Mycorrhizae are a symbiosis between the roots of some plants and certain soil fungi, where both participants in this association obtain benefits. The plant provides the fungus with carbohydrates, proteins, and lipids, and in turn, the fungus allows the plant to better uptake nutrients from the soil and water. The objective was to determine the existence of mycorrhizal associations between native fungi and forage crops in the Central Valley of Catamarca. Root colonization of Melilotus officinalis, Avena sativa, Hordeum vulgare, Secale cereale, Panicum maximum, and Cenchrus ciliaris from field crops in the Central Valley of Catamarcawas studied. Fungal infection or colonization was quantified using the line intersection method and the frequency of appearance of fungal structures. Endomycorrhizal structures of the arbuscular vesicle type were observed, with continuous mycorrhizal hyphae, some of them with rosary lipids inside and vesicles with fat globules. In addition, dark septate endophytic fungi were observed. The association of mycorrhizae is described in six forage species cultivated in the Central Valley of Catamarca and the co-occurrence of arbusculo-vesicular mycorrhizae and native dark septate endophytes.
## I. INTRODUCTION
Mycorrhizae are symbioses between plant roots and certain soil fungi. The term mycorrhiza describes the group made up of the absorption structures of plants and the fungal symbionts that colonize them in a distinctive way (Sánchez de Prager, 2007; Perez et al., 2010), generating a structural and metabolic integration typical of a symbiosis. The absorption of nutrients is affected by these associations that also influence plant health, productivity, and the adaptation of plants to environmental conditions (Sánchez de Prager, 2007).
The plant provides the fungus with carbohydrates, proteins, and lipids, necessary for its development, and in turn, the fungus allows the plant to better capture water and nutrients from the soil, both macronutrients and micronutrients, especially those few mobile, such as phosphorus (Sánchez de Prager, 2007).
Endo mycorrhizae and ectomycorrhizae are two types of mycorrhizae that occur naturally (Sieverding, 1983; García et al., 2000; Biaus, 2017). Endomycorrhizae are more frequent in nature and are characterized by the colonization of cortical cells by a mycosymbiont, which lives between them and within them, inter and intracellularly (Montenegro Gómez et al., 2017). Among the different types of endomycorrhizal, the most widespread within plant species are vesicular-arbuscular mycorrhizae (VAM) or arbuscular mycorrhizae (AM) and some more specific forms such as orchidoids and ericoid (Sánchez de Prager, 2007; Montenegro Gómez et al., 2017).
Arbuscular mycorrhizal fungi (AMF) and vesicular-arbuscular fungi (HMVA) do not develop in pure culture media because they are not capable of growing in the absence of a host plant (Aguilar-Ulloa et al., 2016), so they are considered obligate symbionts, this biological condition that represents an obstacle to their massive propagation. These fungi are propagated using trap plants, which after a period of growth, the roots and soil are collected for use as inoculant of endomycorrhizal fungi (De la Rosa-Meraet al., 2012). In recent decades, the study of these organisms has become important for apply them to the ground, as biofertilizers to increase the productivity in crops (Covacevich and Echeverria, 2010; Reyes Tena et al., 2015; Aguilar-Ulloa et al., 2016; Ordoñez-Castaneda et al., 2021) and in phytoremediation, programs to correct contaminated soils (Pérez et al., 2021; Colombo et al., 2020; Quiroz-Mojica et al., 2021). The colonization of roots by endomycorrhizal fungi is the most used parameter as a quick indicator of the presence of mycorrhizal symbiosis (Covacevich and Echeverria, 2010).
Several plant species have been investigated as trap crops to produce mycorrhizae, to find plants that can be colonized and allow the rapid growth of mycorrhizae (Aguilar-Ulloa et al., 2016). The appropriate trap crop must be a fast-growing plant that adapts to the environmental conditions where it will grow, must be easily colonized by the mycorrhizal fungus, and produce many roots in a relatively short time (45-60 days) (Siqueira Martins et al., 2017).
The association established by AMF is not specific, which allows the same fungus to colonizes different plant species to generate symbiosis (Aguilar-Ulloa et al., 2016). Also, there is a preference that certain AMF colonize and spread better in certain plant species (Covacevich and Echeverria, 2010). Furthermore, using the same fungal species is probably not optimal for all crops. For this reason, it is convenient to evaluate the MVA in each trap crop species.
Because the interactions between different VMAs with soil microorganisms are complex, it is necessary to determine their behavior in the field under the growth conditions of each crop. The use of native MVA is recommended due to its adaptation to prevailing conditions, avoiding ecological risks associated with introducing exotic species (Sánchez de Prager, 2007).
The objective was to determine the existence of mycorrhizal associations between autochthonous soil fungi and forage crops planted in the Central Valley of Catamarca and to evaluate the biological aspects of the interactions found.
## II. MATERIALS AND METHODS
Colonization of mycorrhizae in plant roots of six commonly cultivated forage species in the region was studied. The sowings were carried out in the locality of Miraflores, Department of Capayán, in the Central Valley of Catamarca, in plots of native forest or with agricultural history without application of mycorrhizal fungi. The species evaluated were: clover (Melilotus officinalis L.), oats (Avena sativa L.), barley (Hordeum vulgare L.), rye (Secale cereale L.), Gatton panic (Panicum maximum Jacq.), and buffel grass (Cenchrus ciliaris L.).
In the laboratory, the roots of each of the collected plants were extracted and washed with running water. The thinnest were selected, those that were clarified and stained following the methodology of Phillips and Hayman (1970) to determine fungal colonization and detection of mycorrhizal structures. Staining was performed with Gueguén's triple dye solution, allowing fungi to stain proteins blue simultaneously, starch violet, fats red (Sarasola and Rocca, 1975), and glycogen in mahogany (Verna and Herrero, 1952).
In each specimen, the percentage of fungal colonization by MVA and the percentages of the content of arbuscules (A) and vesicles (V) were quantified by the line intersection method of Giovannetti and Mosse (1980), and the frequency of appearance of A and V (Covacevich et al., 2001). For each specimen, 15 segments of the colored roots were taken, and distributed randomly on a grid slide. By means of microscopic observation (10x and 40x), the presence absence of mycorrhizal structures (A and V) was recorded in the horizontal and vertical intersections between roots and grid lines. Three repetitions of each species were performed, quantifying at least 100 intersections per preparation to later calculate the frequency of mycorrhizal infection, according to Giovannetti and Mosse (1980).
$$
MGiovannetti\,\%\,=\,\text{No.\,SI}\,\times\,100\,\/\,\text{No.\,SO}
$$
Where:
SI: number of infected segments (hyphae + arbuscules + vesicles) and
SO: numberof total segments observed (hyphae + arbuscules + vesicles + no infection).
Also, the percentage of hyphae of dark septateendophytes (ESO) and the percentage of microsclerotia were recorded.
## III. RESULTS AND DISCUSSION
In the six analyzed forage species, typical structures of endomycorrhizal fungi of the MVA type (hyphae, arbuscules, and vesicles), and of ESO fungi (with septate, melanized hyphae, and numerous microsclerotia) were observed.
In all species there were continuous thin and thick hyphae, with intracellular and intercellular growth and some of them with lipids in a rosary inside.
In clover, arbuscular distributed throughout the bark were observed, involved in the bidirectional transfer of nutrients (Smith and Read, 1997). Numerous vesicles of diverse morphology (spherical, oval, tapered) were also observed in this legume, so it is inferred that the roots are colonized by various species or genera of native HMVA. Vesicles with light blue (saccule) and red (single or multiple globules) colorations were observed. These structures are related to the carbon storage of in the form of lipids and fatty acids. For this reason, vesicles are defined as reserve organs of the fungal symbiont (Sieverding, 1983).
A high level of fungal colonization was determined in the six forage plants, obtaining the highest values of mycorrhizal colonization (MC) in clover and barley roots (Table 1). The highest frequency of appearance of arbuscules (FAA) was observed in clover, while the lowest number of arbuscules was determined in oats. The highest frequency of vesicle appearance (FAV) was also observed in clover, with average values of $20\%$ (Table 1). These results coincide with the statements of Covacevich and Echeverria (2010) that indicate that there is a preference for certain AMF to better colonize certain plant species. The more significant colonization in clover is explained by the fact that Rhizobios-arbuscular mycorrhizae (AM) associations occur in legumes that act synergistically in infection, mineral nutrition, and plant growth (Fitter & Garbaye, 1994; Barea, 1997). The main effect of AM in enhancing rhizobia, activity is through a generalized stimulation of plant nutrition, some more localized
effects may occur at the root (Melo de Miranda et al., 2008; Spagnolletti et al., 2021).
Table 1: Fungal colonization in forage species from the Central Valley of Catamarca
<table><tr><td rowspan="2">Species</td><td colspan="3">Mycorrhizal Fungi</td><td rowspan="2">Fungi ESO FAM4 (%)</td></tr><tr><td>MC1 (%)</td><td>FAA2 (%)</td><td>FAV3 (%)</td></tr><tr><td>Clover</td><td>89</td><td>47</td><td>20</td><td>15</td></tr><tr><td>Barley</td><td>89</td><td>17</td><td>15</td><td>15</td></tr><tr><td>Rye</td><td>83</td><td>27,7</td><td>16</td><td>13</td></tr><tr><td>Buffalograss</td><td>76,5</td><td>20</td><td>8</td><td>2</td></tr><tr><td>gatton panic</td><td>75,8</td><td>32</td><td>9,8</td><td>1,7</td></tr><tr><td>Oatmeal</td><td>75</td><td>10</td><td>12</td><td>4</td></tr></table>
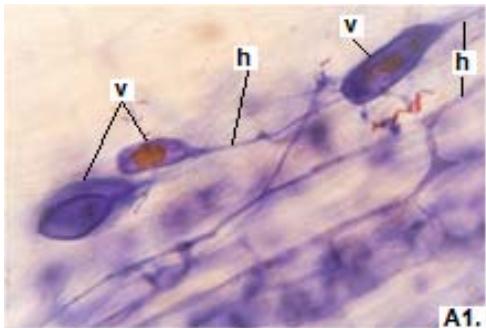
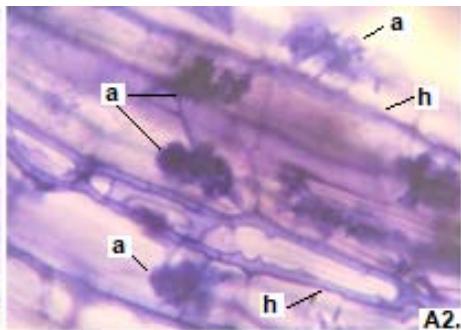
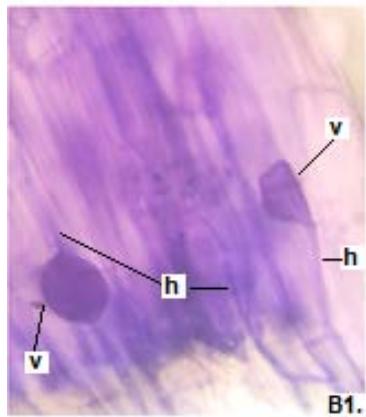
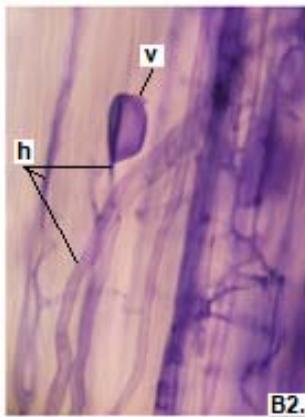
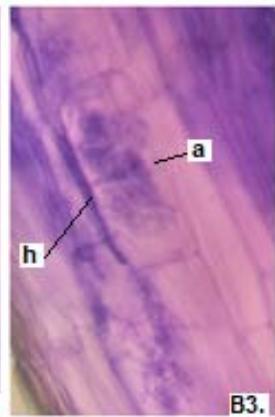
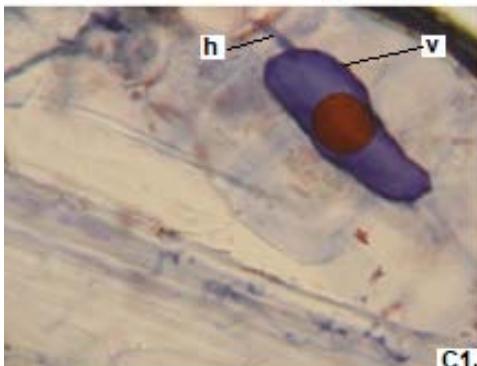
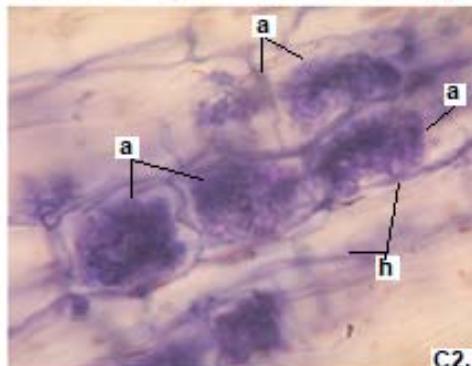 Figure 1: Photomicrographs of mycorrhizal structures in roots of forage species. (A: Clover; B: Barley; C: Rye). (a: Arbuckle; h: hyphae; v: Vesicle).(Magnification: 40x)
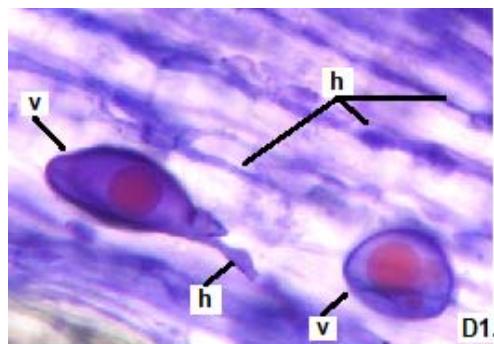
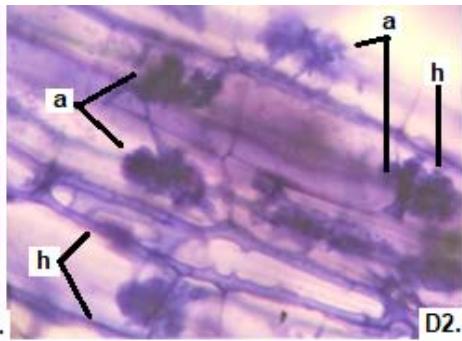
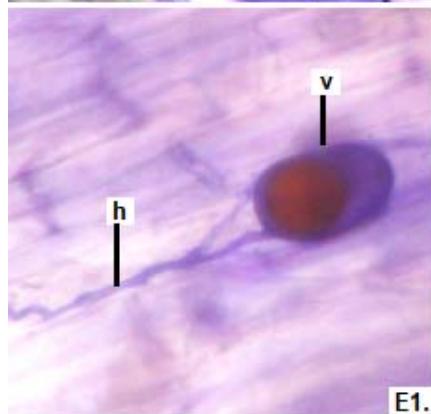
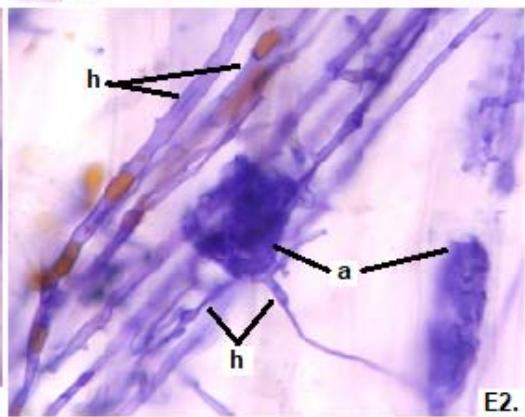
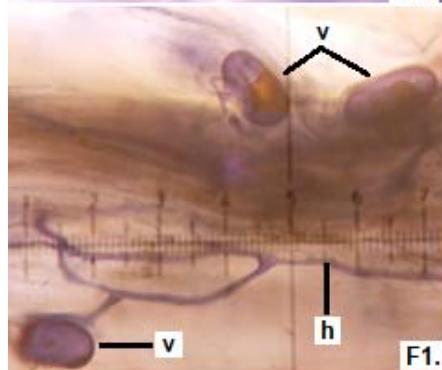
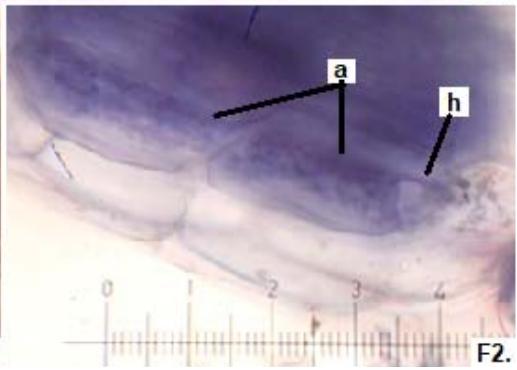 Figure 2: Microphotographs of mycorrhizal structures in roots of forage species. (D: Oats; E: Buffel Grass and F: Gatton Panic). (a: Arbuckle; h: hyphae; v: Vesicle). (Magnification: $40 \times$ ) In addition, in all the forage species analyzed, another type of hyphae was observed: septate, melanized, and with numerous microsclerotia of the kind ESO fungi (Peterson et al., 2004). In clover, barley, and rye roots, similar values of FAM of ESO (between 15 and $13\%$ were determined (Table 1).
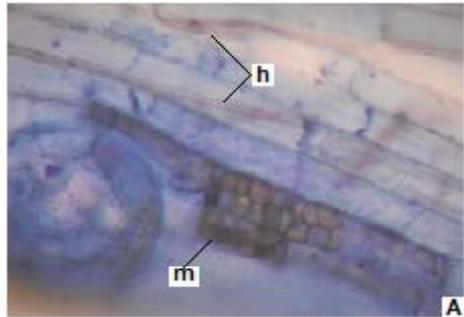
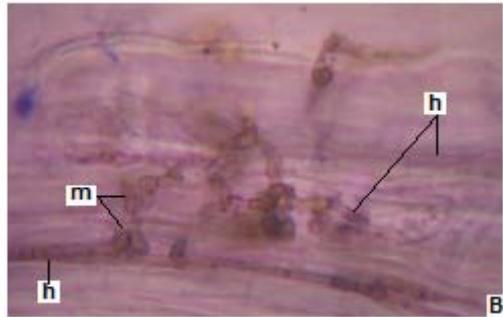

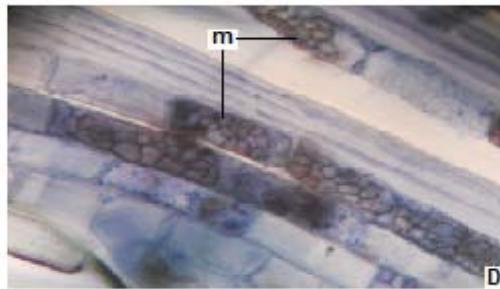

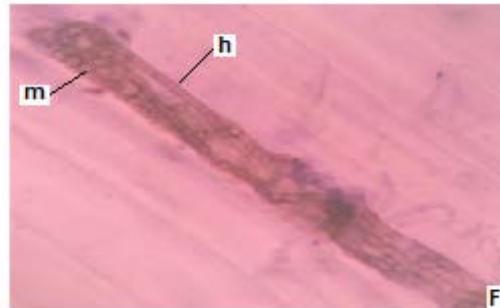 Figure 3: Photomicrographs of dark septate fungal structures on roots of forage species. (A: Clover; B: Barley; C: Rye; D: Oats; E: Buffel Grass and F: Gatton Panic). (m: Microsclerosia; h: hypha). (Magnification: $40x$ ).
In all the plants studied, their roots were simultaneously colonized by both endophytes, MVA and ESO hyphae, also detecting the presence of vesicles, arbuscules typical of VA mycorrhizae, and ESO microsclerotia. This was also observed in other plants including ferns, mono, and dicots that are simultaneously colonized by MVA and ESO. (Urcelay et al., 2005; Lugo et al., 2011; Lizárraga et al., 2015; Di Barbaro et al., 2017).
Therefore, MVA and other fungal structures developed in all the forage species studied. This coincidence could be due to the fact that all these crops were carried out in contiguous lots with identical soil characteristics and environmental conditions, and because the fungal colonization developed from the native microflora, with the ability to associate and generate MVA, which is consistent with what expressed by Aguilar-Ulloa et al. (2016) where they explain that the same fungus can colonize different plant species to generate symbiosis.
These mycorrhizal-forming fungi can be considered as potential constituents of biofertilizers. Diaz Franco et al. (2019) achieved the reduction of inorganic fertilization through the inoculation of FAM in the sorghum crop. The higher yield of sorghum obtained allows considering the inclusion of FAM as a viable practice that guarantees greater profitability, as well as the agroecological conservation of production systems.
## IV. CONCLUSION
The association of mycorrhizae in six forage species cultivated in the Central Valley of Catamarca and the co-occurrence of vesicular-arbuscular mycorrhizae and native dark septate endophytes is described.
High levels of mycorrhizal colonization were obtained in all the evaluated forage species. The suitability of Melilotus officinalis as a trap species is highlighted as it is easily colonized by mycorrhizal fungi and generates rapid mycorrhizal growth.
Generating HTML Viewer...
References
31 Cites in Article
W Aguilar-Ulloa,P Arce-Acuña,F Galiano-Murillo,T Torres-Cruz (2016). Aislamiento de esporas y evaluación de métodos de inoculación en la producción de micorrizas en cultivos trampa.
J Barea,A Ogoshi,I Kobayashi,Y Homma,F Kodama,N Kondon,S Akino (1997). Plant Growth Promoting Rhizobacteria in Sustainable Agriculture: Present Status and Future Prospective.
Daiana Zanardi (2017). Análisis de calidad profesional en graduados universitarios.
Roxana Colombo,Matias Benavidez,Laura Fernandez Bidondo,Vanesa Silvani,María Bompadre,Marina Statello,Maria Scorza,Adalgisa Scotti,Alicia Godeas (2020). Arbuscular mycorrhizal fungi in heavy metal highly polluted soil in the Riachuelo river basin.
F Covacevich,H Echeverría,L Aguirrezabal (2001). Comparación de dos técnicas de cuantificación de infección micorrícica.
F Covacevich,H Echeverría (2010). Indicadores para seleccionar inóculos de hongos micorrícicos arbusculares eficientes en suelos moderadamente ácidos.
Claudia De La Rosa-Mera,Ronald Ferrera-Cerrato,Alejandro Alarcón,María Sánchez-Colín,Alicia Franco-Ramírez (2012). Aislamiento de consorcios de hongos micorrícicos arbusculares de plantas medicinales y su efecto en el crecimiento de vinca (Catharanthus roseus).
Gabriela Di Barbaro,Horacio Andrada,Valeria González Basso,Ana Alurralde,Eleodoro Del Valle,Celia Brandán De Weht (2017). Micorrizas arbusculares y hongos septados oscuros nativos en topinambur (Helianthus tuberosus L.) en Catamarca, Argentina.
Arturo Díaz Franco,Martin Espinosa Ramírez,Flor Ortiz Cháirez (2019). REDUCCIÓN DE LA FERTILIZACIÓN INORGÁNICA MEDIANTE MICORRIZA ARBUSCULAR EN SORGO.
Federico Spagnoletti,Agustina Di Pardo,Natalia Tobar Gómez,Viviana Chiocchio (2013). Las micorrizas arbusculares y Rhizobium: una simbiosis dual de interés.
A Fitter,J Garbaye (1994). Interactions between fungi and other soil organisms.
C García,M Franco,J Quintero,I Rao (2000). Catálogo de cepas de micorrizas arbusculares.
M Giovannetti,B Mosse (1980). AN EVALUATION OF TECHNIQUES FOR MEASURING VESICULAR ARBUSCULAR MYCORRHIZAL INFECTION IN ROOTS.
S Lizarraga,A Ruiz,S Salazar,Díaz Ricci,J Albornoz,P (2015). Micorrizas vesículoarbusculares, endófitos septados oscuros y anatomía radical en Fragaria ananassavar. Camino Real (Rosaceae), en la provincia de Tucumán, Argentina.
M Lugo,P Giordano,C Urcelay,E Crespo (2011). Colonización radical por endófitos fúngicos en Trithrinaxcampestris (Arecaceae) de ecosistemas semiáridos del centro de Argentina.
Elias Miranda,Orivaldo Saggin Júnior,Eliane Silva (2008). Seleção de fungos micorrízicos arbusculares para o amendoim forrageiro consorciado com braquiária.
Montenegro Gómez,S Barrera Berdugo,S Valencia,C (2017). Bioprospección de hongos micorrízicosarbusculares como alternativa para el fortalecimiento del cultivo de aguacate (Persea americana Miller) en Colombia.
Yuli Ordoñez Castañeda,Isabel Ceballos Rojas,Alia Rodriguez Villate,Ian Sanders (2021). Efecto de la inoculación Rhizophagus irregularis y de la fertilización fosfatada sobre la comunidad local de hongos formadores de micorrizas arbusculares.
Alexander. Perez C,Johanna. Rojas,Justo Fuentes (2010). Determinación de un modelo logístico para evaluación in situ de la colonizacion de micorrizas en pasto dichanthium aristatum (l).
R Peterson,H Massicote,L Melville (2004). Dark septate fungal endophytes.
J Phillips,D Hayman (1970). Improved procedures for clearing roots and staining parasitic and vesicular-arbuscular mycorrhizal fungi for rapid assessment of infection.
Laura Quiroz-Mojica,María Daza-Mendoza,Luis Díaz-Muegue,Aslenis Melo-Rios,Gustavo Peñuela-Mesa (2021). Efecto de biochar, micorrizas arbusculares y Guazuma ulmifolia, en la rehabilitación de suelos mineros.
Mayra Núñez-Aguilar,Elvira Brambila-Torres,Victoria González-Ramírez,Ferran Padrós-Blázquez (2015). Evaluación de la sintomatología depresiva y el estado de ánimo en futbolistas lesionados e ilesos.
Rodrigo Pérez,Yasna Tapia,Monica Antilén,Manuel Casanova,Catalina Vidal,Christian Santander,Humberto Aponte,Pablo Cornejo (2021). Interactive effect of compost application and inoculation with the fungus Claroideoglomus claroideum in Oenothera picensis plants growing in mine tailings.
Sánchez De Prager,M (2007). Las micorrizas: estrategia compartida para colonizar el suelo. Capítulo 4. Las Endomicorrizas: Expresión bioedáfica de importancia en el trópico.
A Sarasola,M Rocca (1975). Fitopatología. Curso moderno. Tomo IV. Fisiogénicas -Práctica en Fitopatología.
E Sieverding (1983). Proyecto Micorriza.
R Siqueiramartins,R Melloni,Guimarães Pereira Melloni,E (2017). Crescimento micelial de fungos micorrízicosarbusculares e formação de micorriza em solo contaminado por cádmio.
Sally Smith,David Read (1997). Vesicular-arbuscular mycorrhizas in agriculture and horticulture.
C Urcelay,P Tecco,F Chiarini (2005). Micorrizas arbusculares del tipo 'Arum' y 'Paris' y endófitos radicales septados oscuros en Miconiaioneuray Tibouchinaparatropica(Melastomataceae).
Rocio Gaiser,Carolina Robles,Natalia Skronski,Jesica Kobashigawa,Cecilia Carmarán (1952). HISTORICAL HERITAGE IN ANTARCTICA, THE CASA MONETA MUSEUM: A FIRST APPROACH TO THE CHARACTERIZATION OF XYLOPHAGOUS FUNGI AND THEIR POTENTIAL ROLE IN WOOD DAMAGE.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Di Barbaro Gabriela. 2026. \u201cNative Mycorrhizh-Forming Fungi Associated with Cultivated Forage Plants in the Central Valley Of Catamarca, Argentina\u201d. Global Journal of Science Frontier Research - D: Agriculture & Veterinary GJSFR-D Volume 22 (GJSFR Volume 22 Issue D1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.