Mercury is the most toxic in the organic form like methylmercury (MeHg), with an ability to accumulate in the cells of aquatic organisms like fish. This has been known to adversely affect the functions of almost every aspect of life. This study examined the effects of MeHg exposure on certain antioxidant defences in the liver and muscle of climbing perch (Anabas testudineus) adults and juveniles; the effect on DNA of erythrocytes were also studied. After 15 d of MeHg exposure (87.6 µg L -1 ), the effect of MeHg on DNA of the fish were analysed using comet assay and the activities of SOD (superoxide dismutase), catalase (CAT) and glutathione (GSH) alongwith lipid peroxidation levels (LPO) in the form of TBARS were used as antioxidant markers. The juvenile erythrocytes showed more DNA damage in the form of comets than adults. The activities of SOD significantly increased in all the experimental groups and tissues whereas catalase (CAT) activity increased in adult liver and musclesbut juvenile liver reported a decrease. Glutathione (GSH) levels showed difference in induction depending on the age of the fish with adult liver and muscle showing decrease and juveniles showing significant increase at 15 days of exposure.
## I. INTRODUCTION
Aquatic ecosystems act as a sink for many contaminants that could cause various physiological damage which is not restricted to molecular, biochemical, cellular and physiological damage[1]. According to International Agency for Research on Cancer (IARC) methylmercury (MeHg) is classified as an element belonging to the group 2B, making it a possible human carcinogen[2]. Many studies have been conducted on the effect of such toxic chemicals upon fish genome and antioxidant system[3]. The high toxicity of heavy metals along with their bioaccumulative property make them very important when it comes to environmental contamination as they are able to induce damage in the genes[4]. Genotoxicity studies are extremely important in aquatic ecosystems, as the toxic substances can accumulate in water [5] and fish respond in manner similar to higher vertebrates, and may be used to estimate genotoxic effects that such compounds may cause in animals especially the humans [6]. However, research into clastogenic or mutagenic effects in tropical air-breathing fishes exposed to contaminants is scarce [7].
Mercury and some organomercurial compounds cause genomic damaged due to their effect on tubulin, the component of spindle fibers and important for cytoplasmic organization. The impairment of polymerization causes metaphasic chromosome contraction, delay of centromeric division with slow anaphasic movement[8]. MeHgalso increases the reactive oxygen species (ROS) or free radicals which accelerates reactions that induce genotoxicity.
The main line of defence against pollutants and xenobiotics is the antioxidant system, with components that prevent ROS from being formed or removing it when formed, preventing damage to the vital components of the cell [9]. ROS are also involved in cell functions like signalling; hence, antioxidant system does not entirely remove oxidants, but instead, keeps the quantity at the optimum level [10]. MeHg is known to produce ROS and generate oxidative stress leading to stress damage [11]. Inorganic mercury showed high oxidative-stress-inducing potential in B. amazonicus[12]. Such changes in the enzyme activity have been used as an early warning system to detect the potential to cause adverse effects well before the onset of serious pathological damage in the animals exposed to toxicants [13]. The cell defences against ROS include scavenger compounds such as glutathione and enzymes with antioxidant activities such as SOD, CAT and GPx[14]. The ROS have the capacity to react with lipids, proteins and nucleic acids, which leads to several biochemical injuries. The changes in the antioxidant activities show a shift in the equilibrium in the organism [15] and the induction or decrease in the enzyme activities in relation to oxidative stress is used as the marker for the presence of hazardous substances [16]. These changes are more pronounced in aquatic organisms as they are more sensitive compared to those on land [15].
This study aimed to evaluate genotoxicity by using comet assay on the erythrocytes and the impact on a few components of the antioxidant system in muscles and liver of Anabas testudibesus (Bloch, 1840) exposed to a sublethal concentration of MeHg.
## II. MATERIALS AND METHODS
### a) Test Animal
The test organism A. testudineus (Fig. 1.) was selected as per the criteria put forth by Rand and Petrocelli [17].
### b) Test Chemical (Toxicant)
In the aquatic environment, methylmercury (MeHg) is produced by the action of bacteria on elemental and inorganic mercury. MeHg is a very potent neurotoxin with potential to accumulate in the body of the organism. An organic compound of mercury is used in the experiment. The chemical in the form of the iodide salt (MeHgl) of $98\%$ purity was acquired from Alfa Aeasar, England. The stock solution of 100 ppm was prepared from the salt which was diluted as needed for the experiment.
### c) Median lethal concentration
The median lethal concentration $(96\mathrm{h}\mathrm{LC}_{50})$ for the animal was observed to be $438.21~\mu \mathrm{g}~\mathrm{L}^{-1}$ (95% fiducial CI 314.65 - 610.32 $\mu \mathrm{g}~\mathrm{L}^{-1}$ ) or $0.438~\mathrm{mg}~\mathrm{L}^{-1}$ by probit analysis [18].
For the experimental exposure, 25 fish each of juvenile (average length $9.27 \pm 0.62$ cm and weight $11.96 \pm 2.36$ g) and mature (average length $13.96 \pm 0.93$ cm and weight $36.86 \pm 10.10$ g) life stages were utilised as experimental and control groups. The sublethal concentration was $(\mathrm{LC}_{50} / 5)$; $87.6 \mu \mathrm{g} \mathrm{L}^{-1}$ of MeHg. The fish were exposed as per the semi-static renewal test with the toxicant water renewed every four days and with intermittent feeding.
### d) Comet Assay
At the end of the 15-day exposure period, the exposed fish were sampled along with the control fish. Five fish each were used for the analysis. The blood from the fish was obtained by cardiac puncture and transferred into heparinized tubes. For the comet assay, the method of Singh et al.[19] was used. The slides were photographed using inverted epifluorescent microscope (Olympus CKX41) attached with camera (Opitka Pro5 CCD). Comets were scored using Tritek comet scoring software and correlated statistically.
### e) Antioxidant assay
After exposing to MeHg, five fish from exposure groups along with those from the control experiment were sacrificed by spinal dislocation. The fish were weighed, and the liver and muscle were removed and
We have previously reported that the antioxidant enzymes of the plant are mainly expressed in the plasma membrane (GSH) and in the cytosol. The antioxidant enzymes were detected by HPLC analysis on a Waters 1000 MS spectrometer equipped with an $80^{\circ}\mathrm{C}$ oven for 24 hours. The results showed that the antioxidant enzymes were enriched in the plasma membranes of plants, which is consistent with previous reports [15]. In addition, we found that the antioxidant enzymes were enriched in the plasma membranes of plants under ambient conditions. We also found that the antioxidant enzymes were enriched in the plasma membranes of plants under high temperature conditions. These results suggest that the antioxidant enzymes are enriched in the plasma membranes of plants under high temperatures. However, the antioxidant enzymes were not enriched in the plasma membranes of plants under low temperatures. This suggests that the antioxidant enzymes are enriched in the plasma membranes of plants under low temperatures.
## III. RESULT
### a) Comet Assay
The results showed an increase in the number of comets in both the juveniles and the adults when compared with those of the control. The DNA damage could be visualised as comets with a tail and a head, and the juveniles had more pronounced comets, whereas the control showed minimal comets but pronounced heads. In the case of the length of comets (Fig. 2) the juveniles showed more induction when compared to the adults. The lengths of the comets of the juveniles are significantly longer when compared to the controls and so are those of the adults. Both results showed high significance at $p < 0.01$ revealing that the MeHg exposure for 15 days induces significantly higher DNA damage in the erythrocytes of both juveniles and adults. Even though there are more comets formed in the juveniles than in adults, the results do not show significant difference when compared to each other.
The results do not show significant difference except in the case of juveniles compared to controls. The tail lengths are not significantly different in this case. The erythrocytes of control juvenile fish showed few comets and more of the pronounced (Fig. 3). The comets in the juvenile fish were pronounced (Fig. 4), which are caused by the single strand and double strand damage causing the smaller DNA fragments to move out into the matrix creating the illusion of tails when stained. After 15 days of exposure to sublethal concentration of MeHg, comets were formed in most of the cells in the adults and the DNA damage was pronounced in most cases with the formation of "hedgehogs" (Fig. 6) that are considered to indicate necrotic and apoptotic cells and may not be analysed by the software used for the analysis hence, the insignificant values. Such damage was not observed in the control fish (Fig. 5).
### b) Antioxidant Enzyme Levels
## i. Superoxide dismutase activity
The results of the SOD activity (mean $\pm$ SD) are presented in Fig. 7. All the values increased and are highly significant when compared with the control data. The liver and muscle of the juvenile fish were analysed and found to exhibit increase at $15^{\text{th}}$ day of exposure. In the case of the adults, the liver and muscle showed trend in the SOD levels similar to that in the juveniles.
## ii. Catalase activity
The juvenile liver showed a decrease in activity after 15 days of exposure in comparison with the control values. In the adult liver, activity is significantly increased when compared to the control. The adult muscle showed an increase over the entire time of exposure similar to that seen in the adult liver. After 15 days of exposure, the values kept increasing in both juvenile and adult muscle (Fig. 8). All the values are significant $(p < 0.01)$ with all tissues other than juvenile liver showing increase in the values at 15 days of exposure.
### c) Non-enzymatic Antioxidant Levels
## i. Lipid peroxidation activity via TBARS levels
The quantity of MDA (measured as TBARS) formed in the control liver of the juvenile fish was analysed to be at $276.97 \mu \mathrm{mol} \, \mathrm{mg}^{-1}$ protein and in the mature liver, it was $541.33 \mu \mathrm{mol} \, \mathrm{mg}^{-1}$ protein. The formation of TBARS increased in both the juvenile and adult fish liver. The juvenile liver had the value at 7706.7 $\mu \mathrm{mol} \, \mathrm{mg}^{-1}$ on day 15, whereas in adult liver it increased to $7719.9 \mu \mathrm{mol} \, \mathrm{mg}^{-1}$ protein on day 15. The same was the case with the muscles from both the groups showing increase in the levels of TBARS formed; from values of $66.73 \mu \mathrm{mol} \, \mathrm{mg}^{-1}$ protein to $990 \mu \mathrm{mol} \, \mathrm{mg}^{-1}$ protein in juvenile muscle and $33.28 \mu \mathrm{mol} \, \mathrm{mg}^{-1}$ protein to $729.30 \mu \mathrm{mol} \, \mathrm{mg}^{-1}$ protein in adult muscle (Fig. 9). All the values obtained for TBARS are highly significant ( $p<0.01$ ) when compared to the control values and showed an increase which means the rate of lipid peroxidation also increased during exposure.
## ii. GSH activity
GSH was observed to be 0.02 mmol $\mathrm{mg}^{-1}$ tissue in the liver of juveniles and 0.185 mmol $\mathrm{mg}^{-1}$ in the adult liver at the start of the experiment. With the increase in the duration of exposure to 15 days, GSH levels in the juvenile liver increased, whereas in the adult liver, it decreased. The change in the values is significant ( $p<0.01$ ) when compared to the control. The GSH content in the muscles of the juvenile fish in the control group was found to be 0.00974 mmol $\mathrm{mg}^{-1}$ tissue weight and in the mature fish, the value was 0.0178 mmol $\mathrm{mg}^{-1}$ (Fig. 10). The values decreased in the case of both mature and juvenile fish with significant values ( $p<0.01$ ) and ( $p<0.001$ ), respectively.
 Figures Fig. 1: Test animal - Anabas testudineus
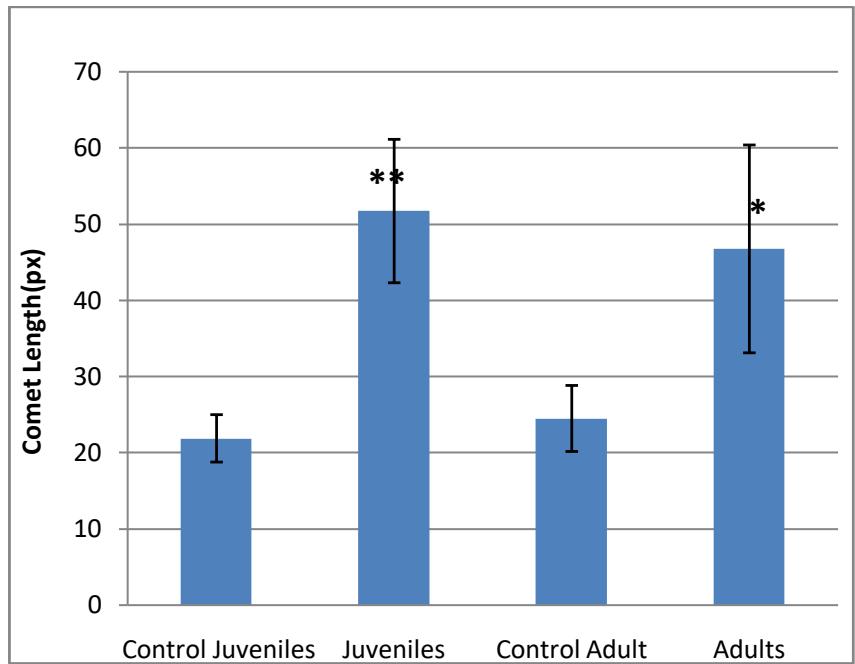 Fig. 2: Comet length in px(mean±SD) obtained in juvenile and adult test fish (\*\*p< 0.001)
 Fig. 3: Erythrocytes from the control juvenile fish
 Fig. 4: Erythrocytes of control adult fish
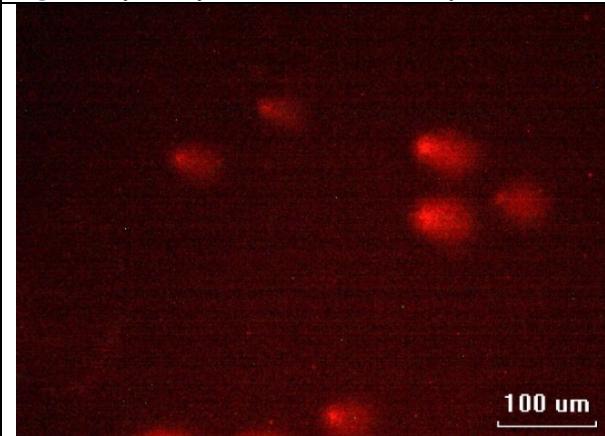 Fig. 5: Comets seen in the juvenile fish after 15 days of exposure to sublethal concentration of MeHg
 Fig. 6: Erythrocytes from adult fish after exposure showing "hedgehogs"
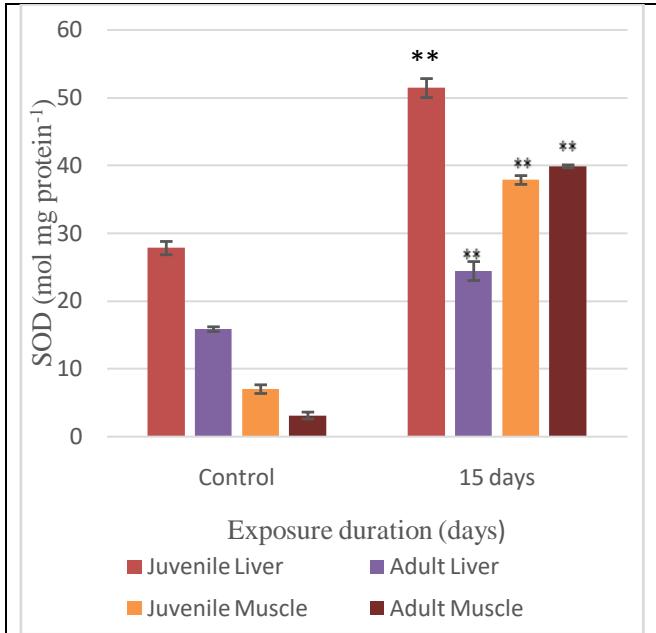 Fig. 7: SOD activity in the liver and muscles
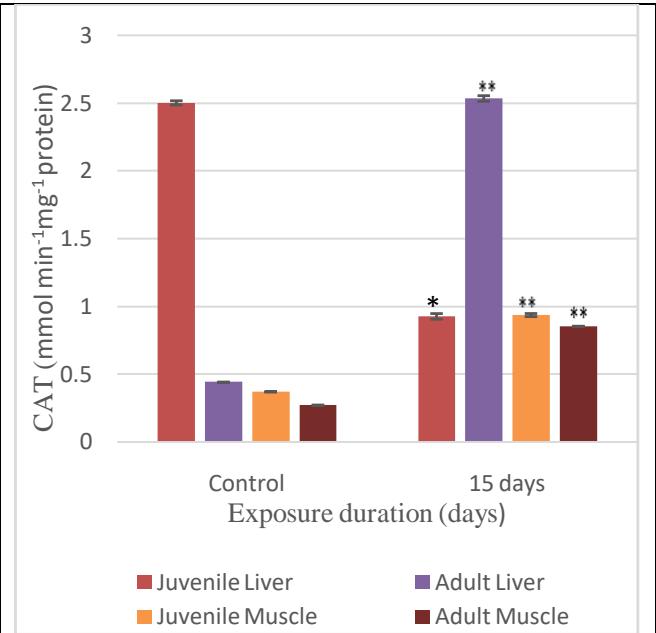 Fig. 8: CAT activity in the liver and muscle
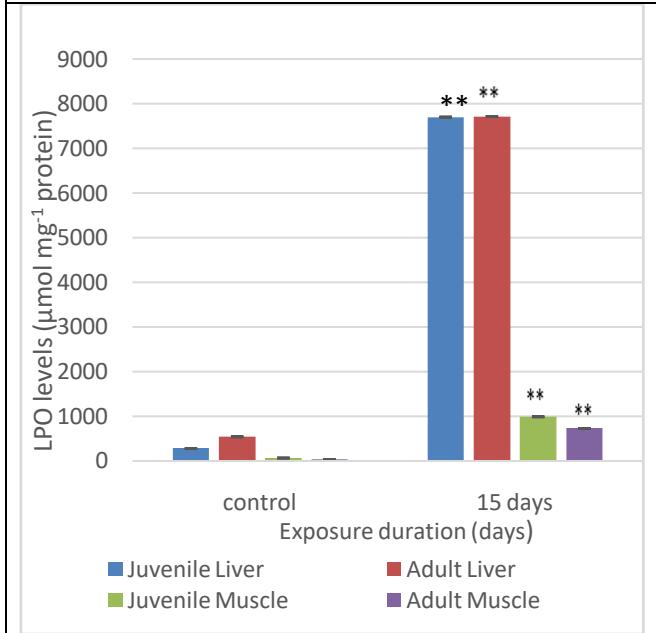 Fig. 9: MDA formed as a result of varying exposure duration of MeHg on the liver and muscle
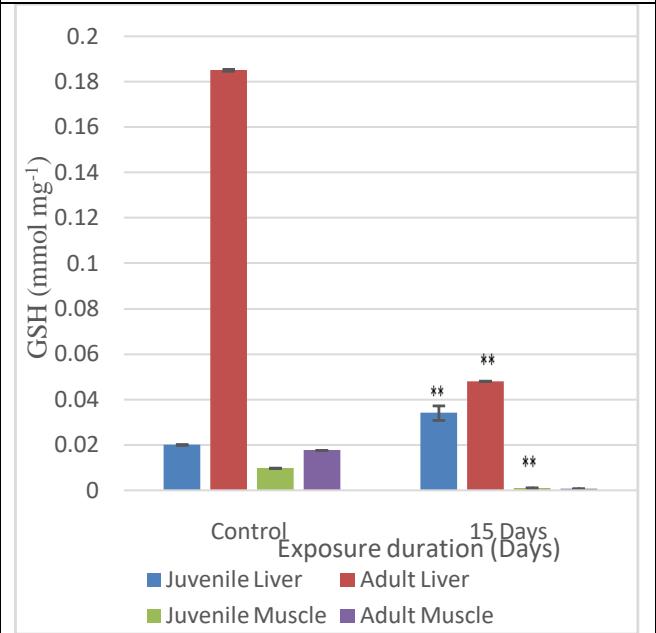 Fig. 10: Levels of GSH in the liver and muscle of juvenile and adult fish on MeHg exposure
## IV. DISCUSSION
Exposure to toxins can affect the genes and alter DNA which is the carrier of information from generation to generation and the altered DNA will be inherited by the future progeny. According to Almeida et al.[24], mutations are any changes or damages in the DNA structure that can be inherited, create carcinogenesis or even cause cell death. Comet assay or SCGE is a rapid and sensitive method to analyse the DNA damage in any tissue [25]. Comet assay is the best procedure for the detection of the damaging effect of chemicals and pollutants under the laboratory and field conditions. It can detect genetic damage due to low levels of toxicant exposure, especially heavy metals like mercury. The types of damages assessed are strand breaks, alkali-labile sites, DNA cross linking and erroneous as well as incomplete DNA excision repair sites. This method was used to assess the DNA damage in the lymphocytes exposed to mutagenic agents[26]. Metal salts create genotoxic effects by the generation of ROS that damage the DNA, and interfere with the DNA repair and replication processes. Creating damages like single and/or double strand breaks, DNA-DNA cross-links, DNA protein cross-links and base modifications[27]. Comet assay has been used to quantify DNA damage in single cells [28] under laboratory conditions [29].
The study conducted on the DNA damage caused due to the exposure of the juvenile and adult fish to the toxicant reveals that MeHg is highly genotoxic. In this study, the lengths of the comets of the juveniles are seen to be significantly higher when compared to those of the controls; same is the case with the adults. Both the results are highly significant at $p < 0.01$. This result shows that MeHg exposure induces significantly higher instances of DNA damage in the erythrocytes of both juveniles and adults. Though more comets were formed in the juveniles than in adults, the statistical analyses do not show significant difference when compared to each other. It is considered that the longer the comet length, the more damaged the DNA is. The DNA damage as per the comet assay shows that the more affected are the juveniles, but the DNA in the adults seem to be highly fragmented creating more of ghosts than comets. Genotoxic agents produce DNA damage that can be either repaired or the damage is irreversible and continues, and such the damage can eventually lead to cell death. MeHg has the ability to damage cellular macromolecules like lipids and DNA by oxidative damage[30].
The sublethal concentration of MeHg in static conditions to which the A. testudineus were exposed to, was sufficient to induce significant alterations in SOD antioxidant enzymes such as and CAT inducing probable oxidative damages in lipids and proteins and, consequently, oxidative stress. SOD and CAT activities increased after 96 hours of exposure to mercuric chloride in all the tissues of B. amazonicus[12]. SOD catalyses the dismutation of $O^2$ -to water and hydrogen peroxide, which is detoxified by CAT. Due to the inhibitory effects on ROS formation, the SOD-CAT system provides the first defence-line against oxygen toxicity due to metals [31] and is usually used as a biomarker indicating ROS production [32; 16]. The induction of the SOD-CAT system indicates a fast adaptive response of the redox-defence system in the liver, gills, white muscle and heart of B. amazonicus after exposure to mercury [12]. On the exposure to effluents, CAT in channel catfish (Ictalurus punctatus) showed a significant increase. The increase or decrease in the activities of antioxidant enzymes depends on the intensity and duration of the exposure of the metals they are exposed to [33].
The most important and easily available redox balance buffer in the living body happens to be glutathione [34]. It is a scavenger of the oxyradical and is important in the antioxidant defence, and also is a very important measure of the detoxification capacity of an organism [35]. The GSH molecule can also scavenge other ROS directly and is involved in various processes vital for normal cellular function such as DNA and protein synthesis [36]. The cells of the organism in contact with metals usually expel them by coupling directly with GSH.Mercury binds to the sulfhydryl groups of glutathione stopping it from functioning as a free radical scavenger [37] and causes the collapse of the antioxidant mechanisms in the cell. This can result in cell degeneration, loss of membrane integrity and eventually, cell necrosis [38], but various studies show that the GSH activities in fishes respond differently to the exposure to mercury. In M. cephalus and L. aurata, mercury exposure shows increase in the GSH content [39], whereas in A. testudineus, the GSH levels decrease on exposure to $0.166\mathrm{mgL}^{-1}\mathrm{Hg}^{2+}$ for 24 hours [40]. The levels of GSH in snakeheads increase when exposed to subacute concentrations of mercury but decrease under longer exposure duration [41]. During long-term exposure, GSH could be involved in the formation of conjugates with $\mathrm{Hg}^{2+}$ leading to the formation of linear II covalent complexes [42]. The cause of the depletion in GSH could be the consumption during the phase II biotransformation (involving GSH-dependent enzymes) observed more in the muscles than in the liver and gill [43]. These complexes are excreted as mercapturic acid leading to the decrease in GSH in order to eliminate mercury [44]. Thus, the decrease in the GSH content could be due to oxidative stress, accumulation and even elimination process. Theliver is the site of GSH synthesis and is exported to extraheptic tissues like kidney, brain and muscle. White muscle has fewer number of mitochondria and are not as efficient at recharging GSH due to lowered transport from the liver [45]. Thus, the depletion of GSH in the muscle is a better indicator of pollutant-based oxidative stress than that in the liver. The levels of GSH normally tend to decrease with the increase in age of the organisms [46], which further decreases during oxidative stress. The increase in the juvenile liver GSH could be due to the increased replenishment of GSH by the uptake of amino acid substrates and the activities of biosynthetic enzymes in the liver. In the case of B. amazonicus, the GSH content in the white muscle decreased [12] similar to the observation in the present study. This decline in GSH content may be due to the ability of GSH to bind directly with mercury or due to the non-conversion of the thiol group back to GSH by GR. In most cases, if the initial increase in the free radicals is small, the antioxidant system is capable to neutralize the effects and readjust the balance between the ROS production and ROS scavenging capacities [47].
The present study revealed higher LPO levels in liver and muscle, a clear oxidative stress indication. LPO is a complex process resulting from reactions in biological membranes causing the formation of lipid hydroperoxides. These molecules are able to fragment the double bonds of unsaturated fatty acids and disintegrate the lipids of the cellular membrane [48]. The oxidative stress was confirmed by the increases in lipid peroxidation in liver, gills, white muscle and heart of B. amazonicus after exposure to mercuric chloride [12]. Similarly, after mercury exposure, increases in LPO levels were also detected in the brain and kidney of the Atlantic salmon [49] and in the liver of Russian sturgeon
Acipenser gueldenstaedti[50]. This study also emphasizes the capability of antioxidants such as CAT, SOD and GSH to be biomarkers of contaminant mediated oxidative stress in a variety of aquatic organisms, and the variations in their levels are a reflection of the response of the organism to pollutants[51].
## V. CONCLUSION
This study showed that MeHg at very low concentration is capable of inducing various major damages to the genetic makeup as well as the antioxidant architecture of exposed fish. MeHg induced the damage at a concentration which is very much below the permissible limits put forth by health agencies and at a very short span of time. Such damage could be detrimental to the organism, and can even create possible defects in the future generations leading to change in the population structure and the genetic makeup of the species as well as the diversity of the ecosystem. This study also reports the formation of highly fragmented DNA beyond computer recognition (ghosts) as a drawback for the comet assay. Organic mercury even at sublethal concentrations and short-term exposure is potent enough to induce an oxidative stress in the case of the air-breathing fish that is otherwise capable of surviving under extreme conditions. These changes can impair DNA and lipid membrane functions, thus affecting the homeostasis. The protective mechanisms against the toxicant-induced stress fall short of maintaining the physiological integrity. The data obtained suggest that mercury at the concentrations that are irrelevant in terms of environmental and health points of view are capable of inducing negative impact on behaviour, health status, reproduction and overall success of A. testudineus, a native air-breathing fish of India, making its survival vulnerable.
### Funding
This research was funded by the Junior Research Fellowship of The University of Kerala
Informed Consent Statement Not applicable.
Data Availability Statement Not applicable.
#### ACKNOWLEDGEMENTS
We would like to thank the Vizhinjam centre of Central Marine Research Institute for the help towards conducting analyses.
Conflicts of Interest The authors declare no conflict of interest.
Generating HTML Viewer...
References
53 Cites in Article
Aline Kirschbaum,Robson Seriani,Camilo Pereira,Andrea Assunção,Denis Abessa,Matheus Rotundo,Maria Ranzani-Paiva (2009). Cytogenotoxicity biomarkers in fat snook Centropomus parallelus from Cananéia and São Vicente estuaries, SP, Brazil.
(2012). Unknown Title.
Da Rocha,C,A,M,Dos Santos,R,A Bahia,M,D,O Da Cunha,L,A Ribeiro,H,F Burbano,R,M,R (2009). The micronucleus assay in fish species as an important tool for xenobiotic exposure risk assessment-A brief review and an example using neotropical fish exposed to methylmercury.
A Bücker,W Carvalho,J Alves-Gomes (2006). Evaluation of mutagenicity and genotoxicity in Eingenmanniavirescens (Teleostei: Gymnotiformes) exposed to benzene.
Marcos Ferraro,Alberto Fenocchio,Mario Mantovani,Ciro Ribeiro,Marta Cestari (2004). Mutagenic effects of tributyltin and inorganic lead (Pb II) on the fish H. malabaricus as evaluated using the comet assay and the piscine micronucleus and chromosome aberration tests.
K Al-Sabti,C Metcalfe (1995). Fish micronuclei for assessing genotoxicity in water.
Marta Cestari,Priscilla Lemos,Ciro Ribeiro,João Costa,Emilien Pelletier,Marcos Ferraro,Mário Mantovani,Alberto Fenocchio (2004). Genetic damage induced by trophic doses of lead in the neotropical fish Hoplias malabaricus (Characiformes, Erythrinidae) as revealed by the comet assay and chromosomal aberrations.
R Their,D Bonacker,T Stoiber,K Böhm,M Wang,E Unger,H Bolt,G Degen (2003). Interaction of metal salts with cytoskeletal motor protein systems.
Kelvin Davies (1995). Oxidative stress: the paradox of aerobic life.
Sue Rhee (2006). H <sub>2</sub> O <sub>2</sub> , a Necessary Evil for Cell Signaling.
D Monteiro,F Rantin,A Kalinin (2009). Inorganic mercury exposure: Toxicological effects, oxidative stress biomarkers and bioaccumulation in the tropical freshwater fish, matrinxã, Bryconamazonicus (Spix& Agassiz).
M Moore (1988). Cytochemical responses of the lysosomal system and NADPH-ferrihemoprotein reductase in molluscan digestive cells to environmental and experimental exposure to xenobiotics.
O Ribeiro,E Pelletier,W Pfeiffer,C Rouleau (2000). Comparative uptake, bioaccumulation, and gill damages of inorganic mercury in tropical and Nordic freshwater fish.
Athanasios Valavanidis,Thomais Vlahogianni,Manos Dassenakis,Michael Scoullos (2006). Molecular biomarkers of oxidative stress in aquatic organisms in relation to toxic environmental pollutants.
Ron Van Der Oost,Jonny Beyer,Nico Vermeulen (2003). Fish bioaccumulation and biomarkers in environmental risk assessment: a review.
G Rand,S Petrocelli (1985). Fundamentals of Aquatic Toxicology: Methods and Applications.
D Finney (1971). Probit Analysis. 3 rd Edn.
N Singh,M Mccoy,R Tice,E Schneider (1988). Simple technique for quantification of low levels of DNA damage in individual cells.
Yasuhisa Kono (1978). Generation of superoxide radical during autoxidation of hydroxylamine and an assay for superoxide dismutase.
Asru Sinha (1972). Colorimetric assay of catalase.
John Buege,Steven Aust (1978). [30] Microsomal lipid peroxidation.
J Buege,S Aust (1978). Microsomal lipid peroxidation.
S Colowick,N Kaplan Unknown Title.
M Moron,J Depierre,B Mannervik (1979). Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver.
Catarina Almeida,Catarina Pereira,Tânia Gomes,Cátia Cardoso,Maria Bebianno,Alexandra Cravo (2013). Genotoxicity in two bivalve species from a coastal lagoon in the south of Portugal.
B Burlinson,R Tice,G Speit,E Agurell,S Brendler-Schwaab,A Collins (2007). Invivo Comet Assay Workgroup, part of the Fourth International Workgroup on Genotoxicity Testing. Fourth International Workgroup on Genotoxicity Testing: Results of the In Vivo Comet Assay Workgroup.
T Capriglione,S De Iorio,F Gay,A Capaldo,M Vaccaro,M Morescalchi,V Laforgia (2011). Genotoxic effects of the fungicide thiophanate-methyl on Podarcis sicula assessed by micronucleus test, comet assay and chromosome analysis.
Paweł Jałoszyński,Maciej Kujawski,Maria Czub-Świerczek,Janina Markowska,Krzysztof Szyfter (1997). Bleomycin-induced DNA damage and its removal in lymphocytes of breast cancer patients studied by comet assay.
Vanessa De Andrade,Juliana Da Silva,Fernanda Da Silva,Vanina Heuser,Johnny Dias,Maria Yoneama,Thales De Freitas (2004). Fish as bioindicators to assess the effects of pollution in two southern Brazilian rivers using the Comet assay and micronucleus test.
Christina Emmanouil,Daniel Smart,Nikolas Hodges,J Chipman (2006). Oxidative damage produced by Cr(VI) and repair in mussel (Mytilus edulis L.) gill.
Deepmala Joshi,Mittal Kumar,Shakya Kumar,Shukla Sangeeta (2014). Reversal of Methylmercury-Induced Oxidative Stress, Lipid Peroxidation, and DNA Damage by the Treatment of N-Acetyl Cysteine: A Protective Approach.
S Pandey,S Parvez,I Sayeed,R Haque,B Binhafeez,S Raisuddin (2003). Biomarkers of oxidative stress: a comparative study of river Yamuna fish Wallago attu (Bl. & Schn.).
Francesco Regoli,Gary Winston,Stefania Gorbi,Giada Frenzilli,Marco Nigro,Ilaria Corsi,Silvano Focardi (2003). Integrating enzymatic responses to organic chemical exposure with total oxyradical absorbing capacity and dna damage in the european eel <i>Anguilla anguilla</i>.
E Padmini,M Rani (2009). Evaluation of oxidative stress biomarkers in hepatocytes of grey mullet inhabiting natural and polluted estuaries.
Dean Jones (2002). [11] Redox potential of GSH/GSSG couple: Assay and biological significance.
Yuanyuan Sun,Hongxia Yu,Jingfei Zhang,Ying Yin,Hua Shen,Hongling Liu,Xiaorong Wang (2006). Bioaccumulation and antioxidant responses in goldfish Carassius auratus under HC Orange No. 1 exposure.
Alton Meister,Mary Anderson (1983). GLUTATHIONE.
Bruce Shenker,Tai Guo,Irving Shapiro (2000). Mercury-Induced Apoptosis in Human Lymphoid Cells: Evidence That the Apoptotic Pathway Is Mercurial Species Dependent.
F Schurz,M Sabater-Vilar,J Fink-Gremmels (2000). Mutagenicity of mercury chloride and mechanisms of cellular defence: The role of metalbinding proteins.
Antonia Elia,Roberta Galarini,Maria Taticchi,Ambrosius Dörr,Luciana Mantilacci (2003). Antioxidant responses and bioaccumulation in Ictalurus melas under mercury exposure.
Shibani Chatterjee,Shelley Bhattacharya (1984). Detoxication of industrial pollutants by the glutathione glutathione-S-transferase system in the liver of Anabas testudineus (Bloch).
S Rana,R Singh,S Verma (1995). Mercury-induced lipid peroxidation in the liver, kidney, brain and gills of a fresh water fish Channa punctatus.
S Guilherme,M Válega,M Pereira,M Santos,M Pacheco (2008). Antioxidant and biotransformation responses in Liza aurata under environmental mercury exposure – Relationship with mercury accumulation and implications for public health.
Y Zheng,L Qiu,S Meng,L Fan,C Song,D Li,C Zhang,J Chen (2016). Effect of polychlorinated biphenyls on oxidation stress in the liver of juvenile GIFT, Oreochromis niloticus.
Federico Rubino (2015). Toxicity of Glutathione-Binding Metals: A Review of Targets and Mechanisms.
Samuel Peña-Llopis,M Ferrando,Juan Peña (2003). Fish tolerance to organophosphate-induced oxidative stress is dependent on the glutathione metabolism and enhanced by N-acetylcysteine.
Laurent Mosoni,Denis Breuillé,Caroline Buffière,Christiane Obled,Philippe Mirand (2004). Age-related changes in glutathione availability and skeletal muscle carbonyl content in healthy rats.
W Droge (2002). Free Radicals in the Physiological Control of Cell Function.
Antonio Ayala,Mario Muñoz,Sandro Argüelles (2014). Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondialdehyde and 4-Hydroxy-2-Nonenal.
Marc Berntssen,Aase Aatland,Richard Handy (2003). Chronic dietary mercury exposure causes oxidative stress, brain lesions, and altered behaviour in Atlantic salmon (Salmo salar) parr.
E Milaeva (2006). The role of radical reactions in organomercurials impact on lipid peroxidation.
Slavica Borković,Jelena Šaponjić,Sladjan Pavlović,Duško Blagojević,Slaviša Milošević,Tijana Kovačević,Ratko Radojičić,Mihajlo Spasić,Radoslav Žikić,Zorica Saičić (2005). The activity of antioxidant defence enzymes in the mussel Mytilus galloprovincialis from the Adriatic Sea.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Swetha, S.. 2026. \u201cEffects on DNA and antioxidant system of Anabas Testudineus on Experimental Exposure to a Sublethal Concentration of Methylmercury\u201d. Global Journal of Science Frontier Research - E: Marine Science GJSFR-E Volume 23 (GJSFR Volume 23 Issue E1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.