High-resolution infrared absorption spectra of the H2CO•••HF complex and its monomers are recorded in a gas phase in C=O and C-H regions using Bruker HR 125 spectrometer recorded at 0.05 cm-1 resolution. After complex formation about the HF stretching band complicated shapes are not observed due to weak interaction couplings between C=O and C-H stretching of the H2CO•••HF complex. Also, quantum chemical calculations are performed in mp2//6311++g(3df, 3pd) approximation, and energetic, geometrical, and harmonic spectral parameters have been calculated.
## I. INTRODUCTION
Hydrogen bond (HB) is vital in many physical, chemical, and biological systems in life, and hydrogen fluoride (HF) is one of the strong proton donor molecule which is worthy of investigation nature of (HB). In [1] v(HF) stretching band formation mechanisms of B...HF complexes were investigated both experimentally and theoretically in detail. Recently, computational efforts performed on the relevant complexes [2]. In [3] for $(\mathrm{H}_2\mathrm{CO})_2\bullet \bullet \bullet \mathrm{HF}$ and $\mathrm{H}_2\mathrm{CO}\bullet \bullet \bullet (\mathrm{HF})_2$ trimers accurate spectral parameters were presented through a very high level of quantum chemical calculations. Optimal geometry, harmonic spectral parameters and energetic evaluations of the $\mathrm{H}_2\mathrm{CO}\dots \mathrm{HF}$ complexes were shown in [4]. This $(\mathrm{CH}_2\mathrm{O})_2$ complex has been investigated both experimentally and theoretically by THz spectroscopic and CCSD(T)-F12/aug-cc-pV5Z and MP2/aug-cc-pVQZ quantum chemical calculations [5]. In this work dissociation energy of the dimer is estimated as $D_0 = 13.7\pm 0.3$ kJ·mol $^{-1}$ for the global potential energy minimum.
Anharmonic calculations in mp2/6-311++G(3df,3pd) approximation and low temperature matrix-isolation experimental studies were performed in [6]. For the $\mathrm{CH}_3\mathrm{CN}\cdots \mathrm{HF}$ complex, both experimental and computational results have showed in our paper [7]. In [8], spectral and geometrical parameters of the $[\mathsf{F}(\mathsf{HF})_2]$ and $[\mathsf{F}(\mathsf{DF})_2]$ -complexes were performed by using the mp2 theory and multidimensional variation methods. Anharmonic calculations are play an essential role in the investigation of hydrogen bonded molecular complexes due to well agreement with the experimental results. Recently, numerous works have been devoted to solving anharmonic multidimensional problems with the help of Schrödinger equations with the variational methods [9-11]. In [12] authors utilized fully automated code for the establishing of interatomic force constants for the identification ro-vibrational spectral parameters for several HB molecular complexes. The results in the report are significantly important to analyze internal dynamics, to recognize spectral manifestation of molecular complexes, and to distinguishing overlapping spectral lines of noncovalent bonded molecular complexes.
In this work, $\mathrm{H}_2\mathrm{CO}\cdots\mathrm{HF}$ complex and monomers are studied with the help FTIR spectroscopy in the gas phase and quantum chemical calculations. Obtained results are coincided with relevant literature.
## II. EXPERIMENTAL AND COMPUTATIONAL METHODS
### a) Experimental Methods
The high-resolution absorption spectra of the $\mathrm{H}_2\mathrm{CO}\cdots\mathrm{HF}$ complex and $\mathrm{H}_2\mathrm{CO}$ monomer were recorded with the help Bruker 125 HR spectrometer at the resolution of $0.05\mathrm{cm}^{-1}$ in the gas phase. Formaldehyde molecule transferred from the powder to the gas phase by the phase changer vacuum equipment. The vacuum atmosphere has created inside of cavity cell, and the samples were placed into stainless steel cavity according to the real gas lows. The stainless-steel cavity was $20~\mathrm{cm}$ in length with sapphire and ZnSe windows. Total pressure of the mixture was about 20-100 Torr.
### b) Computational Methods
Quantum chemical calculations are carried out by the help of the latest version of GAUSSIAN 16 software [15] with mp2/6-311++G(3df,3pd) approximation. Equilibrium geometries, interaction energies, and various harmonic spectral parameters of the complex have been determined.
## III. RESULTS AND DISCUSSION
### a) Experimental Part
Gas phase vibrational spectra of molecular complexes yield adequate information about the rotational dynamics and complicated intermolecular interactions. Recently, in [6] the $\mathsf{H}_2\mathsf{CO}\cdots\mathsf{HF}$ complex and its monomers have been studied in the matrix with the help of FTIR spectroscopy. Infrared spectra in matrix in the $C = O$ and $C - H$ regions were analyzed in detail. Within $2v_{4}$ and $v_{3}$ regions a new spectral features have observed.
Fig.1. shows $\nu (C = 0)$ stretching region of the $\mathrm{H}_2\mathrm{CO}\dots \mathrm{HF}$ complex and $\mathrm{H}_2\mathrm{CO}$ molecule. In the figure,
R, Q, and P branches observed. The yellow spectrum belongs to the pure $\mathrm{H}_2\mathrm{CO}$ molecule at pressure $p = 0.006\mathrm{kgf/cm^2}$ (c), the green spectrum belongs to the complex recorded at $p = 90$ Torr (b) and blue is belongs to the complex at 65 Torr (a), for all three spectra recorded at a resolution of $0.05\mathrm{cm^{-1}}$. Even through the fact that there were no observations of spectral changes upon the complex formation in the band, the intensity of lines increased when the pressure increased from 65 to 95 Torr. The line parameters of the band were in good agreement with HITRAN [13] database.
Intensities of the transitions can be determined through the following formula
$$
S = 2. 5 0 6 4 3 \nu \left[ \left\langle i \left| \mu_ {x} \right| f \right\rangle^ {2} + \left\langle i \left| \mu_ {y} \right| f \right\rangle^ {2} \left\langle i \left| \mu_ {z} \right| f \right\rangle^ {2} \right], \tag {1}
$$
Where, S is the absorption intensity of the lines (in km/mol), i-initial state, f-final state, \\nu-is the transition frequency (in cm^{-1}), \\langle i|\\mu_x|f\\rangle^2, \\langle i|\\mu_y|f\\rangle^2, \\langle i|\\mu_z|f\\rangle^2 - the dipole moment components (in D).
Herein the line intensities of the bands increasing when upgrade the pressure from 65 to 95 Torr, respectively.
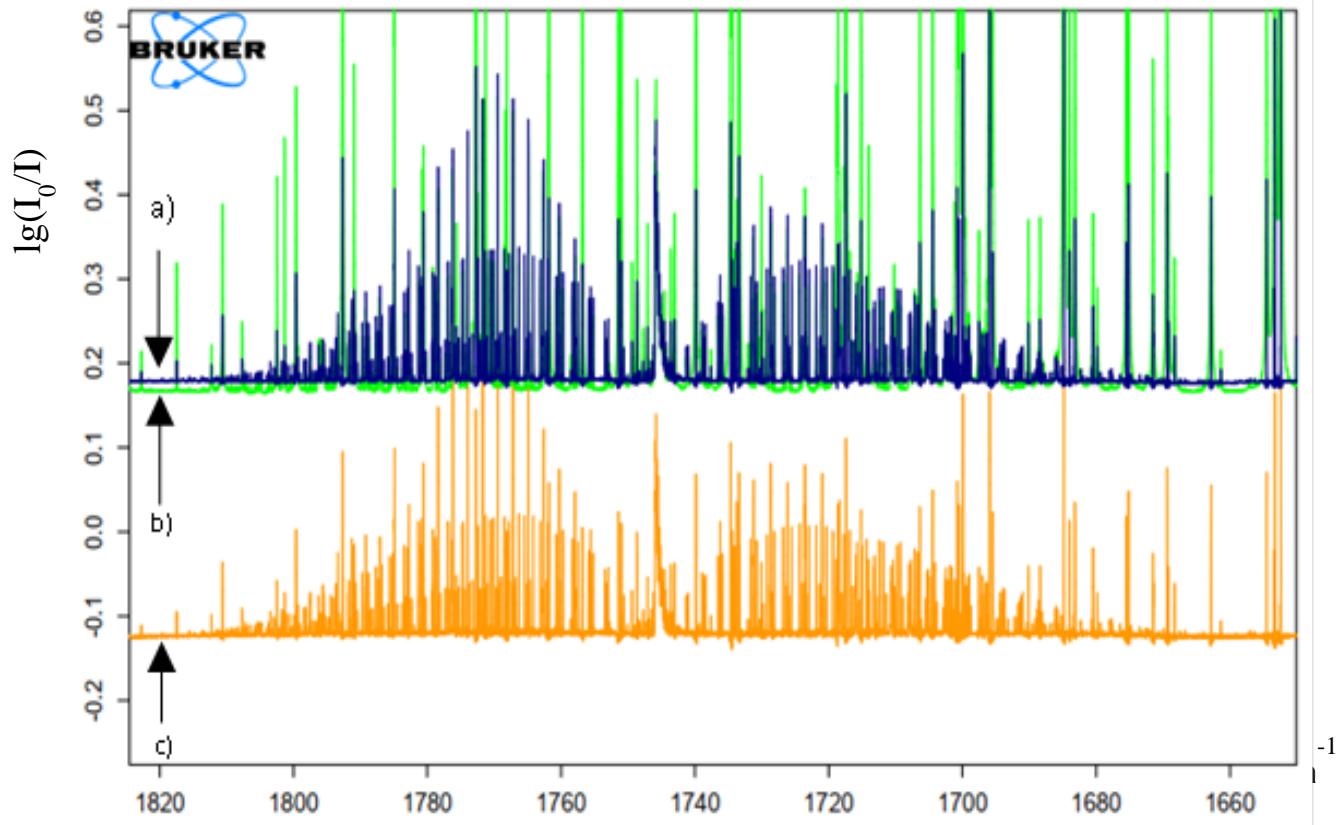 Figure 1: Absorption spectra of pure H2CO and HF+ H2CO complex in gas phase in the v(C[]O) stretching region. (yellow, pure H2CO p=0.006 kgf/cm2, green, HF+ H2CO complex p=90 Torr and blue, HF+ H2CO complex 65 Torr, resolution 0.05 cm-1)
All the spectral features related to the H-F, C=O, and C-H stretches and the HF librational vibrations in the $\mathrm{H}_2\mathrm{CO}\cdots\mathrm{HF}$ complex.
In contrast with studied complexes in [1], in this work, the v(HF) region is complicated spectral features are not observed upon the complex formation. Due to following reasons, complicated spectral patterns have not observed: firstly, C=O and HF stretching vibrations are weakly interacting mutually in the complex. Secondly, the mechanical anharmonic constant of the HF molecule is sufficiently large and equals $85~\mathrm{cm}^{-1}$. Thirdly, the $\mathsf{H}_2\mathsf{CO}$ molecule keeps the inclination to polymerization even in the case of low concentrations.
Therefore, we are not considering the focus $v(HF)$ region of the complex. It requires further experimental investigations at a convenient condition and with the help of unique spectroscopic techniques.
Fig. 2. shows $v(C - H)$ stretching region of the $\mathsf{H}_2\mathsf{CO}\dots \mathsf{HF}$ complex and $\mathsf{H}_2\mathsf{CO}$ molecule. In the spectrum, R, Q, and P branches have observed, respectively. The yellow spectrum (c) belongs to the pure $\mathsf{H}_2\mathsf{CO}$ molecule at pressure $p = 0.006~\mathrm{kgf/cm^2}$, and green (b) belongs to the complex which is recorded at $p = 90$ Torr and at blue (a) belongs to the complex $p = 65$ Torr, respectively. All spectra were recorded at the $0.05~\mathrm{cm^{-1}}$ spectral resolution.
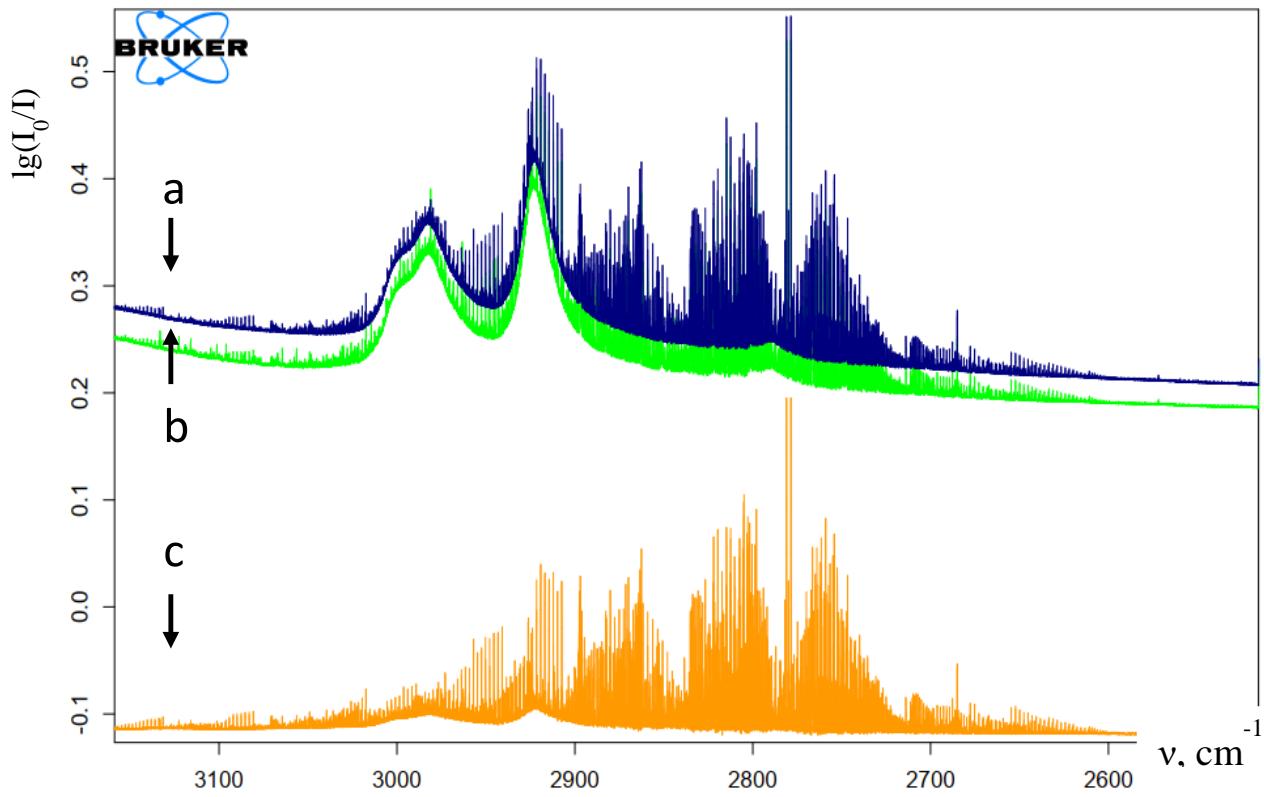 Figure 2: Infrared absorption spectra of pure $\mathsf{H}_2\mathsf{CO}$ and $\mathsf{HF} + \mathsf{H}_2\mathsf{CO}$ complex in the gas phase in the v(CH) stretching region. (yellow-(c), pure $\mathsf{H}_2\mathsf{CO}$ $p = 0.006\mathrm{kgf / cm^2}$, green (b) $\mathsf{HF} + \mathsf{H}_2\mathsf{CO}$ complex $p = 90$ Torr and blue (a), $\mathsf{HF} + \mathsf{H}_2\mathsf{CO}$ complex 65 Torr)
### b) Computational Part
Quantum chemical calculations on the $\mathrm{H}_2\mathrm{CO}\cdots\mathrm{HF}$ complex have been carried out with using the GAUSSIAN 16 software in the mp2/6-311++G(3df,3pd) approximation with the basis set superposition error taken into account. This approximation provides accurate information for the spectral parameters of the complex. The band changes (blue or red), of the frequency, and intensity changes have been explained in [4] in detail. The equilibrium geometry of the $\mathrm{H}_2\mathrm{CO}\cdots\mathrm{HF}$ complex is presented in Fig.3. Selected geometrical parameters of the complex have shown in table 1.
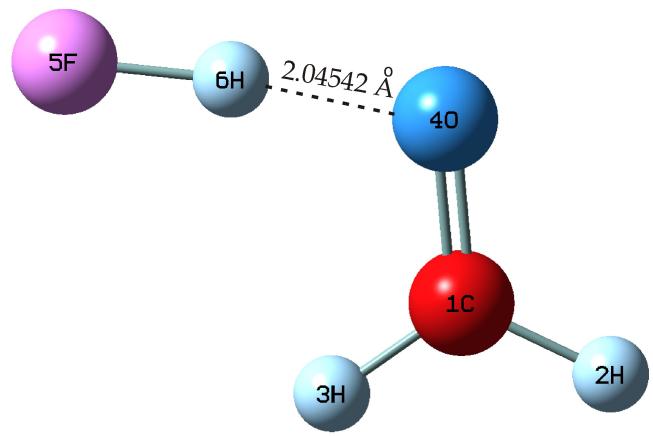 Figure 3: The Equilibrium Geometry of $\mathsf{H}_2\mathsf{CO}\dots \mathsf{HF}$ Complex Calculated at mp2/6-311++g(3df,3pd) Approximation
Upon the formation of the complex, H-F stretching frequency shortened by $9.89~\mathrm{cm}^{-1}$ and the $\mathbf{C} = \mathbf{O}$ stretching band of formaldehyde also shortened about $9.7~\mathrm{cm}^{-1}$.
The C-H stretching frequency is blue shifted by about $39~\mathrm{cm}^{-1}$, and the atomic distance between C and O is increased. The H-F frequency is equal $\nu_{12}$ (3546 cm $^{-1}$ ) according to the computational results and this value equals to $3573~\mathrm{cm}^{-1}$ which is calculated in experiment [14]. The frequency of HF molecule according to experimental investigation equals to $3961.40~\mathrm{cm}^{-1}$. This value shifts toward the low energy side upon the complex formation by about $345.6~\mathrm{cm}^{-1}$ according to quantum chemical calculation findings. Geometrical and spectral parameters of the complex are in good agreement with the calculated parameters in [6]
Table 1: Calculated Stabilization Structural Parameters of the ${\mathrm{H}}_{2}\mathrm{{CO}}\cdots \mathrm{{HF}}$ Complex
<table><tr><td>Distances, (Å)</td><td>Monomers</td><td>Complex</td><td>Angles, (°)</td><td>Monomer</td><td>Complex</td></tr><tr><td>r(H3–F)</td><td>0.915</td><td>0.936</td><td>∠OCH1</td><td>121.5</td><td>121.3</td></tr><tr><td>r(C=O)</td><td>1.207</td><td>1.215</td><td>∠OCH2</td><td>121.5</td><td>121.1</td></tr><tr><td>r(C–H1)</td><td>1.101</td><td>1.096</td><td>∠OH3F</td><td></td><td>166.4</td></tr><tr><td>r(C–H2)</td><td>1.102</td><td>1.097</td><td>∠COH3</td><td></td><td>111.2</td></tr><tr><td>r(O...H3)</td><td></td><td>1.753</td><td>∠COF</td><td></td><td>141.4</td></tr><tr><td>r(O...F)</td><td></td><td>2.649</td><td></td><td></td><td></td></tr></table>
Dipole moment of the complex calculated in CCSD(T)6311++G(3df,3pd) approximations equals to
3.81D. Change of geometrical parameters upon complex formation of the complex are shown in Table 2.
Table 2: The Changes of the-Selected Geometrical Parameters of the ${\mathrm{H}}_{2}\mathrm{{CO}} \cdot \cdot \cdot \mathrm{{HF}}$ upon the Complex Formation
<table><tr><td colspan="4">H2CO···HF</td></tr><tr><td colspan="4">Interatomic distances (Å)</td></tr><tr><td>Δr(C=O)</td><td>Δr(CH2)</td><td>Δr(CH3)</td><td>Δr(HF)</td></tr><tr><td>0.0057</td><td>-0.0047</td><td>-0.0039</td><td>0.0186</td></tr></table>
In Table 3. calculated and experimental determined frequency and intensities are shown. The banding energy of the complex equals to $-4.91\mathrm{kJ}\cdot \mathrm{mol}^{-1}$, and $r(\mathsf{H - F})$ atomic distance to be 0.921 Å.
Table 3: Selected Harmonic Fundamental Transition Frequencies N(Cm $^{-1}$ ) And Intensities S(Km·Mol $^{-1}$ ) and Experimental Results of the H $_2$ CO $\cdots$ HF Complex and Monomers
<table><tr><td rowspan="2">Assignments</td><td colspan="2">Harmonic</td><td colspan="2">Experiment</td></tr><tr><td>v</td><td>S</td><td>v</td><td>S</td></tr><tr><td>C=O stretch</td><td>1758</td><td>75</td><td>1744</td><td>61</td></tr><tr><td>C-H in phase stretch</td><td>3017</td><td>47</td><td>2763</td><td>42</td></tr><tr><td>C-H out of phase</td><td>3120</td><td>44</td><td>2882</td><td>39</td></tr></table>
## VI. CONCLUSION
In this work $\mathsf{H}_2\mathsf{CO}\dots \mathsf{HF}$ complex is studied with the help of FTIR spectroscopy and quantum chemical calculations. High-resolution absorption spectra of the complex in regions of $C = O$, and C-H are presented. Although, the fact that no changing reasons in $\nu (HF)$, stretching bands upon the complexation are explained.
Quantum chemical calculations are carried out at mp2/6-311++g(3df,3pd) approximation. Dipole moment is equal to 3.81 D. The complex formation energy equals to $-4.91\mathrm{kJ}\cdot \mathrm{mol}^{-1}$. A good agreement was found between experimental and computational results and the results coincide with the relevant literatures. We believe that the experimental and computational results of this work can be used to better realize the internal dynamics of the B...HF complexes.
Generating HTML Viewer...
References
16 Cites in Article
V Bulychev,K Tokhadze (2014). Determination of mechanisms of the ν1(H–F) band shape formation in the absorption spectra of H-bonded complexes of HF from combined experimental studies and nonempirical calculations.
V Bulychev,A Koshevarnikov,K Tokhadze (2017). The structure and vibrational spectral parameters of a complex of HF with the planar (H2CO)2 dimer.
R Asfin,V Bulychev,M Buturlimova,K Tokhadze (2022). Trimers formed by formaldehyde with hydrogen fluoride: Structures, energetics, and infrared absorption spectra.
A Amonov,G Murodov,K Tokhadze,A Jumabaev,G Nurmurodova (2020). A study of H2CO•••HF Complex by Advanced Quantum Mechanical Methods.
J Andersen,A Voute,D Mihrin,J Heimdal,R Berg,M Torsson,R Wugt Larsen (2017). Probing the global potential energy minimum of (CH2O)2: THz absorption spectrum of (CH2O)2 in solid neon and <i>para</i>-hydrogen.
R Asfin,V Bulychev,M Buturlimova (2021). Theoretical and matrix isolation studies of infrared spectra of the H 2 CO•••HF hydrogen-bonded complex.
A Amonov,G Murodov,A Jumabaev,G Nurmurodova (2022). Exploration of the СH<sub>3</sub>CN<sup>…</sup>HF complex in the gas phase with high-resolution FTIR spectroscopy and computational methods.
V Bulychev,M Buturlimova,K Tokhadze (2018). Calculation of vibrational spectroscopic and geometrical characteristics of the [F(HF)2]− and [F(DF)2]− complexes using the second-order vibrational perturbation theory and a 6D variational method.
F( Df (2018). -complexes using the second-order vibrational perturbation theory and a 6D variational method.
Valentin Bulychev,Marina Buturlimova,Konstantin Tokhadze (2013). Anharmonic Calculation of the Structure, Vibrational Frequencies and Intensities of the NH<sub>3</sub>···<i>trans</i>-HONO Complex.
V Bulychev,M Buturlimova,K Tokhadze (2017). Quantum-Mechanical Prediction of the Averaged Structure, Anharmonic Vibrational Frequencies, and Intensities of the H 2 O•••trans-HONO Complex and Comparison with the Experiment.
J Bloino (2015). A VPT2 route to near-infrared spectroscopy: the role of mechanical and electrical anharmonicity.
Vincenzo Barone (2005). Anharmonic vibrational properties by a fully automated second-order perturbative approach.
I Gordon,L Rothman (2022). Editorial Board.
S Bach,B Ault (1984). Infrared matrix isolation study of the hydrogen-bonded complexes between formaldehyde and the hydrogen halides and cyanide.
M Frisch,G Trucks,H Schlegel,G Scuseria,M Robb,J Cheeseman (2016). Unknown Title.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Akhtam Amonov. 2026. \u201cInvestigation of Formaldehyde+Hydrogen Fluoride Complex in Gas Phase by the Help of Ftir Spectroscopy and Computational Methods\u201d. Global Journal of Science Frontier Research - H: Environment & Environmental geology GJSFR-H Volume 23 (GJSFR Volume 23 Issue H1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
High-resolution infrared absorption spectra of the H2CO•••HF complex and its monomers are recorded in a gas phase in C=O and C-H regions using Bruker HR 125 spectrometer recorded at 0.05 cm-1 resolution. After complex formation about the HF stretching band complicated shapes are not observed due to weak interaction couplings between C=O and C-H stretching of the H2CO•••HF complex. Also, quantum chemical calculations are performed in mp2//6311++g(3df, 3pd) approximation, and energetic, geometrical, and harmonic spectral parameters have been calculated.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.