A thermodynamic model based on cluster of four atoms is considered to obtain conditional probability enumerating the higher order atomic correlation in the nearest neighbor shell of liquid binary alloys. This has enlightened the discussion of how the higher order atomic correlation is related to pairwise distribution. The Cd-Ga liquid alloy is a candidate of homo-coordination. Bi-Cd liquid alloy indicates partly homo-coordination. The higher order conditional probability of Bi-Cd binary alloy is uniformly displayed. There is band-like formation of the higher order conditional probability of Cd-Ga liquid alloy. The values of concentration-concentration fluctuation and higher order conditional probabilities computed for Bi-Cd and Cd-Ga, are presented.
## I. INTRODUCTION
The neutron diffraction experiment is always carried out to determine some structural details and information pertaining to some easily detectable thermodynamic properties of binary liquid alloys [1]. The exceptional cases might occur if the experimental data for some alloys is not available due to cumbersome tasks involved and experimental difficulties. For instance, the Short Range Order (SRO) [1] which has a connection with Concentration-Concentration Fluctuation in the long wavelength limit Scc(0) can be experimentally determined from the knowledge of concentration-concentration partial structural factor, $S_{\mathrm{cc}}(\mathbf{q})$, and the number-number partial structural factor $S_{\mathrm{NN}}(\mathbf{q})$ [2]. However, these structures are not easily measurable in most diffraction experiments. Hence SRO is usually computed without necessarily making reference to its experimental values. Additionally, a direct experimental determination of Scc (0) is often avoided due to the complexities involved. For this reason, the option of a thermodynamic model, which is readily used, was employed.
The thermodynamic model is commonly used to extract the macroscopic chemical structure of some thermodynamic systems. By considering a Quasi-chemical Model of the Four Atoms Cluster (say FACM),
with the view to obtain Higher Order Conditional Probabilities (HOCP) enumerating the atomic distribution in the nearest neighbor shell. (For example, i/iji, is the probability of finding $i$ atoms in the given lattice site. While other three neighboring sites in the cluster are occupied by atoms $i$, $j$, and $l$ atoms. This should give a more realistic of ordering energy (w) and the concentration-concentration fluctuation [Scc(0)] and thus a qualitative discussion is attempted for either hetero-coordination (i.e. preference for unlike atoms as to pair as nearest neighbors) or self-coordination (preference for like atoms as the nearest neighbors) in molten alloys [3].
In this study, ordering energy values were determined from Scc (0) [3] of which two alloys were considered. In the application of the thermodynamic model, the determined values of ordering energy, the Scc(0), and HOCP were recorded at different temperatures.
The calculations of the thermodynamic quantities at different temperatures involve getting the available experimental data and employing a suitable theoretical model. The alloys were preferred because the two alloys represent the classes of metal and semiconductor.
Investigations of liquid metallic alloys on the basis of the Quasi Chemical Model (QCM) and its usage for extracting values is also found in [3, 4] where this model was used for the calculations of some thermodynamic properties of the compound forming binary molten alloys at their melting temperatures[4].
In this observation, the QCM is applied to Bi-Cd and Cd-Ga liquid alloys for the qualitative investigation of their thermodynamic properties. Ordering energy values determined from Scc (0) are recorded in Table 1. The $C^{++}$ Computer programs were inscribed to generate data for thermodynamic expressions as functions of concentration, c, using ordering energy values, w, coordination number, Z, Boltzmann constant, K and temperature, T presented in Table 1.
Therefore, in the observation write up the attention is geared towards determining the ordering energy values of two binary alloys from values of deviations in $\mathrm{Scc}(0)$ [4] and calculating of some thermodynamic properties.
The determined values of ordering energy are displayed in Table 1.
Table 1: Ordering energy (w) in eV of Binary Liquid Alloys
<table><tr><td>Alloy</td><td>Temperature (°K)</td><td>Z</td><td>w (eV)</td></tr><tr><td>Bi-Cd</td><td>773</td><td>10.0</td><td>-0.0147</td></tr><tr><td>Cd-Ga</td><td>700</td><td>10.0</td><td>+0.1133</td></tr></table>
## II. EXPRESSION FOR CONCENTRATION-CONCENTRATION FLUCTUATION IN THE LONG WAVE LENGTH LIMIT (SCC (0))
$$
S _ {c c} (0) = \frac {S _ {c c} ^ {i d} (0)}{1 + \frac {Z}{2 \beta} (1 - \beta)} \tag {1}
$$
Where $S_{cc}^{id}(0)$ is the ideal concentration-concentration fluctuation in the long wavelength unit
$Z$ is the coordination number, and $\beta$ has an interwoven parameters such as w, $\eta,k_{B},T$ and c.
c is the concentration of atom i and 1-c is the concentration of atom j, w is ordering energy, $K_B$ is Boltzmann's constant, T is the melting temperature.
$$
\mathbf {S} _ {c c} ^ {i d} (0) = c (1 - c) \tag {2}
$$
$$
\eta = e ^ {\frac {w}{z k _ {T} T}} \tag {3}
$$
$$
\boldsymbol {\beta} ^ {2} = 1 - 4 c (1 - c) \left(\boldsymbol {\eta} ^ {2} - 1\right) \tag {4}
$$
Expression for Higher Order Conditional Probability (HOCP)
$$
\frac {B / A A A}{A / A A A} = a \sigma^ {z - 3} \eta^ {- 3} \tag {5}
$$
$$
\frac{B / AAB}{A / AAB} = a \sigma^{z - 3} \eta^{- 1} \tag{6}
$$
$$
\frac{B / ABB}{A / ABB} = a \sigma^{z - 3} \eta \tag{7}
$$
$$
\frac{B / BBB}{A / BBB} = a \sigma^{z - 3} \eta^{3} \tag{8}
$$
Where
$$
a = \left(\frac{1 - c}{c}\right) \sigma^{- z}
$$
$$
\sigma = \frac {\beta - 1 + 2 c}{2 \eta c} \tag {10}
$$
$$
\eta = e ^ {\frac{w}{z k _ {T} T}}
$$
$$
(\mathrm {A} / \mathrm {A A A}) + (\mathrm {B} / \mathrm {A A A}) = 1 \tag {12}
$$
$$
(\mathrm{A}/\mathrm{AAB}) + (\mathrm{B}/\mathrm{AAB}) = 1
$$
$$
(\mathrm{A}/\mathrm{ABB}) + (\mathrm{B}/\mathrm{ABB}) = 1
$$
$$
(\mathrm {A} / \mathrm {B B B}) + (\mathrm {B} / \mathrm {B B B}) = 1 \tag {15}
$$
$$
(A / AAA) = \frac{1}{1 + a \sigma^{(z - 3)} e^{-3\beta w / z}} \tag{16}
$$
$$
(A / AAA) = \frac{1}{1 + a \sigma^{(z - 3)} e^{-\beta w / z}} \tag{17}
$$
$$
(A / AAA) = \frac{1}{1 + a \sigma^{(z - 3)} e^{-\beta w / z}} \tag{18}
$$
$$
(A / AAA) = \frac{1}{1 + a \sigma^{(z - 3)} e^{-\beta w / z}} \tag{19}
$$
## III. RESULTS AND DISCUSSION
Table 1: Calculated concentration-concentration fluctuation of Bi-Cd alloy using Four Atoms Cluster Model (FACM). $C_{Bi}$ is the concentration of Bismuth in the alloy.
<table><tr><td>C<sub>Bi</sub></td><td>Scc(0)</td><td>Scc(0)id</td></tr><tr><td>0.1</td><td>0.089</td><td>0.09</td></tr><tr><td>0.2</td><td>0.166</td><td>0.16</td></tr><tr><td>0.3</td><td>0.250</td><td>0.21</td></tr><tr><td>0.4</td><td>0.296</td><td>0.24</td></tr><tr><td>0.5</td><td>0.296</td><td>0.25</td></tr><tr><td>0.6</td><td>0.256</td><td>0.24</td></tr><tr><td>0.7</td><td>0.284</td><td>0.21</td></tr><tr><td>0.8</td><td>0.156</td><td>0.16</td></tr><tr><td>0.9</td><td>0.065</td><td>0.09</td></tr></table>
<table><tr><td>CBI</td><td>Scc(0)</td><td>Scc(0)id</td></tr><tr><td>0.1</td><td>0.089</td><td>0.09</td></tr><tr><td>0.2</td><td>0.166</td><td>0.16</td></tr><tr><td>0.3</td><td>0.250</td><td>0.21</td></tr><tr><td>0.4</td><td>0.296</td><td>0.24</td></tr><tr><td>0.5</td><td>0.296</td><td>0.25</td></tr><tr><td>0.6</td><td>0.256</td><td>0.24</td></tr><tr><td>0.7</td><td>0.284</td><td>0.21</td></tr><tr><td>0.8</td><td>0.156</td><td>0.16</td></tr><tr><td>0.9</td><td>0.065</td><td>0.09</td></tr></table>
Table 2: Calculated concentration-concentration fluctuation of Cd-Ga alloy using Four Atoms Cluster Model (FACM). $C_{\mathrm{cd}}$ is the concentration of Cadmium in the alloy.
<table><tr><td>Ccd</td><td>Scc(0)</td><td>Scc(0)id</td></tr><tr><td>0.1</td><td>0.371</td><td>0.09</td></tr><tr><td>0.2</td><td>0.437</td><td>0.16</td></tr><tr><td>0.3</td><td>0.776</td><td>0.21</td></tr><tr><td>0.4</td><td>0.718</td><td>0.24</td></tr><tr><td>0.5</td><td>0.745</td><td>0.25</td></tr><tr><td>0.6</td><td>0.143</td><td>0.24</td></tr><tr><td>0.7</td><td>0.038</td><td>0.21</td></tr><tr><td>0.8</td><td>0.347</td><td>0.16</td></tr><tr><td>0.9</td><td>0.212</td><td>0.09</td></tr></table>
Table 3: Calculated Higher Order Conditional Probability of Bi-Cd alloy using Four Atoms Cluster Model (FACM). $C_{Bi}$ is the concentration of Bismuth in the alloy
<table><tr><td>C_Bi</td><td>A/AAA</td><td>A/AAB</td><td>A/ABB</td><td>A/BBB</td></tr><tr><td>0.1</td><td>0.0879</td><td>0.0922</td><td>0.0967</td><td>0.1014</td></tr><tr><td>0.2</td><td>0.1805</td><td>0.1884</td><td>0.1965</td><td>0.2050</td></tr><tr><td>0.3</td><td>0.2772</td><td>0.2879</td><td>0.2968</td><td>0.3099</td></tr><tr><td>0.4</td><td>0.3774</td><td>0.3899</td><td>0.4024</td><td>0.4151</td></tr><tr><td>0.5</td><td>0.4302</td><td>0.4934</td><td>0.5065</td><td>0.5197</td></tr><tr><td>0.6</td><td>0.5848</td><td>0.5975</td><td>0.6100</td><td>0.6225</td></tr><tr><td>0.7</td><td>0.6900</td><td>0.7011</td><td>0.7120</td><td>0.7227</td></tr><tr><td>0.8</td><td>0.7549</td><td>0.8034</td><td>0.8115</td><td>0.8194</td></tr><tr><td>0.9</td><td>0.8965</td><td>0.9032</td><td>0.9077</td><td>0.9121</td></tr></table>
Table 4: Calculated Higher Order Conditional Probability of Cd-Ga alloy using Four Atoms Cluster Model (FACM). $C_{cd}$ is the concentration of Cadmiun in the alloy
<table><tr><td>Ccd</td><td>A/AAAA</td><td>A/AAB</td><td>A/ABB</td><td>A/BBB</td></tr><tr><td>0.1</td><td>0.2403</td><td>0.1757</td><td>0.1256</td><td>0.0882</td></tr><tr><td>0.2</td><td>0.3838</td><td>0.2974</td><td>0.2219</td><td>0.1612</td></tr><tr><td>0.3</td><td>0.4898</td><td>0.3928</td><td>0.3036</td><td>0.2271</td></tr><tr><td>0.4</td><td>0.5726</td><td>0.4744</td><td>0.3782</td><td>0.2907</td></tr><tr><td>0.5</td><td>0.6438</td><td>0.5491</td><td>0.4508</td><td>0.3561</td></tr><tr><td>0.6</td><td>0.7092</td><td>0.6217</td><td>0.5255</td><td>0.4273</td></tr><tr><td>0.7</td><td>0.7728</td><td>0.6963</td><td>0.6071</td><td>0.5101</td></tr><tr><td>0.8</td><td>0.8387</td><td>0.7790</td><td>0.7025</td><td>0.6141</td></tr><tr><td>0.9</td><td>0.9117</td><td>0.8943</td><td>0.8242</td><td>0.7537</td></tr></table>
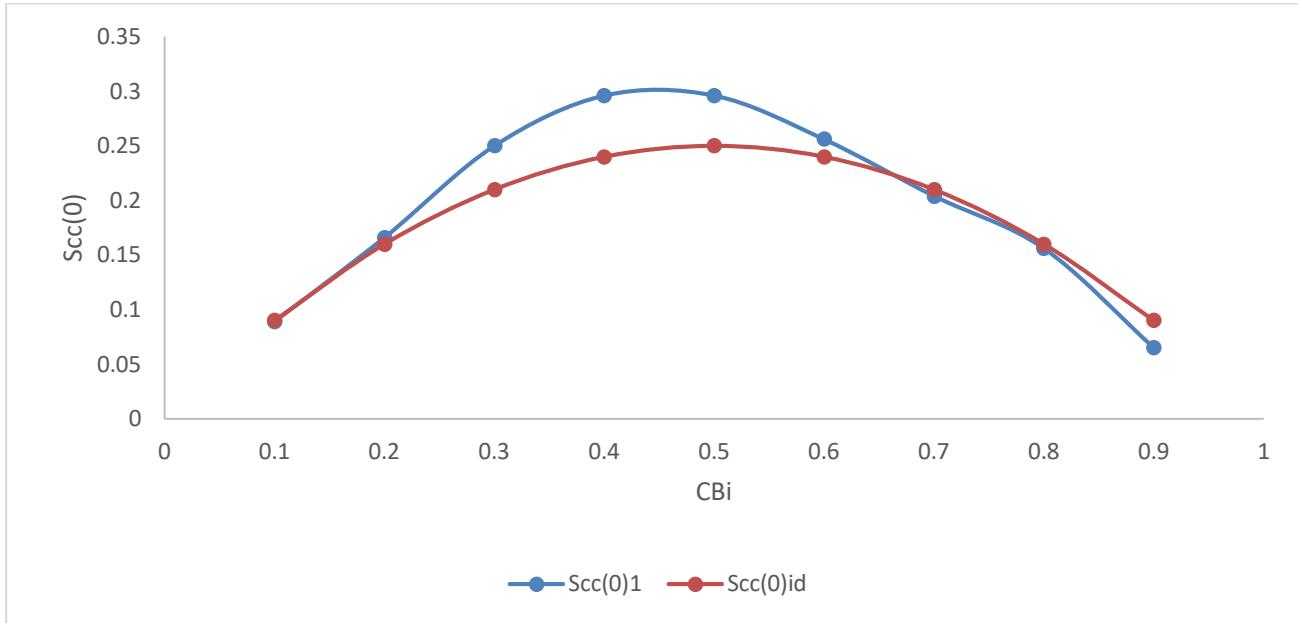
Fig. 1: Concentration-concentration fluctuation Scc(0) versus concentration $C_{\mathrm{Bi}}$
In Figure 1, the values of $\mathrm{Scc}(0)$ for Bi-Cd liquid alloy portray homocoordination at some concentrations $C_{\mathrm{Bi}} = 0.3$, 0.4, 0.5 and 0.6. At the remaining concentrations, their exits perfect agreement between the ideal solution and the $\mathrm{Scc}(0)$ of Bi-Cd liquid binary alloy. In the alloy at elevated temperature same atom are bound to pair-up with each other in the nearest
neighbourhood at some specific composition of bismuth. The Scc(0) of these alloy increases initially to a maximum (owing to the charge transfer between neighboring atoms) within the entire concentration range [5, 6]. This is due to chemical alternation of positive and negative charges with length scale approximately twice the nearest neighbor distance.
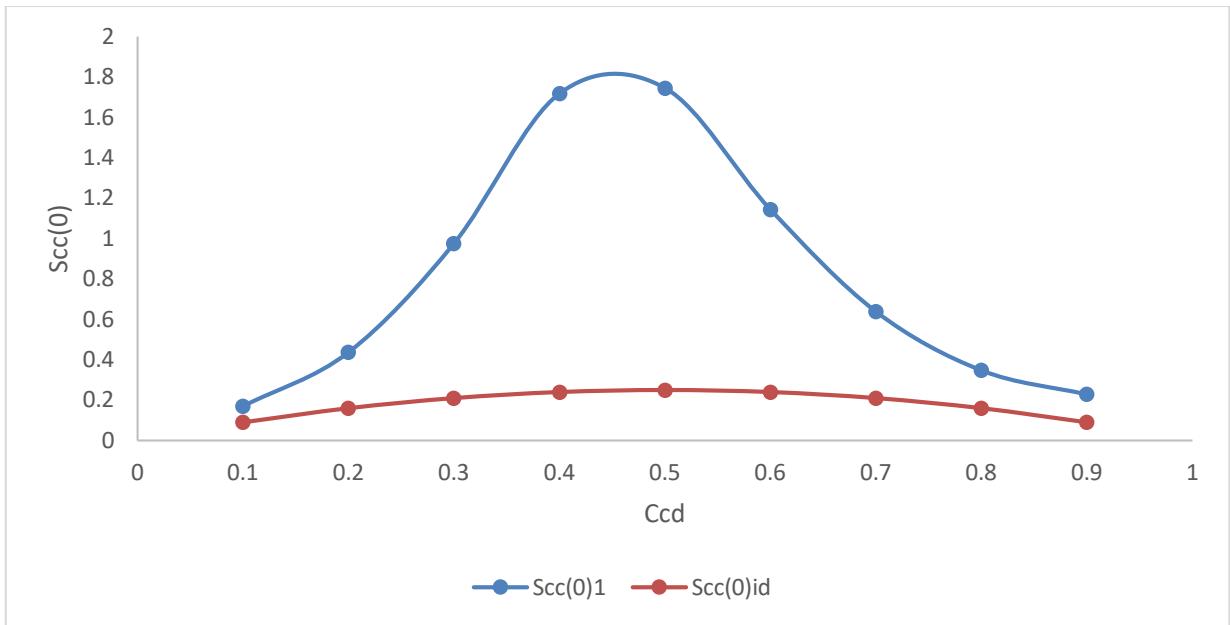
Fig. 2: Concentration-concentration fluctuation Scc(0) versus concentration $C_{\mathrm{cd}}$
In Figure 2, the values of $\mathrm{Scc}(0)$ for Cd-Ga liquid alloy signifies homocoordination throughout the entire composition of Cadmium. The values of $\mathrm{Scc}(0)$ of Cd-Ga liquid binary alloy are higher than the ideal case representing the complete homocoordination in the
nearest neighbour shell. In the alloy at elevated temperatures, the same atoms are bound to pair up with each other in the nearest neighborhood at the entire concentration of cadmium. This is possibly due to the disordered potential which too large [7].
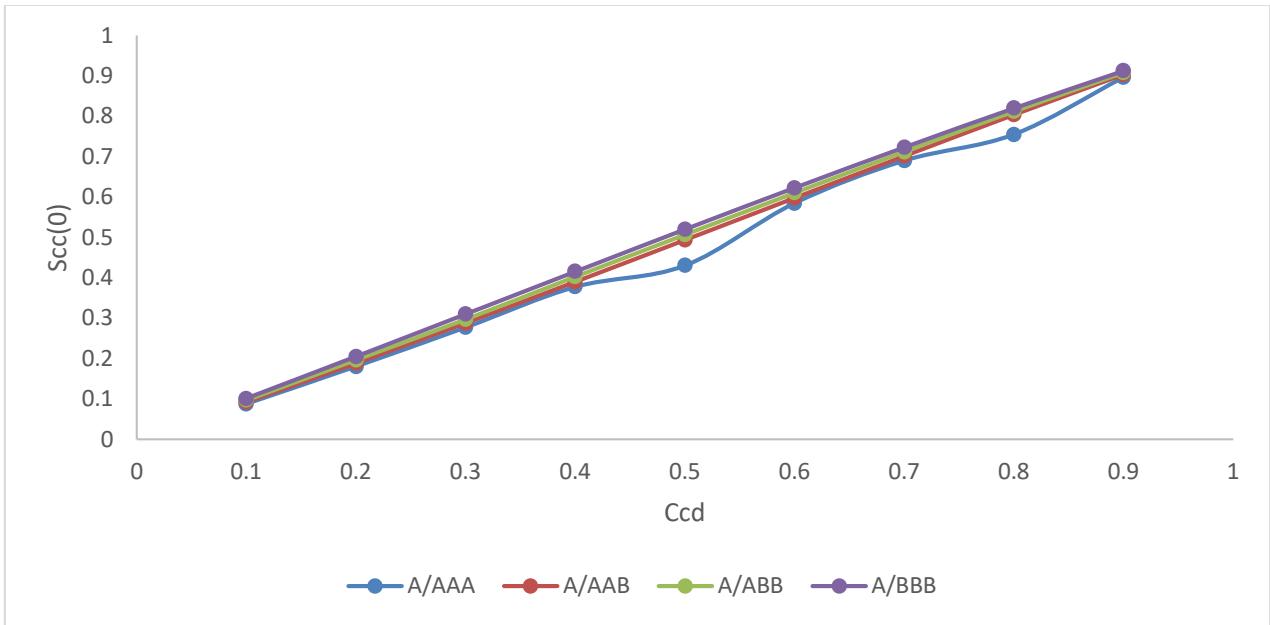
Fig. 3: Higher Order Conditional Probability (HOCP) versus concentration $C_{\mathrm{Bi}}$
The three probabilities (A/AAB, A/ABB and A/BBB) have formed unified paths undeviated at higher order cluster of four atoms irrespective of the possibilities. Only one of the possibilities has slight deviations at concentrations $C_{\mathrm{Bi}} = 0.5$ and 0.8. The three possibilities agree with the one another on the distributions of a particular atom as reference atom having either the same or and different atom as the nearest neighbour shell in the cluster. Because of the
topological short range order at $\mathrm{Ccd} = 0.5$ and 0.8. i.e. geometrical arrangements of atoms) it indicates and supports homocoordination (Preference of like atoms as the nearest neighbor) [8].
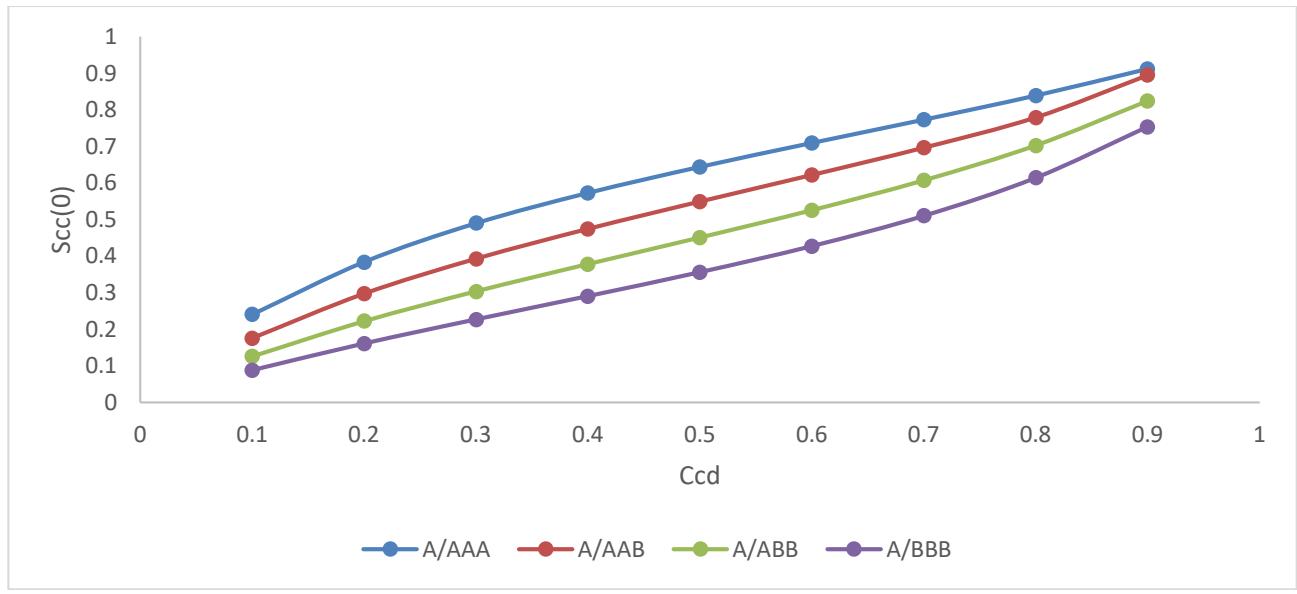
Fig. 4: Higher Order Conditional Probability (HOCP) versus concentration $C_{\mathrm{cd}}$
The four possibilities of higher order conditional probability (i.e A/AAA, A/AAB, A/ABB and A/BBB) are not uniform because there is a near cancellation of the ionic potentials while at large distances ionic potentials was screened in Figure 4. In addition, in this perspective it has calculated values for HOCP of Cd-Ga been spaced for the four possibilities which are also in support of homocoordination or self-coordination. There are other reasons of directional bonding, which are also responsible for slight curve of the plot of HOCP of Cd-Ga binary liquid alloy [9, 10].
Generating HTML Viewer...
References
10 Cites in Article
R Singh (1987). Short-range order and concentration fluctuations in binary molten alloys.
A Bhatia,W Hargrove (1974). Concentration fluctuations and thermodynamic properties of some compound forming binary molten systems.
S Lele,P Ramachandrarao (1981). Estimation of complex concentration in a regular associated solution.
O Awe,I Akinwale,J Imeh,J Out (2009). Calculation of experimental concentrationconcentration fluctuations of liquid binary alloys using free energy of mixing and experimental activities.
D Adhikari,B Singh,I Jha,B Singh (2010). Thermodynamic Properties and Microscopic Structure of Liquid Cd-Na Alloys by Estimating Complex Concentration in a Regular Associated Solution.
P Singh,K Khanna (1987). Entropy of mixing calculations for compound forming liquid alloys in the hard sphere system.
Natalia Kotova,Natalia Usenko,Natalia Golovata (2018). Thermochemical properties and glass forming tendency modeling for the ternary Ge–Mn–Gd melts.
K Khanna,C Pal,P Singh (1987). Volume of Mixing in Binary Liquid Alloys.
P Singh,K Khanna (1984). Entropy of mixing calculations for compound forming liquid alloys in the hard sphere system.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
O.W Abodunrin. 2026. \u201cHigher Order Conditional Probability and Concentration-Concentration Fluctuation in the Long Wavelength Limit of Molten Binary Alloys\u201d. Global Journal of Science Frontier Research - A: Physics & Space Science GJSFR-A Volume 22 (GJSFR Volume 22 Issue A4): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
A thermodynamic model based on cluster of four atoms is considered to obtain conditional probability enumerating the higher order atomic correlation in the nearest neighbor shell of liquid binary alloys. This has enlightened the discussion of how the higher order atomic correlation is related to pairwise distribution. The Cd-Ga liquid alloy is a candidate of homo-coordination. Bi-Cd liquid alloy indicates partly homo-coordination. The higher order conditional probability of Bi-Cd binary alloy is uniformly displayed. There is band-like formation of the higher order conditional probability of Cd-Ga liquid alloy. The values of concentration-concentration fluctuation and higher order conditional probabilities computed for Bi-Cd and Cd-Ga, are presented.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.