. 2: CRAO with cherry red spot, attenuated blood vessels, pale retina and disc Fig. 3: CRAO
These patients had a minimum follow up of 5 years. One experienced Consultant Vitreoretinal and Ophthalmic Surgeon performed all the procedures and followed up the patients on regular basis. Institutional ethical approval was acquired for this research and tenets of Declaration of Helsinki, applied.
Inclusion criteria: Patients in the study were those who were examined and diagnosed at the retina clinic of 37 Military Hospital in Accra, Ghana. Exclusion criteria were other co-morbidities in the affected eye: age related macular degeneration, macular hole, end stage glaucoma, retinitis pigmentosa, history of retinal detachment surgery and trauma. Out of 67 patients whose medical records were reviewed, 27 were excluded from the study because they were either followed up for less than 5 years or lost to follow up.
Some of the patients had been referred from other Sub-Saharan African countries. In addition to general demographic data, information on underlying systemic diseases, visual acuity, indication for procedures and latest Best Corrected Visual Acuity (BCVA) was collected and analyzed.
One Consultant Vitreoretinal and Ophthalmic Surgeon (FKO) did comprehensive eye examination at the Out-Patient Department to diagnose CRAO (fig 2 and 3). Those whose intra-ocular pressures were high (between 22 and $25\mathrm{mmHg}$ ) were given selective alpha-2 adrenergic receptor agonist (alphagan-P bd) and or carbonic anhydrase inhibitors (diamox $250~\mathrm{mg}$ od). Other staff assisted in checking the patients's blood pressures, fasting or random blood sugar, fasting lipid profile and weight with the aim to identifying systemic risk factors. All patients were referred to the medical department emergency unit (MDEU) for further assessment, detection and management of systemic risk factors.
Physician specialists at MDEU, on the other hand, referred patients with underlying systemic diseases to the retina clinic for further evaluation especially if the latter complained of visual impairment. Some of these patients were on nasal oxygen therapy (NOT) and other medications (table 2) on account of acute ischaemic heart disease and myocardial infarction. Another group of patients were on sublingual isosorbide dinitrate (Canadian brand Dilatrate-SR 5mg daily) due to angina from coronary artery disease. Another group of patients were on oral anticoagulation therapy (XARELTO, rivaroxaban) on account of atrial fibrillation (20mg daily), pulmonary embolism (15mg twice daily for 3 weeks, then 20mg daily) and deep vein thrombosis
(10mg daily). Those with hypercholesterolaemia were managed with oral atorvastatin (VIATRIS LIPITOR10-80mg daily). Patients who had diabetes were on metformin (oral glucophage 1g to 3g daily). All clients who had hypertension were put on oral captopril (US brand capoten 25-450mg daily).
Snellen BCVA was converted into logarithm of minimum angle of resolution (logMAR) units in order to get better statistical analysis. Patients whose visual acuities were hand movement were assigned equivalence of 1.7 logMAR units.
## III. STATISTICAL ANALYSIS
For normally distributed variables, paired t-test was used. All tests were considered statistically significant if p-value was 0.05 or less. Chi square test and paired t-test with SPSS and Graph Pad software were used respectively as shown in table 1.
## IV. RESULTS
Although all the patients had underlying systemic diseases, only 24 (60%) knew they had a chronic disease before the onset of CRAO. A total of 40 eyes of 40 patients (15 females and 25 males) were identified. Mean age at diagnosis was $70.4 + 3.5$ (range 68-75 years) with minimum follow up period of 5 years (range 5 - 8).
Pre- and post-treatment BCVA which was generally hand motion did not change from the onset of CRAO until last follow up visit. Mean pre-treatment BCVA was $2.40 + 2.00\log \mathrm{MAR}$ units which depended on stage and severity of disease. The mean difference between final post- and pre-treatment visual acuity was $0.00 \pm 2.00$ log MAR units which was statistically significant $(p < 0.005)$.
Table 1: Shows statistical analysis
<table><tr><td>Syntax</td><td></td><td>T-TEST
/TESTVAL=40
/MISSING=ANALYSIS</td></tr><tr><td></td><td></td><td>/VARIABLES=PRETREATM
ENT POSTTREATMENT
/CRITERIA=CI(.95).</td></tr><tr><td>Resources</td><td>Processor Time</td><td>00:00:00.00</td></tr><tr><td></td><td>Elapsed Time</td><td>00:00:00.02</td></tr></table>
One-SampleStatistics
<table><tr><td></td><td>N</td><td>Mean</td><td>Std. Deviation</td><td>Std. ErrorMean</td></tr><tr><td>PRE-TREATMENT</td><td>40</td><td>.00425</td><td>.002362</td><td>.000373</td></tr><tr><td>POST-TREATMENT</td><td>40</td><td>.00425</td><td>.002362</td><td>.000373</td></tr></table>
One-Sample Test
<table><tr><td colspan="6">Test Value = 40</td><td></td></tr><tr><td>T</td><td>Df</td><td>Sig. (2-tailed)</td><td>MeanDifference</td><td>95% Confidence Interval of the Difference Lower</td><td></td><td></td></tr><tr><td>PRE-TREATMENT</td><td>-107114.068</td><td>39</td><td>.000</td><td>-39.995750</td><td>-39.99651</td><td></td></tr><tr><td>POST-TREATMENT</td><td>-107114.068</td><td>39</td><td>.000</td><td>-39.995750</td><td>-39.99651</td><td></td></tr></table>
In all 40 eyes were managed for CRAO with the most common aetiologies being hypertension (24;60%), hypercholesterolaemia (5;12.5%) and diabetes(5;12.5%) [table-2]. The most common causes of mortality were hypertension induced myocardial infarction (5;12.5%), pulmonary embolism (3;7.5%) and hypertension induced atrial fibrillation (1;2.5%). The use of isosorbide dinitrate, nasal oxygen therapy (NOT), anticoagulation and reduction in intraocular pressure was not able to improve visual acuities in the affected eyes. However, they played important role in prevention of some deaths and preserved vision in affected or contralateral better eyes as shown in table 2.
Table 2: CRAO Management Plan Summary
<table><tr><td>SRL</td><td>Underlying Systemic Disease</td><td>Additional Eye Disease Apart from CRAO</td><td>Number of patient</td><td>Number of Eyes N (%)</td><td>Systemic Intervention</td><td>Primary Ocular Therapy</td><td>Mortality N (%)</td></tr><tr><td>1</td><td>HPT and Angina</td><td>Nil</td><td>10</td><td>10 (25%)</td><td>Captopril and ISDN</td><td>Nil</td><td>0(0%)</td></tr><tr><td>2</td><td>Isolated HPT</td><td>Nil</td><td>7</td><td>7(17.5%)</td><td>Captopril</td><td>Nil</td><td>0(0%)</td></tr><tr><td>3</td><td>Hypercholesterolaemia</td><td>Nil</td><td>5</td><td>5 (12.5%)</td><td>Atorvastatin</td><td>Nil</td><td>0(0%)</td></tr><tr><td>4</td><td>Diabetes Mellitus</td><td>Ocular Hypertension</td><td>5</td><td>5 (12.5%)</td><td>Metformin</td><td>Diamox and alphagan-P</td><td>0(0%)</td></tr><tr><td>5</td><td>HPT and Myocardial Infarction</td><td>Nil</td><td>5</td><td>5 (12.5%)</td><td>Captopril and nasal oxygen therapy</td><td>Nil</td><td>5 (12.5%)</td></tr><tr><td>6</td><td>Pulmonary Embolism</td><td>Nil</td><td>3</td><td>3 (7.5%)</td><td>Anti-coagulation</td><td>Nil</td><td>3 (7.5%)</td></tr><tr><td>7</td><td>HPT and Atrial Fibrillation</td><td>Nil</td><td>1</td><td>1 (2.5%)</td><td>Captopril and Anti-coagulation</td><td>Nil</td><td>1 (2.5%)</td></tr><tr><td>8</td><td>DVT</td><td>Nil</td><td>3</td><td>3 (7.5%)</td><td>Anti-coagulation</td><td>Nil</td><td>0(0%)</td></tr><tr><td>9</td><td>HPT and IHD</td><td>Nil</td><td>1</td><td>1 (2.5%)</td><td>Captopril and nasal oxygen</td><td>Nil</td><td>1 (2.5%)</td></tr></table>
## V. DISCUSSION
The idea behind ocular massage is to create fluctuations in intraocular pressure with intermittent retina arteriolar dilatation resulting in mechanical disintegration of the clot and reperfusion[23]. Although use of ocular massage to treat CRAO started in 1880, no study has demonstrated and established its efficacy. It has therefore been relegated to the background in modern literature since there is no scientific evidence to support its use. In the study hospital, the first two eyes which were massaged became hypotensive with corneal oedema. Subsequently, no other eye was subjected to massaging.
Hyperbaric oxygen therapy (HBOT) is used as a means of protecting retinal tissue in acute CRAO from severe ischaemia. Physiologically, choroidal circulation nourishes the retina with $50\%$ of its oxygenneeds [24], a concentration which increases up to $97\%$ with the use of HBOT [25]. A lot of scientific researchers have arrived at the conclusion that HBOT enhances visual recovery in the acute phase of CRAO [26,27,28] without necessarily re-establishing reperfusion of the retina permanently. Accordingly, another study published by Rozenberg et al substantiated the fact that HBOT improves visual acuity significantly even 9 hours after CRAO [29]. Luca Rosignoli et al, however, have categorically established through their research that HBOT does not improve visual acuity [30]. Additionally, they realized its use is marred by complications like barotrauma, ear pain, tympanic membrane rupture, and generalized seizures due to oxygen toxicity of the central nervous system [30]. As the study hospital lacks HBOT, 6 patients with 6 eyes were subjected to nasal oxygen therapy due to underlying diseases like myocardial infarction (MI) and Ischaemic Heart Disease (IHD) as soon as the events occurred. There was no improvement of their visual acuities.
Carbogen is made up of $5\%$ carbon dioxide (CO2) and $95\%$ oxygen. The theory behind its use in CRAO management is that CO2 dilates the central retinal artery paving the way for passage of oxygen to nourish the retina [31,32]. Its practical use, however, is characterized by contradictions and poor outcomes [33,34]. In the study hospital, this therapy was not applied because it was not available.
Other researchers reduced intraocular pressure (IOP) pharmacologically even if it was normal to restore sight in CRAO but were not successful [35]. In the study hospital, 5 eyes in 5 patients were managed with carbonic anhydrase inhibitors (diamox) and a-2 selective adrenergic receptor agonist (alphagan) due to high IOP. None of the patients had improvement of their visual acuities.
Isosorbide dinitrate (ISDN) helps in generation of nitric oxide and contributes to dilation of choroidal, retinal and optic nerve vessels. Based on this theory, it has been used in combination with other therapies to re-establish retinal blood flow in CRAO [36]. In the study hospital, none of the 10 patients with 10 eyes who were treated with ISDN on account of angina had improvement of their visual acuities.
According to Cochrane review, there is no conservative treatment combined or monotherapy which is superior to placebo in management of CRAO [10,37]. In an attempt to get better results, researchers recruited 419 patients in 8 different studies which disappointedly gave poorer outcomes than the Cochrane study [38]. Accordingly, the American Academy of Ophthalmology recently conducted research concluding that conservative therapies play no role in the management of CRAO [39].
FieB A et al, in their study, established that anterior chamber paracentesis did not contribute to gaining of visual acuity regardless of interval between onset of symptoms and time of treatment [11]. Opremcak E et al also published a study in which lysis of emboli was achieved with YAG laser embolectomy. However, this procedure was marred by complications like vitreous haemorrhage and pseudoaneurysms making the whole procedure unreliable [12]. Similarly, a research conducted by Lu N et al revealed that pars plana vitrectomy with direct massage of the central retinal artery does not yield good results [13]. In the study hospital, none of the above invasive therapies was used since they did more harm than good.
Intra-arterial thrombolysis (IAT) within 4.5 hours of occurrence of CRAO gained some good results [40,41,42,43]. Other studies, however, have shown poor results [44,45]. The NINDS trial [46], which involved 291 participants, found symptomatic intracranial haemorrhage within 36 hrs of treatment in $8\%$ of the thrombolysis treatment group of which $4\%$ was fatal compared with no events in the control group. Moreover, $3\%$ of patients in the treatment group developed asymptomatic intracranial haemorrhage within 36 hrs of treatment and another $3\%$ of the treatment group developed symptomatic intracranial haemorrhage on long-term follow-up. Since there was no IAT facility in the study hospital, pharmacological thrombolysis was made in 7 patients with 7 eyes who had anticoagulation on account of pulmonary embolism, deep vein thrombosis and atrial fibrillation within 4 hours of onset of the disease. None of them had improvement of their visual acuities. Thrombolysis (pharmacological or IAT) is therefore not a reliable modality of management.
Since there is no known established treatment for CRAO, it is therefore mandatory to re-channel its management towards secondary prevention: prevent mortality from peripheral vascular, cardiovascular and cerebrovascular diseases, avoid blindness in the affected eye and stop similar visual impairment occurring in the better eye. Prophylaxis can only be achieved if the underlying risk factors are detected and modified early enough [7]. In the study hospital, $25\%$ $(n = 10)$ of patients died from several causes the most common of which was hypertension with myocardial infarction as shown in table 2.
## VI. LIMITATION
Limitations of this study include its retrospective nature, single-centre focus, variable follow up lengths, and the fact that one retina specialist performed all procedures. Moreover, invasive therapies were not utilized due to lack of equipment or fear of worsening the patients' already poor sight.
## VII. CONCLUSION AND FUTURE THERAPEUTIC APPROACH
Ischaemic cerebrovascular accidents, myocardial infarction and CRAO have similar pathogenesis and therapeutic interventions. Although many ophthalmologists have diverse ways of managing CRAO, the fact remains that currently there is no conventionally accepted treatment modality substantiated by scientific evidence. Treatment dilemma is made worse by the fact that the retina, which is extension of the brain and central nervous system, does not recover from ischaemic injuries because it lacks Schwann cells. Similarly, several comparative studies, as indicated above, have repeatedly established that no therapeutic intervention is superior to placebo or observation. Management of underlying risk factors equally plays a pivotal role as a preventive measure against blindness in same eye, visual impairment in the contralateral eye and death. A multidisciplinary team made up of ophthalmologists, physician specialists, family physicians and neurologists should be formed in all institutions to help in CRAO management.
Future therapeutic approaches should delve into how to revive the retina after CRAO has occurred. The success of such a study will lead to visual recovery, reversal of ischaemic stroke and good prognosis for myocardial infarction.
Generating HTML Viewer...
References
42 Cites in Article
Jacqueline Leavitt,Theresa Larson,David Hodge,Rachel Gullerud (2011). The Incidence of Central Retinal Artery Occlusion in Olmsted County, Minnesota.
Sohan Hayreh,M Zimmerman (2005). Central Retinal Artery Occlusion: Visual Outcome.
S Park,N-K Choi,K Seo (2014). Nationwide incidence of clinically diagnosed central retinal artery occlusion in Korea, 2008 to 2011.
J Leavitt,T Larson,D Hodge,R Gullerud (2011). The incidence of central retinal artery occlusion in Olmsted County, Minnesota.
P Lavin,M Patrylo,M Hollar,K Espaillat,H Kirshner,M Schrag (2019). Stroke risk and risk factors in patients with central retinal artery occlusion.
S Hayreh,P Podhajsky,M Zimmerman (2009). Retinal artery occlusion: associated systemic and ophthalmic abnormalities.
S Sim,Dsw Ting (2017). Diagnosis and Management of Central Retinal Artery Occlusion.
A Fieß,Ö Cal,S Kehrein,S Halstenberg,I Frisch,U Steinhorst (2014). Anterior chamber paracentesis after central retinal artery occlusion: a tenable therapy?.
Emitchel Opremcak,Alan Rehmar,Chet Ridenour,Lisa Borkowski,James Kelley (2008). RESTORATION OF RETINAL BLOOD FLOW VIA TRANSLUMENAL Nd:YAG EMBOLYSIS/EMBOLECTOMY (TYL/E) FOR CENTRAL AND BRANCH RETINAL ARTERY OCCLUSION.
N Lu,N-L Wang,G Wang,X Li,Y Wang (2009). Vitreous surgery with direct central retinal artery massage for central retinal artery occlusion.
Amelie Pielen,Stefanie Pantenburg,Claudia Schmoor,Martin Schumacher,Nicolas Feltgen,Bernd Junker,Josep Callizo (2015). Predictors of prognosis and treatment outcome in central retinal artery occlusion: local intra-arterial fibrinolysis vs. conservative treatment.
the,%3B%20see%20also%20Chapter%2022).
Sudha Cugati,Daniel Varma,Celia Chen,Andrew Lee (2013). Treatment Options for Central Retinal Artery Occlusion.
Kazutaka Shinozuka,T Dailey,N Tajiri,H Ishikawa,D Kim,M Pabon (2013). Stem Cells for Neurovascular Repair in Stroke.
Chao Chen,Yongting Wang,Guo-Yuan Yang (2013). Stem Cell-Mediated Gene Delivering for the Treatment of Cerebral Ischemia: Progress and Prospectives.
Xiaoying Wang,Anna Rosell,Eng Lo (2008). Targeting Extracellular Matrix Proteolysis for Hemorrhagic Complications of tPA Stroke Therapy.
O Angeli,Z Nagy,M Schneider (2019). Spontaneous visual recovery following a central retinal artery occlusion in a patient with a cilioretinal artery.
T Ffytche (1974). Treatment of Nonarteritic Acute Central Retinal Artery Occlusion.
M Landers (1978). Retinal oxygenation via the choroidal circulation.
C Dollery,C Bulpitt,E Kohner (1969). Oxygen supply to the retina from the retinal and choroidal circulations at normal and increased arterial oxygen tensions.
Bekir Bağlı,Sadık Çevik,Mediha Çevik (2018). Effect of hyperbaric oxygen treatment in central retinal artery occlusion.
Ana Lopes,Rita Basto,Susana Henriques,Luísa Colaço,Filomena Costa E Silva,Isabel Prieto,Francisco Guerreiro (2019). Hyperbaric Oxygen Therapy in Retinal Arterial Occlusion: Epidemiology, Clinical Approach, and Visual Outcomes.
Thomas Masters,Bjorn Westgard,Stephen Hendriksen,Alejandra Decanini,Anne Abel,Christopher Logue,Joseph Walter,Joseph Linduska,Kevin Engel (2019). CASE SERIES OF HYPERBARIC OXYGEN THERAPY FOR CENTRAL RETINAL ARTERY OCCLUSION.
Assaf Rozenberg,Aviel Hadad,Alon Peled,Biana Dubinsky-Pertzov,Lior Or,Eva Eting,Shai Efrati,Eran Pras,Adi Einan-Lifshitz (2021). Hyperbaric oxygen treatment for non-arteritic central retinal artery occlusion retrospective comparative analysis from two tertiary medical centres.
S Harino,J Grunwald,B Petrig,C Riva (1995). Rebreathing into a bag increases human retinal macular blood velocity..
Oliver Arend,Alon Harris,Bruce Martin,Mary Holin,Sebastian Wolf (1994). Retinal blood velocities during carbogen breathing using scanning laser ophthalmoscopy.
N Atebara,G Brown,J Cater (1995). Efficacy of anterior chamber paracentesis and Carbogen in treating acute nonarteritic central retinal artery occlusion.
T Deutsch,J Read,J Ernest,T Goldstick (1983). Effects of Oxygen and Carbon Dioxide on the Retinal Vasculature in Humans.
Scott Fraser,Wendy Adams (2009). Interventions for acute non-arteritic central retinal artery occlusion.
S Rumelt,Y Dorenboim,U Rehany (1999). Aggressive systematic treatment for central retinal artery occlusion.
S Fraser,Adams (2009). Interventions for acute nonarteritic central retinal artery occlusion.
Matthew Schrag,Teddy Youn,Joseph Schindler,Howard Kirshner,David Greer (2015). Intravenous Fibrinolytic Therapy in Central Retinal Artery Occlusion.
Christina Flaxel,Ron Adelman,Steven Bailey,Amani Fawzi,Jennifer Lim,G Vemulakonda,Gui-Shuang Ying (2020). Retinal and Ophthalmic Artery Occlusions Preferred Practice Pattern®.
Mac Grory,B Nackenoff,A Poli,S Spitzer,M Nedelmann,M Guillon,B Preterre,C Chen,C Lee,A Yaghi,S (2020). intravenous fibrinolysis for central retinal artery occlusion: a cohort study and updated patientlevel meta-analysis.
Cécile Préterre,Gaelle Godeneche,Xavier Vandamme,Thomas Ronzière,Matthias Lamy,Christophe Breuilly,Cédric Urbanczyk,Valérie Wolff,Pierre Lebranchu,Mathieu Sevin-Allouet,Benoit Guillon (2017). Management of acute central retinal artery occlusion: Intravenous thrombolysis is feasible and safe.
Max Nedelmann,Michael Graef,Frank Weinand,Klaus-Heiko Wassill,Manfred Kaps,Birgit Lorenz,Christian Tanislav (2015). Retrobulbar Spot Sign Predicts Thrombolytic Treatment Effects and Etiology in Central Retinal Artery Occlusion.
Maximilian Schultheiss,Florian Härtig,Martin Spitzer,Nicolas Feltgen,Bernhard Spitzer,Johannes Hüsing,André Rupp,Ulf Ziemann,Karl Bartz-Schmidt,Sven Poli (2018). Intravenous thrombolysis in acute central retinal artery occlusion – A prospective interventional case series.
Rahul Sharma,Nancy Newman,Valérie Biousse (2019). New concepts on acute ocular ischemia.
Mac Grory,B Lavin,P Kirshner,H Schrag,M (2020). Thrombolytic therapy for acute central retinal artery occlusion.
(1995). Tissue Plasminogen Activator for Acute Ischemic Stroke.
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.