From the introduction of human recombinant insulin preparations, insulin allergy has become rare, with a reported prevalence of approximately 2.4% (1). Most insulin injection reactions are immediate and IgE-mediated and can be classified as either Type I or Type IV hypersensitivity (1).
## I. INTRODUCTION
From the introduction of human recombinant insulin preparations, insulin allergy has become rare, with a reported prevalence of approximately $2.4\%$ (1). Insulin injection reactions can be classified as Type I or Type IV. Most reactions are immediate and mediated by IgE (1). Clinically, these reactions may manifest as swelling at the injection site, erythema, urticaria, angioedema, rhinitis, bronchospasm, or, in severe cases, anaphylaxis. (1)
Managing patients with insulin allergies, particularly those who are insulin-dependent poses significant challenges in achieving successful glycemic control. In this case report, we describe a 32-year-old female who developed an immediate allergic reaction to a premixed analog insulin preparation containing Aspart and Degludec.
## II. CASE REPORT
A 32-year-old female with latent autoimmune diabetes in adults (LADA) was treated with multiple oral antidiabetic drugs in combination with four daily injections of recombinant human insulin. This regimen included one injection of intermediate-acting insulin at bedtime and three premeal injections of rapid-acting insulin. Despite dose adjustments based on her blood sugar levels and food intake, the patient experienced recurrent episodes of fluctuating blood glucose levels.
As a result, her treatment was modified to three injections: one premixed analog insulin injection before dinner and two injections of rapid-acting recombinant human insulin before breakfast and lunch. This adjustment successfully stabilized her blood glucose levels. However, the patient began experiencing rash and itching at the injection site 20-30 minutes after administering the dinner insulin dose. Figure 1 shows the rash that developed after injecting the night dose of insulin. Seeking dermatological consultation, she was prescribed antihistamines and steroids, which alleviated her symptoms. Other than the localized rash and itching, she had no systemic symptoms and no history of allergies.
The patient noted that these episodes consistently followed her evening insulin dose, prompting her to revisit the clinic with concern. To investigate a possible allergic reaction, a skin prick test was conducted separately with Aspart and Deglude. The test yielded an immediate positive result for Degludec, Figure 2 shows the rash that developed immediately after injecting Degludec during the skin prick test, confirming the diagnosis of Degludec-induced hypersensitivity. Figure 3 shows no rash at the site of the skin prick with insulin aspart.
Consequently, the patient was transitioned back to her prior basal-bolus insulin regimen. However, this led to spikes in her blood glucose levels. To optimize glycemic control, she was switched to three injections of rapid-acting insulin combined with a long-acting insulin (300 U Glargine). Given the synthetic nature of Glargine, there was a potential risk of an allergic reaction. Therefore, a skin prick test was performed before initiation, which showed no reactivity. The patient was subsequently started on Glargine along with rapid-acting insulin, achieving stable blood glucose levels without further hypersensitivity reactions.
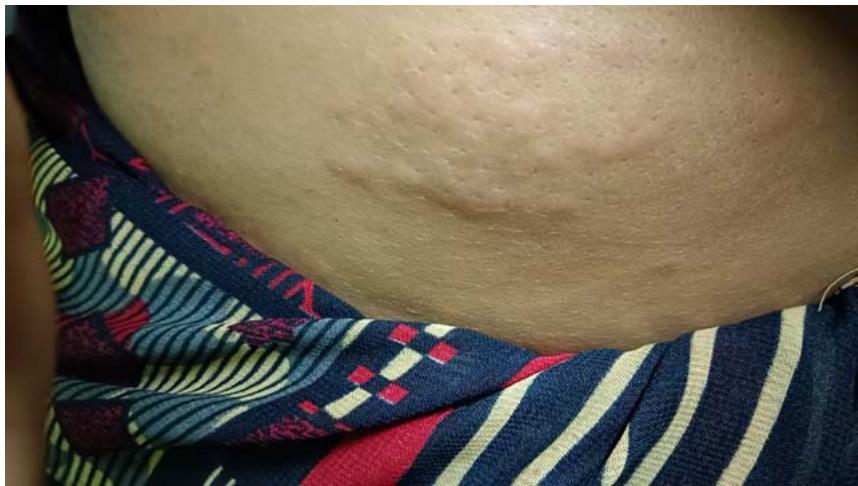 Fig. 1 Rash at the Site of Insulin Injection
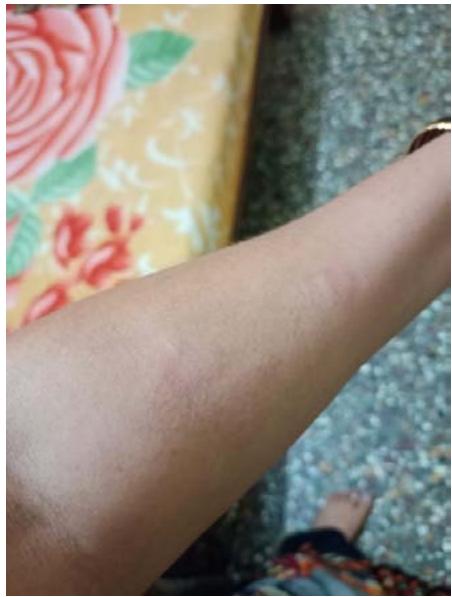 Fig 2: Skin Prick Test Showing Rash at the Site of Degludec Injection
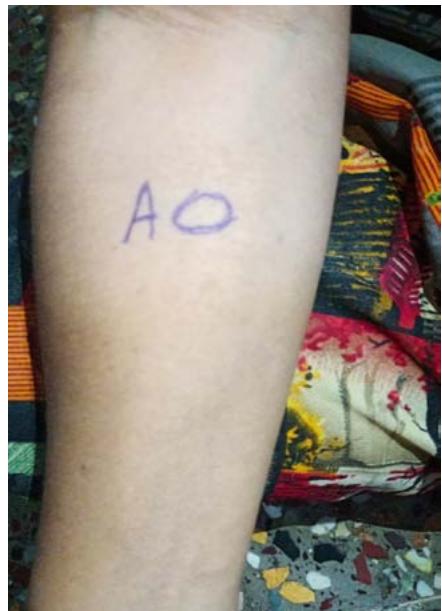 Fig 3: Skin Prick Test Was Not Showing Any Rash at the Site of Aspart Injection
## III. DISCUSSION
The availability of recombinant human insulin and its analogs has revolutionized the treatment of diabetes, substantially decreasing immunogenicity that is linked to traditional animal-derived insulins. However, hypersensitivity remains a clinical challenge, particularly in insulin-dependent patients. This case report presents a rare instance of hypersensitivity caused by insulin degludec in a patient with latent autoimmune diabetes in adults (LADA).
## IV. TRIGERRS AND PATHOGENESIS
Hypersensitivity reactions to insulin can be classified into two major categories: Type I and Type IV. Type I reactions are immediate and mediated by IgE, typically involving mast cells. Symptoms of Type I reactions include urticaria, pruritus, angioedema, and, in severe cases, systemic anaphylaxis. In contrast, Type IV reactions are delayed and T-cell-mediated. These reactions develop more gradually, usually presenting as localized erythema or induration, appearing several hours to days after insulin injection. (2)(3).
Type I hypersensitivity reaction is well demonstrated in this case by the rapid onset of symptoms (particularly, a rash and itching within 20 to 30 minutes of the injection) and positive results of the skin prick test for degluc. This type of hypersensitivity can be caused by the insulin molecule itself or additives like zinc, protamine, or metacresol. Deglucc contains additives like metacresol and zinc, two common allergens that can cause allergic reactions (4).
## V. DIAGNOSTIC APPROACH
The diagnosis of insulin hypersensitivity requires a detailed evaluation. The history and physical examination should be detailed to identify potential triggers, such as latex contamination of syringes or concurrent allergens. When diagnosing allergic reactions, skin tests-skin prick and intradermal tests-are the most appropriate methods (1). In this case, a skin prick test confirmed the immediate allergy to degludec. Measuring serum levels of IgE that are specific to insulin or its parts may also assist in making a diagnosis, but their practical use is limited (4).
## VI. MANAGEMENT APPROACH
"Managing insulin hypersensitivity in insulin-dependent patients is particularly challenging, as discontinuation of insulin therapy is not an option. The primary objective is to maintain glycemic control while minimizing allergic reactions."
### 1. Switching Insulin Formulations
Rapid-acting insulin and glargine (300 U) were added to our patient's basal-bolus insulin regimen. Before starting glargine, a skin prick test was conducted to ensure its safety. Hypersensitivity is frequently resolved by switching insulin formulations with different excipients or by utilizing a different administration method (3).
#### 2. Desensitization Protocols
Desensitization involves administering increasing dosages of the inciting insulin to produce immune tolerance. Although it works well in resistant cases, it is laborious and needs to be carried out under strict supervision in a hospital setting (1)(5).
#### 3. Adjunctive Therapies
Acute allergic reactions can be alleviated by treating symptoms with corticosteroids and antihistamines. (1)(3). In our patient antihistamines adequately controlled the symptoms during hypersensitivity episodes.
#### 4. Alternatives
Non-insulin therapies such as SGLT2 inhibitors or GLP-1 receptor agonists may reduce insulin needs and, hence, the risk of hypersensitivity reactions. Sometimes, hypersensitivity reactions may even be prevented by Continuous Subcutaneous Insulin Infusion (CSII) using an insulin pump (6). For patients with severe, recurrent allergic reactions, omalizumab can be considered, particularly after reducing IgE levels with B-cell-depleting monoclonal antibody Rituximab. When insulin allergies are more severe and unresponsive to these treatment options, the remaining therapeutic options are islet cell transplantation and pancreatic transplantation (1). Figure 4, gives a rough idea about evaluation of insulin allergy and its treatment (1).
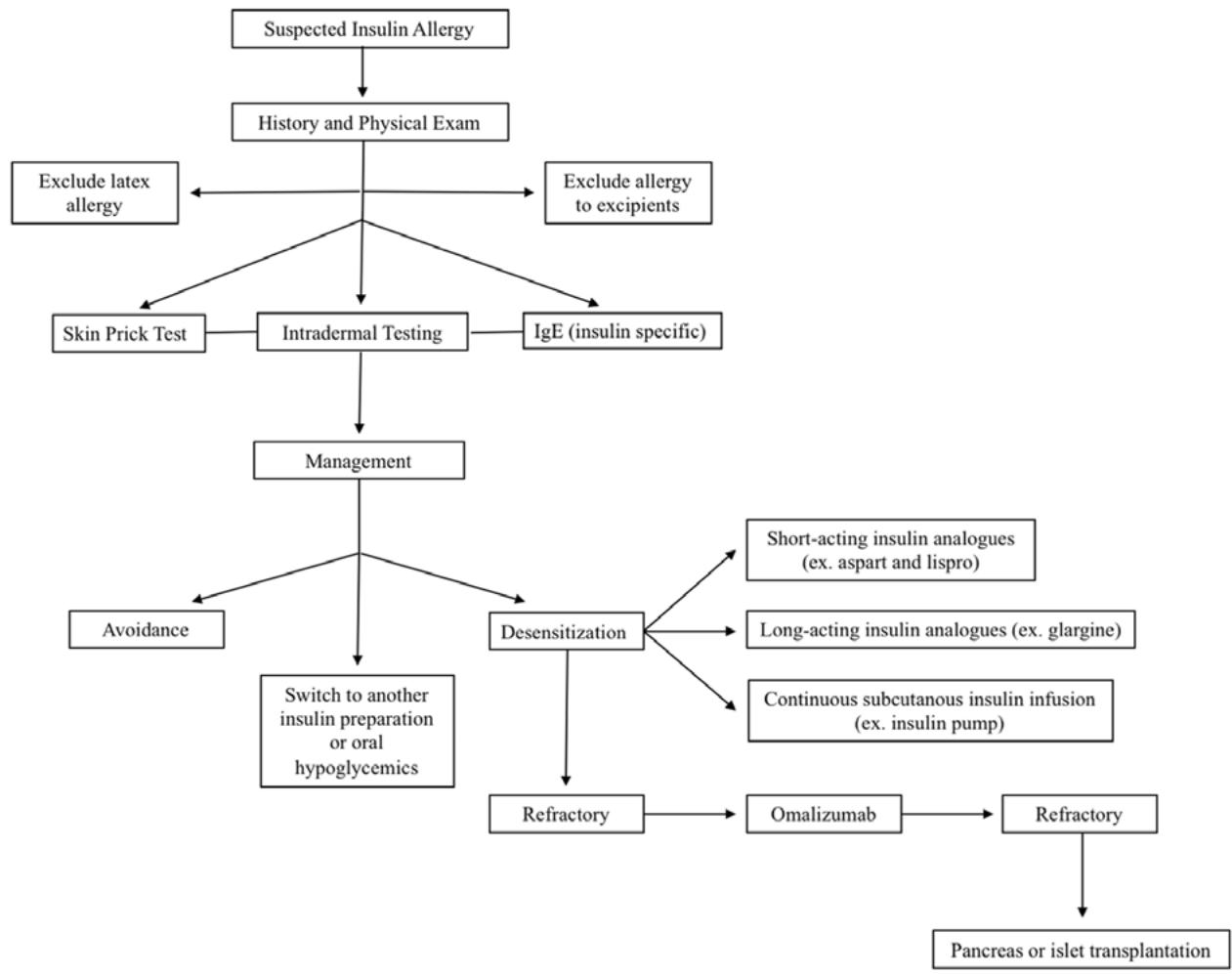 Fig 4: Flow Chart Showing the Evaluation and Treatment of Insulin Allergy (Sourced From Reference 1)
## VII. CONCLUSION
This case underscores the importance of promptly recognizing and managing hypersensitivity reactions in insulin-treated patients. Although rare, such reactions can significantly perturb glycemic control and impair quality of life. A structured diagnostic approach that includes a thorough investigation of potential allergens, confirmatory skin tests, knowledge of alternative insulin formulations and adjunctive therapies will help clinician handle these challenges effectively.
### Future Perspectives:
Further research is essential to deepen our understanding of the immunological mechanisms underlying insulin hypersensitivity, paving the way for the development of more targeted and specific insulin formulations. Investigating genetic predispositions, particularly those associated with human leukocyte antigen (HLA) variants, may offer valuable insights into individual susceptibility to hypersensitivity reactions. Additionally, advancements in the design of hypoallergenic insulin formulations-through techniques such as protein engineering or alternative delivery systems-hold significant promise for reducing the incidence of adverse reactions. Such progress could lead to improved clinical outcomes, enhanced patient adherence, and a better quality of life for those affected by insulin hypersensitivity.
Generating HTML Viewer...
References
4 Cites in Article
Babak Aberumand,Samira Jeimy (2021). The complexities of insulin allergy: a case and approach.
Lucie Heinzerling,Adriano Mari,Karl‐christian Bergmann,Megon Bresciani,Guido Burbach,Ulf Darsow,Stephen Durham,Wytske Fokkens,Mark Gjomarkaj,Tari Haahtela,Ana Bom,Stefan Wöhrl,Howard Maibach,Richard Lockey (2013). The skin prick test – European standards.
L Heinzerling,K Raile,H Rochlitz,T Zuberbier,M Worm (2008). Insulin allergy: clinical manifestations and management strategies.
C Grieco,D Swain,S Colberg,E Dowling,K Baskette,L Zarrabi (2013). Effect of intensity of aerobic training on insulin sensitivity/resistance in recreationally active adults.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Kavya. Jonnalagadda. 2026. \u201cHypersensitivity to Insulin Degludec: Case Analysis and Management Perspectives\u201d. Global Journal of Medical Research - K: Interdisciplinary GJMR-K Volume 25 (GJMR Volume 25 Issue K1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
From the introduction of human recombinant insulin preparations, insulin allergy has become rare, with a reported prevalence of approximately 2.4% (1). Most insulin injection reactions are immediate and IgE-mediated and can be classified as either Type I or Type IV hypersensitivity (1).
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.