The nature of intracellular bone salt is elusive. Its concentration and distribution are frequently fugitive to standard electron microscopy procedures, such that the inorganic phase of bone is widely reported as an extracellular precipitate of hydroxyapatite crystals epitactic on collagen and obedient to the laws of inorganic chemistry. Light microscopy, in contrast, captures the cyclical intracellular mineral loading of “switched on” osteocyte cohorts. Rapidly fabricated within their golgi apparatus are nascent microspheres, approximately 1 micron diameter, calcified with phosphate/carbonate and unloaded at the calcification front in a stress-related process also manifest in a silt-burrowing invertebrate. The calcified objects densely populating the bone matrix may be examined in situ or isolated for density fractionation analysis. They are variably tempered by Si, Mg, Al, K, Na, Fe, stain histochemically for acid phosphatase, bone sialoprotein, osteocalcin encapsulation, osteopontin and nucleic acids, are Gram stain positive for microbes and form a fluorescent complex with the antibiotic tetracycline.
## I. INTRODUCTION
A ge- and sex-related skeletal fragility factors primarily relate to a negative turnover and a low bone mass. However, in addition are less accessible factors of structural "quality" that are independent of the mass; one of these concerns the fundamental nature of bone mineral which is histologically more complex than often supposed. In
Foremost in orchestrating the calcification event is the golgi apparatus and its differential activity between cells of soft and hard tissues, a distinction modified by age [3]. With longevity, golgi-directed force discrimination tends to shift, causing soft tissues to harden and hard ones to soften, with potential pathological consequences. Paradoxically, a broader perspective may be projected on the skeletal stress-related significance of the vertebrate golgi body by reference to calcification and transduction routes in the invertebrates and lower orders in biology.
## II. THE INTRINSIC BIOLOGY OF BONE MINERAL: RELATED FACTORS
### a) Invertebrate adaptation to stress
The origins of an evolutionary supremacy of the golgi apparatus in bone mineral fabrication is illustrated in the invertebrates by the large, contractile protozoan Spirostomum ambiguum[4,5]. This common aquatic organism "switches on" its juxtanuclear organelle, i.e.golgi apparatus, to fabricate numerous micron-sized objects calcified with phosphate resembling those in bone. Following release into the cytoplasm their alignment is polarised relative to spiralling muscle myonemes to facilitate a silt-burrowing phase during arid conditions. With the restitution of their habitual aqueous environment the less stressful swimming mode of the organism is resumed, the golgi body "switches off" and much of the particulate cargo is jettisoned, the remainder stored as a metabolic mineral reserve [6]. This protozoan model of the juxtanuclear golgi apparatus as receptor and biological mediator of extraneous forces and mineral particle generation seems contrary to much established doctrine about vertebrate calcification as an extracellular process of epitactic crystal growth obedient to ritual laws of physics and chemistry.
### b) Vertebrate adaptation to stress
When the quiescent, discrete juxtanuclear golgi bodies of receptive osteocytes are stimulated in bone by extraneous biomechanical signals they expand for the purpose of concerted vesicular bone mineral fabrication across interconnected cell cohorts proximal to the calcification front; this recapitulates on a macrocosmic scale the basic, stress-related pattern of protozoan behaviour. There follows extracellular signal dissemination and repercussions across 3 expansive hierarchical transduction networks, spatially integrated for functional competence [3].
### c) Vertebrate stress transduction networks
1. Inorganic particulates and the osteocyte syncytium. The Golgi-directed cycle of calcium phosphate/carbonate loading and unloading(each cycle about 15 min duration)is most evident in "young" osteocytes[7]at the advancing calcification front of developing bone using von Kossa, GBHA[8]or tetracycline stain[9], Figure 1. In particular, the antibiotic tetracycline forms a fluorescent histochemical complex with bone salt and its specific labelling of the calcification front is a property applied for decades as a histomorphometric marker of bone dynamics introduced by H.M. Frost, Detroit, USA.
In situ, the regional diversity of the osteocyte syncytium in engaging with the extracellular matrix may be captured by a system combining confocal laser microscopy with image analysis software and novel binary masks enabling the definition, separation and mapping of cell features for quantification (as osteocyte number and size, cytoplasmic process volume and density, interconnection and polarity). The results suggest that a less cellular, more widely disconnected syncytium characterises low stress-related osteoporosis, in contrast to high stress osteoarthritis where cytoplasmic volume is raised and disconnection less frequent [10], in keeping with the significant mechanotransduction distinction between the two conditions.
In vitro, the cell dynamics may be recapitulated in substructural detail in cultured osteocytes subjected to extraneous manipulation. Tracking of the prominent golgi-initiated process may be monitored by confocal fluorescence microscopy across interconnected cell cohorts using a specific FGP-tagged construct. Subsequently this pivotal event may be halted by the golgi inhibitor brefeldin A and re-started by the stimulant forskolin[11].
2. Inorganic particulates and microskeletal assemblage. Following their extrusion from the osteocytes, the golgi-fabricated calcified microspheres accumulate in the extracellular matrix as objects of bacterial dimension. They may be discrete or interlinked into budding chains and bridged assemblies, to form an independent microskeletal network of Gram positive-stained objects (crystal violet/iodine microbial method in cryosections). Their properties seem characteristic of a "microbiome" of regional diversity, which encircles, impacts upon and colonises the collagen type I matrix fibrous bundles [12,13]. The inorganic particles are organically enshrouded (lipid, bone sialoprotein [14], osteocalcin [15], Figure 2), variably contain osteopontin (stimulated at remodelling sites) and they remain amorphous or of differentially controlled crystallinity [12]. They apparently integrate spatially into mosaic-like domains (30 microns diameter approx.) of indigenous histochemical diversity e.g., acid phosphatase and carbonic anhydrase activity, and they exhibit widespread RNA content and DNA positivity [13], the latter apparently associated with the calcification front region. In addition is positive autoclastic potential [16] demonstrated as an inherent hydrolytic capacity for rapid self-destruction (autolysis), i.e., constituting strategic "crumple zones" equipped to absorb and dissipate excess energy [17] as an alternative to the more measured process of osteoclasis.
On the one hand, the populations of individual calcified objects (Figure 3) can be harvested from the organic matrix by standard milling and digestion procedures for analysis [18], while on the other, the micro-archaeology of their remains is fossilised for posterity in Khourigba phosphorite, mined as a plant fertiliser. The morphologically organelle-like microspheres have less dense centres surrounded by clusters of electron dense beaded filaments (5nm; Figure 4). They apparently deform under pressure and pair into ladder-like configurations with the potential to further fuse into fenestrated plates. At the calcification front they may also derive from discrete "nests" of precursor nanospheres (40-250nm) that are both intracellular and extracellular in distribution [19, 20]. As well as an inherent capacity to bud into bridged chains the calcified microspheres possess variable traces of Si, Mg, Al, Fe, Na, S as inorganic modulators. Moreover, in expansive growth cartilage and in unloaded regions functional primarily in protection e.g., fish scales of acellular bone, they tend to remain large, discrete and rounded. In contrast, in axially loaded tubular dentine they align and compress into dense, polarised struts and stays, while in the powerful turkey leg tendon they are biomechanically attenuated.
Analysis suggests that relative to stress factors, the microspheres tend to be disproportionately small in low-stress osteoporosis (0.5 microns, Mg deficient) constituting a fine microskeletal assemblage, and disproportionately large in regions of high-stress osteoarthritis (2 microns, relatively Mg, Si deficient) creating a microskeleton that is particularly coarse [21]. As well as influencing structural strength, such textural differences may modulate intraosseous fluid flow and associated mechano-transduction in a manner familiar to gardeners of differing water-retentive clay and sandy soils.
### d) Polarised periosteal Sharpey's fibres and force translation
The long-established birefringent periosteal Sharpey's (Figure 5) formerly assigned the superficial purpose of anchorage, may now also signal incipient force transmission/translation across their extensive intraosseous, uncalcified network. Their ramifications traverse the collagen type I matrix lamellae and marrow spaces to fuse eventually with the endosteal rim and complete the histological loop [22]. They directly integrate intramembranous bone with muscle, ligament and tendon insertions and their prominence, permeation and average insertion angle tend to diminish with age. The identification and tracking of their finest, most distal branches require the application of specific fluorescent antibody markers. Collectively they are most populous and prominent as a substantial, discretely defined proximal femoral domain [23, 24] and they are also a major feature of the powerful porcine mandible [25].
Originating in the periosteum, Sharpey's fibres are apparently basic to embryonic trabecular development and also to skeletal repair in the form of an unmineralised scaffold for osteogenic cell assembly. Augmented by youth and physical activity, the uncalcified fibres, 15-25 microns thick, comprise collagen type III/VI macromolecules beaded with the biological organiser molecule tenascin and encircled with elastin. With female oestrogen decline they progressively calcify and shorten in advance of matrix loss. Their fan-like interspersion among the microskeletal assemblies may direct signal trafficking to engage endosteal remodelling (a turnover process from which they themselves are generally protected until age-related random calcification occurs). In addition, the evidence suggests that in low-stress osteoporosis, the periosteal Sharpey fibre system is fine and fragmented, consistent with poor intraosseous perception of muscle input; in high-stress osteoarthritis the periosteal Sharpey fibre system is coarse and continuous, apparently conducive to maximum perception of muscle input.
### e) Interconnected trabeculae and multiaxial resilience
The golgi-derived calcified microsphere populations influence the intrinsic micro architectural properties of the cancellous network they colonise. This is by virtue of their variable nature and stability which defines not only their remodelling propensity but also determines the likelihood of particle slip or crystal fracture[12]. Cancellous integrity is a major determinant of strength at the most multiaxial, fracture-prone skeletal locations and a susceptibility to trabecular disconnections (particularly of cross struts) reduces strength in excess of the quantity of tissue lost (i.e., doubling the distance between cross struts weakens structural resilience fourfold). Disconnections are rare in youth, except in pregnancy and lactation where their incidence is apparently reversible [26]. In the osteopenic female hip and spine disconnections multiply into predictable "hotspots" of weakness at major microanatomical sites of tension [27,28] ultimately releasing "floating" cancellous segments of minor structural value [17]. Treatment with bisphosphonates apparently provides some protection against hypogonadal cancellous disconnection [29].
In men, age-related trabecular disconnections are fewer than in women and tend to arise in the proximity of the vertebral endplates. However, in high stress osteoarthritis, the distribution of trabecular disconnections is often capricious, with a tendency towards the creation of discrete subchondral bone marrow lesions (BML, about $10\mathrm{mm}$ diameter) as sites of in situ hydrolytic destruction (autoclasis) and associated repair.
### f) The golgi-directed bone mineral biosphere: a stresscompliant prokaryote legacy
The evolutionary significance for backbones of an otherwise apparently undistinguished protozoan by Pautard (1959) is described in section IIa (see also [30]). Combined with the extrapolation to bone cell behaviour of the golgi-body there follows the possibility that minimum trauma fractures may derive from an under performing golgi program imprinted in antiquity to enable delicate aquatic protozoans to tunnel in silt or risk desiccation. Furthermore, a common bacterium may pre-date the protozoan golgi-directed mineral fabrication event (Figure 6). For example, there is evidence for prokaryote-directed assembly of similar microspheres and precursor nanospheres (40-250nm) calcified with phosphate in a silicon-rich intracellular intermediary in the lifecycle of the bacterial model Corynebacterium matruchotii [31, 32]. Perhaps precedent in initiating this milestone event might be envisaged the emergence of copious silicified primordial particles through an ancient geological portal whereby the omnipresence of silicon in bacterium, protozoan and osteocyte alike derive from the singular property of facilitating calcium phosphorylation via carbonate in biological systems resilient to stressful conditions. On the road to backbones [30] the golgi-fabricated inorganic bone mineral microspheres may constitute a Gram positive "petrified microbiome" composed of compacted, mechanically deformable populations of microspherical bionts.
g) Reciprocity between the postulated bone mineral microbiome and the established gut microbiome?
According to recent literature is accumulating evidence from orthopaedic surgery that the gut microbiome often effects bony tissue, influencing the healing process, surgical recovery and even causing impaired cognition trajectories consistent with prospects for targeted intervention with probiotics [33]. When dysbiosis occurs (i.e., disruption of microbial balance) associated clinical conditions extend not only to inflammatory bowel disease, and impaired immune response but also to metabolic bone disorder. The possibility arises of a biochemical reciprocity between two dissimilar microcosms that nonetheless share a metabolic role not only in bone healing and orthopaedic surgical outcome, but also in oestrogen-mediated bone density determination via intermediary endocrinology agents including vitamin D, vitamin K osteocalcin activator, and Gla protein activation, linked to osteoblasts, osteoclasts, mineral density and collagen integrity.
### h) Summary. Another song to sing?
The nature of bone salt is not easily characterised and its inherent sensitivity to preparative manipulation may capture aspects of a modest early evolutionary transformation. Complementary to the widespread view of a collagenous matrix stiffened by uniform sheets of micro crystallites is proposed the alternative of a "petrified microbiome" of hierarchical force translation colonised by biospheres of variable histochemical complexity calcified with calcium phosphate/carbonate.
- Consisting of populations of golgi-fabricated calcified microspherical objects (1micron approximately) the microbiome is the putative evolutionary legacy of a contractile protozoan tunnelling in silt.
- Initially confined to the intracellular "switched on" osteocyte syncytium, their monitored extrusion into a compressed extracellular environment causes microspherical compaction and deformation apparently within the confines of mosaic-like matrix domains (30 microns dimension).
- Exposure to multiaxial forces directs the assembly of the calcified microspheres into a mineral microskeleton permeated by periosteal Sharpey fibre arrays in a partnership with the golgi body that defines the bedrock resilience of each trabecula within the cancellous network.
- Abnormalities in either the recipient microspheres (with the behaviour of functional organelles) or in the force-transmitting periosteal envelope (and Sharpey fibre/endosteal continuum) may predispose to trabecular disconnection, disproportionate weakness and the foundation of stress-related disability.
- If the proposed microcosm seems unlikely, clinical relevance includes particulate populations that are under-sized in under-stressed conditions and oversized in over-stressed conditions. In endochondral cartilage they tend to remain rounded [34] and discrete (as is also the case in the protective acellular bone of fish scales, and in the keratinocytes of hoof and horn [35]).
- Clinical reports include: i) improved bio-implant integration by Sharpey's fibres in the most complex oral maxillofacial surgery named "Embryomimetics" (M. Chin, California [36]); ii) treatment for overloaded periosteal Sharpey's fibres in Schlatter's Syndrome where microfissures accumulate in the over-stimulated knees of teenage footballers during the signal-rich growth spurt (T. Maseide, Norway); iii) excessive torsion in Sharpey's fibre anchorage of degenerate collapsing discs causing vertebral epiphyseal rim erosion found in archaeological remains (M. Dobson, London).
A remarkable developmental time distinction between synthetic chemistry versus biological calcification with phosphate was illustrated by Pautard [37]. By comparison, calcium phosphate precipitation from supersaturated synthetic fluids took about 40 hours under physiological conditions, commencing from ultrastructural, 10-500 angstrom amorphous spheres after 20 hours, which coalesce and recrystallize to apatites after about 40hr. In contrast, bone salt was formed in a quixotic 15 minutes, commencing via golgi vesicle investment, whereby loading with calcium is followed by a spread of phosphate from the nucleus [38] preceding micron-sized, dense particulates (Figure 7). The prospect of an emergent geobiology releasing silicified bone salt bionts may explain the diversity. Chronically modulated by other anions (carbonate and fluoride) and cations (magnesium, potassium, sodium), and following crucial investiture within an organic golgi apparatus precursor, the transition to a microbiome of bone mineral biospheres might be explained (Table 1). Tentative milestones along the rocky, chemical-strewn road to the inanimate/animate calcification frontier of backbones are: 1) geological crystalline/amorphous ultrastructural bone salt deposits; 2) pre-prokaryote microspheres calcified with phosphate/ carbonate; 3) prokaryote calcified with phosphate/carbonate via silicification; 4) protozoan, stress-responsive, golgi body-directed fabrication of bone mineral microspheres calcified with phosphate/carbonate in invertebrates; 5) golgi body-directed fabrication of bone mineral microspheres calcified with phosphate/carbonate (biospheres) in vertebrates; 6) musculoskeletal force translation by a petrified bone mineral microbiome.
Table 1: Evidence-based hypotheses pertaining to the proposed golgi-directed microbiome of bone mineral microspheres.
1. The particulate microskeletal assembly of calcium phosphate by bone cells is predicated by a primordial golgi apparatus programmed for complex mineral fabrication in a contractile, intermittently stressed protozoan.
2. A chronic decline in the substructural delivery of tensile stimulation due to atrophy of a regionally integrated network of intraosseous periosteal Sharpey's fibres "silences" the golgi body and predisposes to "hotspots" of trabecular disconnection (osteoclasis, autoclasis, acute microfracture).
3. The mineral phase of bone is a microcosm of 1 micron geo/biological particulates which colonise the collagenous matrix in a manner suggestive of either a primordial or a regressed microbiome, i.e., inorganic physical chemistry assumes the mantle of biology.
4. Bone mineral microspheres(biospheres) calcified with phosphate/carbonate (i.e. bionts) arrive at the metaphorical inanimate/animate (geo/biological) frontier where their passport to proceed is "stamped" by the golgi apparatus.
#### ACKNOWLEDGEMENTS
The above synopsis is a personal reflection and complementary prelude to the evidence-based paradox presented in technical detail in Reference [3]. It is dedicated to my indefatigable mentors, colleagues etc and students who stoked the research engines for half a century at Leeds and never ceased to amaze me. Support for my Bone Structural Biology Laboratory since inception is gratefully acknowledged from the Medical Research Council, Action Medical Research, Research into Ageing, Help the Aged, EPSRC Challenging Engineering Program in collaboration with the Institute of Mechano biological Engineering Leeds. The following citations are a representative selection and not a comprehensive list.
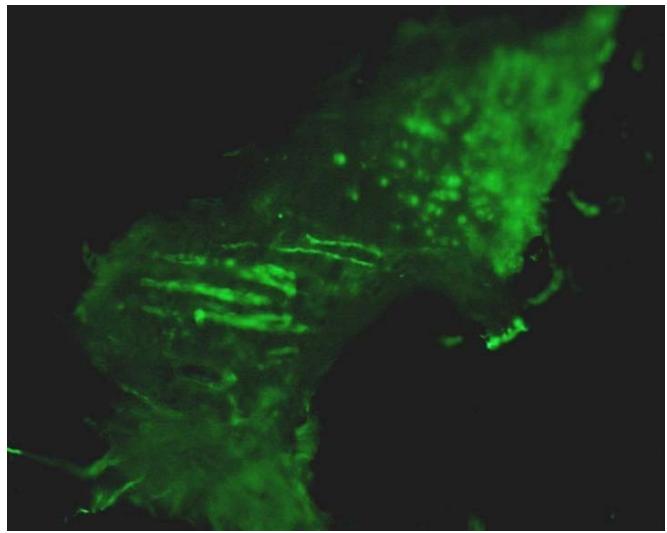
Figure 5: Green, fluorescent periosteal Sharpey's fibres approximately 10 microns thick, rich in collagen Type III and extending from the periosteum, traversing the bone matrix and shown in longitudinal and cross section. Fluorescein isothiocyanate immunostain, cryomicrotomy, epifluorescence microscopy[36].
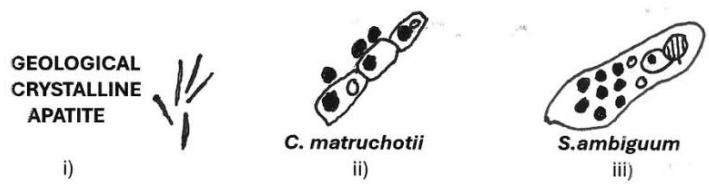
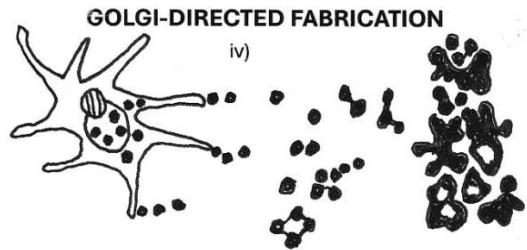
1 micron spheres BONE MINERAL MICROBIOME Figure 6: On the rocky road to the bone mineral microbiome. Diagram illustrating the proposed evolutionary trajectory of bone mineral microspheres calcified with phosphate and colonising the organic matrix as a "petrified microbiome." i) Geological crystalline apatite. ii) Corynebacterium matruchotii calcified with intracellular and extracellular bone mineral microspheres. iii) Spirostomum ambiguum calcified with golgi-fabricated bone mineral microspheres. iv) Osteocyte calcified with golgi-fabricated bone mineral microspheres, their migration into the collagenous extracellular matrix and assembly in groups.
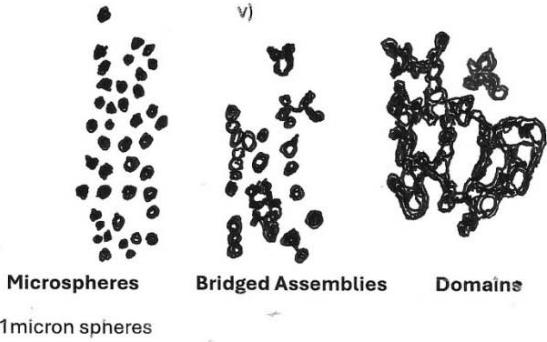
v) Microbiome comprising bone mineral biospheres as discrete objects, "budding" into chains or in bridged assemblies and domains.
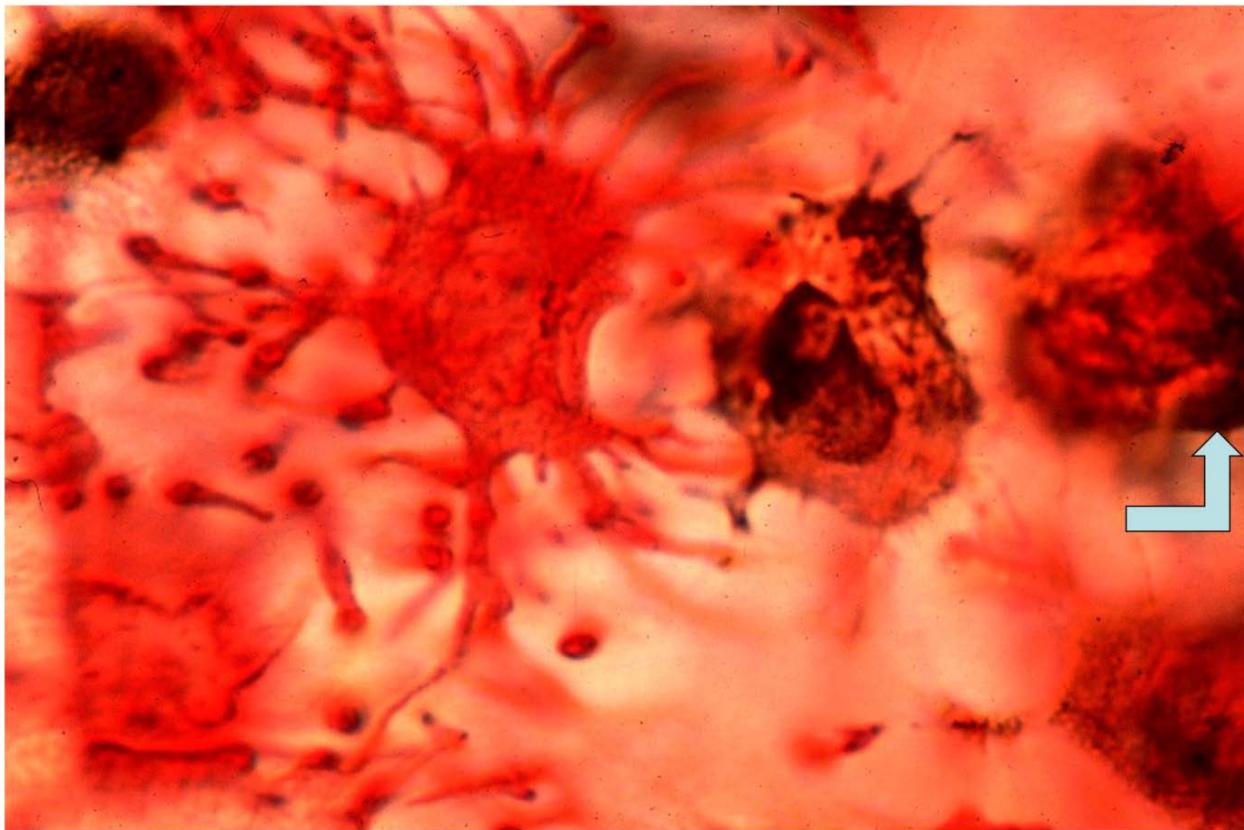
Figure 7: Three osteocytes in intact developing mouse calvarium sequentially loading with bone mineral in a cycle of approximately fifteen minutes. Left cell: loaded throughout with red-stained calcium. Middle cell: brown-stained phosphate released from the nucleus into the cytoplasm. Right cell: densely packed mineral particulates fabricated in the expanded, crescent-shaped juxtanuclear golgi apparatus (arrowed). GBHA and von Kossa stain, Nomarski optics [38].
Generating HTML Viewer...
References
39 Cites in Article
(1969). absorption of amino acids by apatite crystals.
J Aaron (2024). Cancellous skeleton, microskeleton, ultramicroskeleton: a geo/biomorphological bone mineral microbiome of hierarchical force translation and ancient golgi-directed lineage.
F Pautard (1959). Hydroxyapatite as a developmental feature of Spirostomum ambiguum.
Alla Gracheva (1962). MAXIM GORKY: PORTRAIT OF THE UNKNOWN WITH GERMANY AS THE BAC KGROUND.
V Fallon,P Garner,J Aaron (2017). Mineral fabrication and g olgi apparatus activity in Spirostomum ambi guum: a primordial paradigm of the stressed b one cell.
Jean Aaron (1973). Osteocyte types in the developing mouse calvarium.
Herbert Kashiwa (1970). Calcium Phosphate in Osteogenic Cells.
J Aaron,F Pautard Tetracycline staining of bone in normal and pathol ogical states.
P Garner,R Wilcox,J Aaron (2013). A direct method for the spatial 3D mapping of trabecular termini in the spine.
Valerie Fallon,D Carter,Jean Aaron (2014). Mineral Fabrication and Golgi Apparatus Activity in the Mouse Calvarium.
J Aaron (2003). Bone turnover and microdamage.
Jean Aaron (2016). Cellular Ubiquity of Calcified Microspheres: A Matter of Degree, Ancient History and the Golgi body?.
P Bianco,M Riminucci,G Silvestrini,E Bonucci,J Termine,L Fisher,P Robey (1993). Localization of bone sialoprotein (BSP) to Golgi and post-Golgi secretory structures in osteoblasts and to discrete sites in early bone matrix..
S Shahtaheri,J Aaron,B Oakley (2002). Immunolocalization of osteocalcin in calcified microspheres in lamb vertebrae and mouse bones using PAP method.
F Burton,M Neuman,W Neuman (1969). On the possible role of crystals in the origins of life. The adsorption of nucleosides, nucleotides and pyrophosphate by apatite crystals.
F Burton,M Neuman,W Neuman On the possible role of crystals in the origins of life.
Jean Aaron (1977). Autoclasis—a mechanism of bone resorption and an alternative explanation for osteoporosis.
Patricia Shore,Roger Shore,Jean Aaron (2023). Morphogenesis of Floating Bone Segments: A Legacy of Serial Tensile Cross-Strut Microdamage in Trabecular Disconnection “Crumple Zones”?.
J Aaron,B Oliver,N Clark,D Carter (1999). Calcified microspheres as biological entities and their isolation from bone.
D Carter,P Hatton,J Aaron (1997). The ultrastructure of slam frozen b one mineral.
D Carter,A Scully,R Davies,J Aaron (1998). Evidence for phosphoprotein microspheres in bone.
Kathryn Linton,Lesley Hordon,Roger Shore,Jean Aaron (2014). Bone Mineral “Quality”: Differing Characteristics of Calcified Microsphere Populations at the Osteoporotic and Osteoarthritic Femoral Articulation Front.
Jean Aaron (2012). Periosteal Sharpey’s fibers: a novel bone matrix regulatory system?.
F Luther,H Saino,D Carter,J Aaron (2003). Evidence for an extensive collagen type III/VI proximal domain in the rat femur I. Diminution with ovariectomy.
H Saino,F Luther,D Carter,A Natali,D Turner,M Shahtaheri,J Aaron (2003). Evidence for an extensive collagen type III/VI proximal domain in the rat femur II. Expansion with exercise.
A Al-Qtaitat,R Shore,J Aaron (2010). Structural changes in the ageing periosteum using collagen III immunostaining and chromium labelling as indicators.
Seyed Shahtaheri,Jean Aaron,David Johnson,David Purdie (1999). Changes in trabecular bone architecture in women during pregnancy.
P Garner,R Wilcox,J Aaron (2013). A direct method for the spatial 3D mapping of trabecular termini in the spine.
Jean Aaron,Patricia Shore,Mizuo Itoda,Rory Morrison,Andrew Hartopp,Elizabeth Hensor,Lesley Hordon (2015). Mapping trabecular disconnection “hotspots” in aged human spine and hip.
L Hordon,M Itoda,P Shore,R Shore,M Heald,M Brown,J Kanis,G Rodan,J Aaron (2006). Preservation of thoracic bone microarchitecture by alendronate: Comparison of histology and microCT.
F Pautard (1961). Calcium phosphate microspheres in biology.
Kathryn Linton,Charles Tapping,David Adams,D Carterr,Roger Shore,Jean Aaron (2013). A silicon cell cycle in a bacterial model of calcium phosphate mineralogenesis.
J Ennever,H Creamer (1967). Microbiologic calcification: bone mineral and bacteria.
A Nadeem-Tariq,S Kazemeini,M Michelberger,C Fang,K Nelson,S Maitra (2025). The role of gut microbiota in orthopaedic surgery: a systematic review. Microorganisms. Special Issue: Microbiome Research: Past, Present and Future.
Keith Lester,Major Ash (1980). Scanning electron microscopy of mineralized cartilage in rat mandibular condyle.
F Pautard (1963). Mineralization of keratin and its comparison with enamel matrix.
M Chin,J Aaron (2019). Sharpey biologic model for bone formation.
F Pautard (1981). Calcium phosphate microspheres in biology.
J Aaron (1980). Demineralization of bone in vivo and in vitro: evidence for a microskeletal arrangement.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Dr. Jean E. Aaron. 2026. \u201cMicrobiome of Bone Mineral Microspheres: A Brief History on the Golgi-Directed Road to Force Translation and Geo/Biomorphological Evolution\u201d. Global Journal of Science Frontier Research - C: Biological Science GJSFR-C Volume 25 (GJSFR Volume 25 Issue C1): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
The nature of intracellular bone salt is elusive. Its concentration and distribution are frequently fugitive to standard electron microscopy procedures, such that the inorganic phase of bone is widely reported as an extracellular precipitate of hydroxyapatite crystals epitactic on collagen and obedient to the laws of inorganic chemistry. Light microscopy, in contrast, captures the cyclical intracellular mineral loading of “switched on” osteocyte cohorts. Rapidly fabricated within their golgi apparatus are nascent microspheres, approximately 1 micron diameter, calcified with phosphate/carbonate and unloaded at the calcification front in a stress-related process also manifest in a silt-burrowing invertebrate. The calcified objects densely populating the bone matrix may be examined in situ or isolated for density fractionation analysis. They are variably tempered by Si, Mg, Al, K, Na, Fe, stain histochemically for acid phosphatase, bone sialoprotein, osteocalcin encapsulation, osteopontin and nucleic acids, are Gram stain positive for microbes and form a fluorescent complex with the antibiotic tetracycline.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.