Schrödinger’s “searchlight” made it possible to enlighten the atom and cast shadows on the “Screen of the Observer”. But the main trouble of the modern “Quantum Theory” is precisely that it mistook the shadows from the “Elephant” for the “Elephant” itself, for Reality. This led to a catastrophic discrepancy between the allowed calculated energy levels of electrons and the experimental ionization potentials with an increase in the mass of atoms. But the inclusion of “normal illumination” -UNDERSTANDING gives a REAL Quantum Theory, which can be built on the path laid by Planck-de Broglie-Einstein-Heisenberg-Bohm. Quantization adds a new INVARIANT and strictly mathematically, according to Planck, expands complements the Classics, and does not deny it -Resonant orbits of electrons in atoms -Electronic Orbitals correspond to resonant de Broglie waves. And Pontryagin’s Dualism of Functional Sets translates Schrödinger’s Uncertainty Principle into the Principle of CERTAINTY=OBSERVABILITY.
## I. INTRODUCTION
I introduced by Bohr in the planetary model of the atom, the orbits of electrons, he, without realizing it, himself pushed, moving away from the FOUNDATIONS of Quantization of his FOUNDERS - Planck and Einstein. Fascinated by the mysticism of the solutions of the Schrödinger equation, Bohr replaced his orbits, due to a poor understanding of mathematics [1], with interpretations of the "wave function". So, this mysticism entered their very canonized definition: Atomic orbital (electron orbital) is a one-electron wave function, $\psi$, obtained by solving the Schrödinger equation for a given atom; is given by the principal n, orbital I and magnetic m - quantum numbers [2].

$\odot$ 1993 International Union of Pure and Applied Chemistry and published for them by Blackwell Science Ltd Editorial Offices: Oseyd Mead, Oxford OX2 0EL 25 John Street, London WC1N 2BL 23 Ainslie Place, Edinburgh EH3 6AJ 350 Main Street, Malden MA 02148 5018, USA 54 University Street, Carlton Victoria 3053,Australia 10, rue Casimir Delavigne 75006 Paris, France Other Editorial Offices: Blackwell Wissenschafts-Verlag GmbH Kurfurstendann 57 10707 Berlin, Germany
Blackwell Science KK MG Kodenmacha Building 7-10 Kodenmacho Nihombashi Chuo-ku, Tokyo 104, Japan
First published 1988 Reprinted 1988 Reprinted as paperback 1989 Russian translation 1988 Hungarian translation 1990 Indian reprint edition 1990 Japanese translation 1991 Second edition 1993 Reprinted 1995, 1996, 1998
The Blackwell Science Logo is a trade mark of Blackwell Science Ltd, registered at the United Kingdom Trade Marks Registry
DISTRIButors
Marston Book Services Ltd
PO Box 269
Abingdon
Oxon OX14 4YN (0000)
1E 01235465500
Fax:01235463555)
### USA and Canada
CRC Press, Inc.
2000 Corporate Blvd, NW
Boca Raton
Florida 33431
Australia
Blackwell Science Pty Ltd
Carlton, Victoria 3053
(Orders: Tel: 393470300
Fax:393475001)
A catalogue record for this title is available from the British Library
#### ISBN 0-632-03583-8
Library of Congress
Cataloging in Publication Data
Quantities, units and symbols in physical chemistry prepared for publication by
Jan Mills...fet al.1-2nd ed.
P. C
At head of title: International Union of Pure and Applied Chemistry
International Union of Pund and Applied Chemistry, Physical
Chemistry Division,
Commission on Physicochemical Symbols,
Terminology, and Units-P. facing t.p.
Includes bibliographical references and index.
ISBN 0-632-03583-8
#### 1. Chemistry, Physical and
theoretical-Notation.
#### 2. Chemistry,
Physical and theoretical-Terminology.
## I. Mills, Ian (Ian M.)
## II. International Union of Pure and Applied
Chemistry.
## III. International Union of Pure and Applied
Chemistry.
Commission on Physicochemical Symbols,
Terminology, and Units
QD451.5.Q36 1993
### 541.3'014-dc20
But this definition of ORBITAL included in the ABC books, strictly speaking, is not a definition, but is its redefinition, moreover, it is looped, like a snake devouring its own tail. So, the whole explanation of Chemistry by "Quantum Mechanics" is built on the initially false definition [3]. Indeed, this "definition" does not appeal to the known measured waves - to the Heaviside-Maxwell electromagnetic wave quantized by Planck or to the acoustic wave quantized by Einstein, and not to the de Broglie matter wave, albeit somewhat mystical (yet), but experimentally observed, quantization which, as will be shown, gives the resonant orbits of electrons. No, this "definition" appeals to a fundamentally UNOBSERVABLE "wave function". Those without losing the inherent "meaning", this "definition" (with the same success and with the same errors) could be rewritten and vice versa: "Wave function is a one-electron atomic orbital (electron orbital)". But the physicists who gathered in Copenhagen over a bottle of cheap port came up with "their" special name for the "wave function", in order to somehow tie it to reality, "their own" special name: probability density. But they didn't tie it tightly - Schrodinger turned out that it could be not only non-zero, but also maximum inside the nucleus! There are other reasons indicating that the "wave function" has nothing to do with the probability density [4]. And in general, it turned out the same as when a reference to the Principle of Causality is simply masked, the reason for something is not understood. If we talk about the probability density, then we must add - WHAT! So the probability density of the kinetic energy of molecules in a gas can also describe a sound wave. The probability density can be compared not only to electromagnetic waves, but also to the static Coulomb-Newton Laws [4], if, in accordance with the principle of Logarithmic Relativity, we compare to it the subparticles that form these fields, and the substructure of the corresponding fields, even if so far unmeasured by us. And for electron orbitals, if we talk about the probability density of the distribution of an electron in an orbit, then we must honestly say - WHAT! What parameter? And do not hide behind the words UNKNOWN WHAT, when the calculations give the maximum "wave" function of the electron inside the nucleus.
So, at the dawn of the last century, the times of Newton, when the Physicist and the Mathematician were in the same vial of Reality, have already passed. And their misunderstanding of some aspects of Classical Physics [5], multiplied by the misunderstanding of the Planck-Einstein QUANTUM, the developers of the "Quantum Theory" hid behind an imaginary unit from the mat. Heaviside's physics, which was also not fully understood, having supplemented the picture for "completeness" with "unobservable zero-point oscillations."
And, thus, they turned the entire "Quantum Mechanics" into a schizophrenic Game of the Mind, cut off from Reality. And now it took a hundred years for the intoxicating "charm" of mysticism in the ELEMENTARY Harmonic Oscillator, which, in fact, was not even strictly analyzed [6], passed and a heavy hangover with "Black Holes" and "Particles of God" set in. Ate to drink, it's better still a good cognac. And on a "sober head" it becomes obvious that the primary quantization of spatial waves in extended media is done correctly - according to the Principles laid down by Planck and Einstein. This determined the progress of the Quantum Theory of Solids. But the "quantum" transition to local fluctuations has not been carefully worked out in the basics. And he, even in the simplest case of the Ideal Harmonic Oscillator, gives mystical ("immeasurable") ZERO vibrations. So both "Secondary Quantization" and Atomic Physics and after them, Quantum Field Theory for a hundred years have been engaged not in correcting the Basic Models [7], but, in fact, in sucking corrections out of the finger, exceeding what the corrections correct.
That is why the really observed DUALISM [8] even of Planck's light quanta, and even more so of de Broglie particles, was actually attributed to mysticism [9, 10], which does not need to be UNDERSTAND, but must be ACCEPTED.
Whereas the first coherent waves arising due to Quantization, into which the packets of de Broglie waves that fill the particle are converted, Nature "invented" in the form of electron orbitals long before people mastered radio waves and invented the maser laser, where the property of Bose particles is manifested be in the same state without limiting their number. And the manifestation of both the Pauli principle and the same property of Bose particles can be easily shown using the example of electron orbitals, but not at all Schrödinger, but the original ones - Bohr. But first, let us show that the opposition of the classical Bohr orbits to quantum physics is far-fetched, simply dictated by the struggle for the priority of the "invention" of the QUANTUM. Bohr or Planck? Half a century later, Pontryagin's dualism of functional sets "reconciled" them. But the canonization of Schrödinger's "shadows", despite the catastrophic discrepancies between calculations based on "shadows" and experiment, prevented us from noticing the difference between the shadow of the Elephant and the Elephant for another half a century.
## II. CLASSIC ORBITAL
It follows from Classical Physics that in the hydrogen atom the centrifugal force acting on the electron is equal to the force of the Coulomb attraction of the electron and the nucleus. So the force acting on an electron in orbit is the centrifugal force
$$
F _ {C} = m \omega^ {2} r = m \frac {v ^ {2}}{r} \tag {1}
$$
and the force of the Coulomb attraction of the electron to the hydrogen nucleus
$$
F _ {1 1} = \frac {e ^ {2}}{4 \pi \varepsilon_ {0} r ^ {2}} \tag {2}
$$
From the equality of these forces acting on an electron in orbit, we obtain the dependence of the electron velocity in orbit on its radius (Fig. 1)
$$
m \frac{v ^ {2}}{r} = \frac{e ^ {2}}{4 \pi \varepsilon_ {0} r ^ {2}} \Rightarrow v _ {1} ^ {2} = \frac{e ^ {2}}{4 \pi \varepsilon_ {0} r _ {1} m} \Rightarrow v _ {1} = \pm \frac{e}{2 \sqrt{\pi \varepsilon_ {0} m}} \frac{1}{r _ {1} ^ {1 / 2}}
$$
$$
\frac{v_{1}}{c} = \pm \frac{e}{2c\sqrt{r_{1}\pi\varepsilon_{0}m}} \frac{1}{(r/r_{1})^{1/2}} = \pm 0.007291686044471947 \frac{1}{(r/r_{1})^{1/2}}
$$
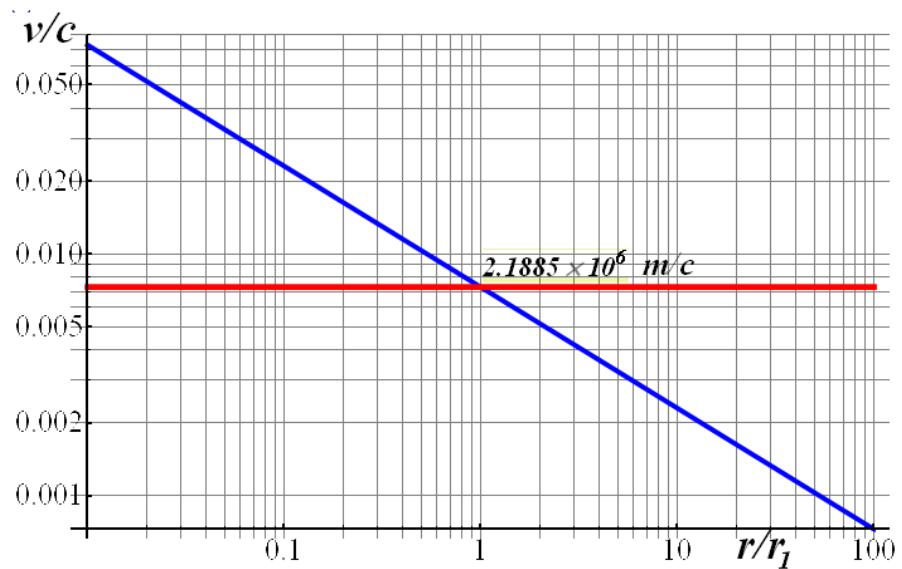 Fig. 1: The dependence of the relative (reduced to the speed of light) velocity of the steady motion of an electron in the orbit of a hydrogen atom on the relative radius of the electron orbit, reduced to its experimental radius (blue line):
$$
\begin{array}{l} e = - 1. 6 0 \cdot 1 0 ^ {- 1 9} C, \\m = 9. 1 1 \cdot 1 0 ^ {- 3 1} k g, \\\varepsilon 0 \approx 8. 8 5 1 0 ^ {- 1 2} \mathrm {m} ^ {- 3} \mathrm {k g} ^ {- 1} \mathrm {s} ^ {4} \mathrm {A} ^ {2}, \\r _ {1} = 5 3 1 0 ^ {- 1 2} \mathrm {m} \\\end{array}
$$
The resulting dependence of the electron speed, as well as the speed reduced to the speed of light, are determined not only by world constants, but also by the absolute value of the orbit radius. So, using the tabular value of the size of the hydrogen atom as the diameter of the orbit, we get the red line shown in Fig. 1, a quite reasonable value for the speed of stable rotation of an electron in orbit (the reduced radius is used on the graph). But relatively weak influences can take the electron away from the initially given radius of the classical orbit to neighboring ones.
In this orbit, the potential (Coulomb) energy of an electron, if we set it to zero for an electron at infinity, is equal to
$$
U _ {1 1} = \frac {e ^ {2}}{4 \pi \varepsilon_ {0} r _ {1}} \tag {5}
$$
With the speed obtained in formula 3, the kinetic energy of an electron in this orbit is equal to half the potential:
$$
E_{c}=m\frac{v_{1}^{2}}{2}=m\frac{\left(\frac{e}{2\sqrt{\pi\varepsilon_{0}m}}\frac{1}{r_{1}^{1/2}}\right)^{2}}{2}=\frac{e^{2}}{8\pi r_{1}\varepsilon_{0}}=\frac{U_{11}}{2}=I_{1}\square13.55eV
$$
and, of course, is also equal, as shown in Fig. 2, to the energy of the electron's exit into vacuum.
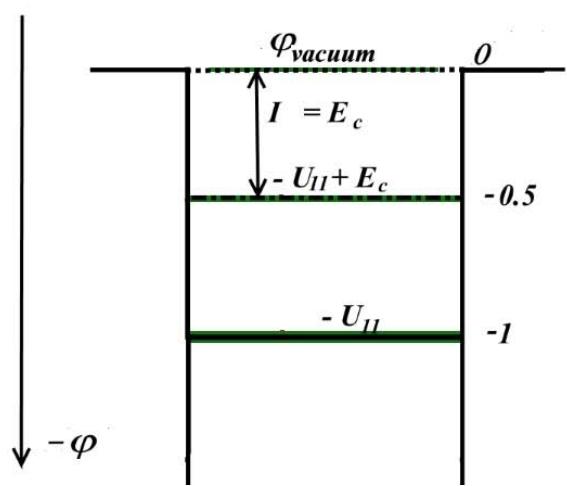
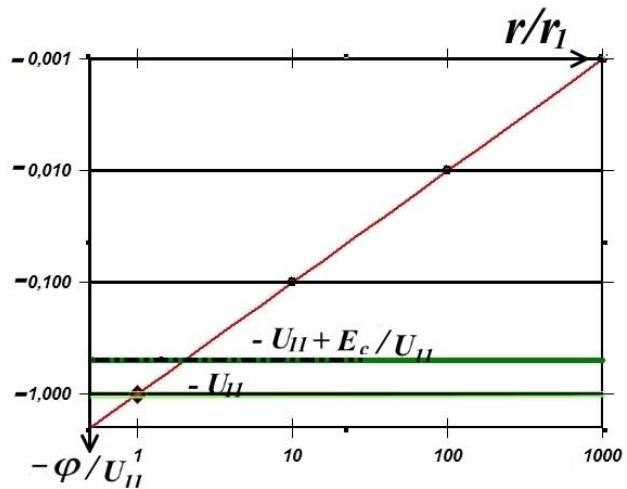 Fig. 2: Electron energy level diagram on a linear scale (left) and on a logarithmic scale (right) the edge of the potential Coulomb well (red line) with exponentially decreasing equipotentials with the electron energy level superimposed on them (green line).
At the same time, the purely classically obtained numerical value of the electron work function (6), which is in good agreement with the tabular value of the Ionization Potential for the hydrogen atom - 13.595 eV.
The whole question is: Why did the electron "choose" from a continuous series of orbits exactly the radius of rotation that corresponds to the ionization Potential of hydrogen? Although it is likely that the "choice" of this "quantum" radius was simply hidden behind the classical calculation, based on the most experimentally determined value of the ionization Potential.
So, in the classical representations for the hydrogen atom, as well as in space calculations, we have a continuous series of orbits, with an easy transition from one to another. And the first cosmic velocity for the Earth's satellites is calculated simply on the basis of their minimum radius, equal to the radius of the Earth.
But the "cosmic" velocities of an electron can be tried to be calculated in the model of particle rotation around an infinitely small point. In the simplest particular case - at a fixed speed of rotation of an electron around the nucleus (which, for clarity, we assume equal to unity), the trajectory of its movement relative to the nucleus is described by a modified Archimedes spiral, the radius of which is determined by the ratio of its "start from the nucleus" speed and the total radial acceleration: the balance of the centrifugal forces and forces of the Coulomb attraction of the electron to the nucleus and something else, which gives the binding of the calculated values to the experimentally observed values of the Ionization Potential in the resonant orbit:
$$
r (\theta) = v _ {0} - \frac {a \theta^ {2}}{2} \tag {7}
$$
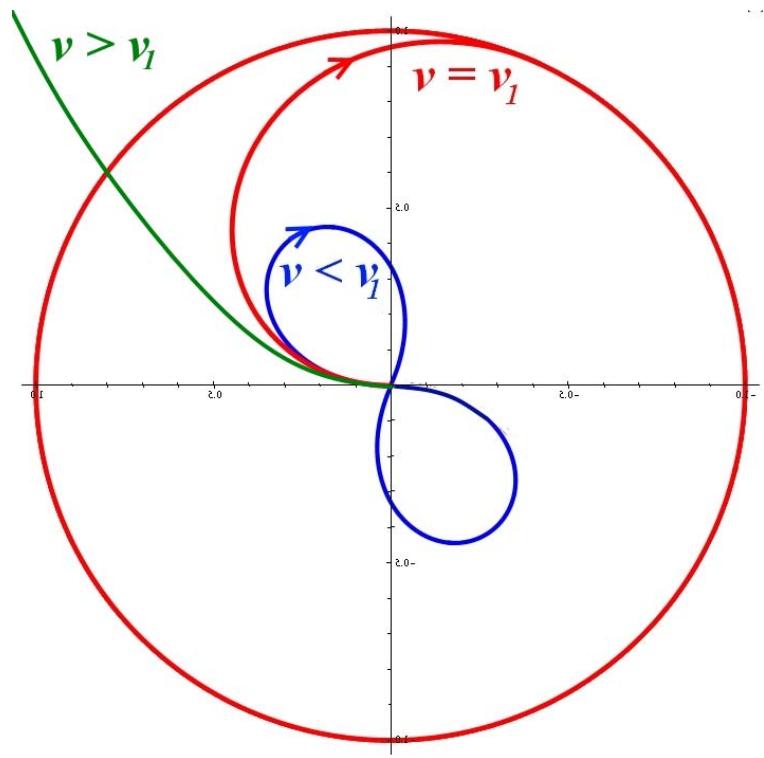 Fig. 3: The trajectories of an electron that "started from the nucleus" at different speeds
As can be seen from Figure 3, the electron at low speeds will return to the center of rotation and (if it can) skip it. It enters a stationary circular orbit at an initial speed equal to the speed of movement along the orbit, which is its "first cosmic speed". At high starting speeds, the electron also goes into a stationary, but elliptical orbit. And when the "second/third" cosmic velocity is reached, the electron becomes free - it goes to infinity and no longer returns to the nucleus.
We will not carry out this model calculation in detail, since the model with an infinitely small rotation point is idealized and, in accordance with the Principle of Relativity, must also take into account the limitation of the electron speed by the speed of light, while the speed obtained above on the basis of classical concepts is far from the speed of light. But the obtained value for the speed of an electron in a stationary circular orbit (Fig. 1.) is quite real and is "the first space velocity for a hydrogen electron. So the reason for the "quantization" of orbits proposed by Bohr - the stabilization of only a few "selected" orbits from the entire series of orbits - must be sought in something else, and not in the limitation of speed by the Principle of Relativity.
But the value of the speed on the orbit itself was obtained from its radius, which is set equal to the radius of the hydrogen atom (which, of course, is a tautology). And, as will be shown below, the calculation path indicated by Planck gives even without "Schrödinger shadows" not an abstract one, which, as shown in the work, leads to a catastrophic discrepancy between the calculated allowed energy levels and the First Ionization Potentials with an increase in the mass of atoms [11], but a real the value of the "chosen" radius, consistent, as will be shown later, with the experimental values of the Ionization Potentials.
And, thus, Bohr's orbital model of the atom is restored in rights!
## III. QUANTUM ADDITION TO THE CLASSICS
From the equality of the forces acting on an electron in orbit, in the classical consideration, similarly to formula 3 (or simply by dividing formula 3 by the radius), we can also obtain the dependence of the frequency of its rotation around the nucleus on the radius
$$
m \omega^ {2} r = \frac {e ^ {2}}{4 \pi \varepsilon_ {0} r ^ {2}} \Rightarrow \omega_ {1} ^ {2} = \frac {e ^ {2}}{4 \pi \varepsilon_ {0} r _ {1} ^ {3} m} \Rightarrow \omega_ {1} = \frac {e}{2 \sqrt {\pi \varepsilon_ {0} m}} \frac {1}{r _ {1} ^ {3 / 2}} \tag {8}
$$
And so, the classical consideration gives one functional relationship of either the electron velocity in the orbit, or the rotation frequency with its radius, using the experimental value of which, according to Bohr, we simply postulated their separation from the series as stationary.
Now we use the Planck equation and the Planck constant to relate to the frequency of light equal to the energy of the ionization Potential, which, according to formula 6, is equal to the kinetic energy of an electron in orbit
$$
I _ {1} = \hbar \omega_ {P h 1} = E _ {C} = m \frac {v _ {1} ^ {2}}{2} = m \frac {\omega_ {1} ^ {2} r _ {1} ^ {2}}{2} \tag {9}
$$
This equation complements the classical ones and, thus, gives an additional connection between the rotation frequency and the radius of the orbit, if the light quantum correlates with the rotation frequency of the electron in orbit. And, thus, if we ASSUME this correlation of frequencies, then it is possible to obtain a single value of both the frequency and the radius of the classical orbit.
In particular, if we assume that these frequencies are equal, then from (9) we obtain, in addition to equation (8), one more equation relating the frequency to the radius.
$$
\omega_ {1} = \frac {2 \hbar}{m r _ {1} ^ {2}} \tag {10}
$$
And equating the frequencies, we get one equation with one unknown radius:
$$
\frac {2 \hbar}{m r _ {1} ^ {2}} = \frac {e}{2 \sqrt {\pi \varepsilon_ {0} m}} \frac {1}{r _ {1} ^ {3 / 2}} \tag {11}
$$
The solution of the resulting equation gives us the only value of the radius of the RESONANT orbit of an electron in the hydrogen atom
$$
r _ {1} = \frac {1 6 \pi \hbar^ {2} \varepsilon_ {0}}{e ^ {2} m} = 2 1 2. 1 4 \mathrm {p m} \tag {12}
$$
So, the World Constants, supplemented by Planck's constant, uniquely DETERMINE the radius of the RESONANT orbit of an electron in a hydrogen atom. But the obtained value of the radius of the orbit is approximately 4 times larger than the radius of the hydrogen atom obtained from the experiment.
If we assume that the frequency of a light quantum is equal to HALF the frequency of rotation of an electron in orbit, then instead of equations (9-10) we get
$$
\hbar \frac{\omega_1}{2} = m \frac{\omega_1^2 r_1^2}{2} \Rightarrow \omega_1 = \frac{\hbar}{m r_1^2} \tag{13}
$$
And instead of equation (11) we have
$$
\frac {\hbar}{m r _ {1} ^ {2}} = \frac {e}{2 \sqrt {\pi \varepsilon_ {0} m}} \frac {1}{r _ {1} ^ {3 / 2}} \tag {14}
$$
And, then, we obtain the expression of the radius of the orbit determined by the World Constants, which gives a value that coincides with the experimental value of the hydrogen radius given under Figure 1 with good accuracy:
$$
r_{1} = \frac{1}{4} \frac{16 \hbar^{2} \pi \varepsilon_{0}}{e^{2} m} = 53.03 \mathrm{pm} \tag{15}
$$
At the same time, taking into account that the particle is an electron, it is a packet of waves, it is natural to assume that the resonant orbit corresponds to a resonant wave. Plasma, resonant, i.e. standing fluctuations in the charge density lead to the formation of charged layers at the boundaries of the resonator. But there are no boundaries in a circular orbit and, consequently, there are no areas of charge accumulation - there is no dipole. Therefore, again, it is natural that this resonance wave does not give a charge to the radiation of an electromagnetic wave. And the dipole arises only between the entirely displaced orbit and the nucleus. So the excitation of the entire electron orbit is PARAMETRIC, as it should be at half the frequency of its intra-orbital resonance!
So, in fact, the rejection of the Bohr orbital model of the atom was incorrectly "justified" by the instability of the (energy) of this electron orbit due to electromagnetic radiation. But, taking into account the corpuscular-wave dualism, the de Broglie wave packet of an electron in a resonant orbit also enters into resonance - in a self-coherent state, i.e. the electron becomes a resonant undamped wave, which, as noted above, does not have a dipole and does not radiate at the resonant frequency.
And taking into account the obtained parametric relationship between the frequencies of rotation of an electron in a resonant orbit and the quantum of light exciting it from the orbit, one can also imagine the parametric relationship of the de Broglie wave with an electromagnetic wave. In this case, the de Broglie wavelength of an electron in a resonant orbit is determined by the circumference of this orbit
$$
\lambda_{dB} = 2\pi r_{1} = \frac{8\hbar^{2}\pi^{2}\varepsilon_{0}}{e^{2}m}
$$
So the ideal (undamped) resonance of the de Broglie wave DOES NOT radiate energy, if the Planck quantum for electromagnetic waves is extended not only to acoustic (as Einstein did), but also to the de Broglie waves of the electron.
## IV. PRINCIPLES OF FORMATION OF MULTIELECTRON ORBITALS
The representation of an electron orbital as a packet of de Broglie waves of an electron transformed into a resonant wave gives a simple explanation for the Pauli principle. Given that orthogonally polarized (charge) waves do not repel each other, it can be assumed that two electron waves can simultaneously be in the same orbit as one particle with doubled mass and doubled charge. This corresponds to the Pauli principle for electrons as particles. Therefore, for this pair of electrons in orbit, the centrifugal force, in contrast to formula (1), is equal to
$$
F _ {C} = 2 m \omega^ {2} r = 2 m \frac {v ^ {2}}{r} \tag {17}
$$
And the force of the Coulomb attraction of a pair of electrons in this orbit to a nucleus with a double charge (helium atom) is equal to
$$
F_{22} = \frac{e^{2}}{\pi\varepsilon_{0} r^{2}}
$$
From the equality of forces in the orbit of this pair of electrons, similarly to formula (3), but now using the radius of the helium atom $32 \times 10^{-12}$ m, we obtain a large, but also quite reasonable speed of movement of this pair of electrons along the orbit of the order of a percent from the speed of light
$$
v _ {2} ^ {2} = \frac {e ^ {2}}{2 \pi \varepsilon_ {0} r _ {2} m} \Rightarrow v _ {2} = \pm \frac {e}{\sqrt {2 \pi \varepsilon_ {0} m r _ {2}}} = \pm 3 9 7 8 5 6 6. 4 m / s = \pm 0. 0 1 3 3 \cdot c \tag {19}
$$
Wherein, again, the classical consideration gives the first functional relationship between the speed of a pair of electrons in an orbit and its radius and the total potential energy of this pair of electrons
$$
U_{22} = \frac{e^{2}}{\pi\varepsilon_{0} r_{2}} = 179.548356\,eV
$$
where one electron accounts for half of the potential energy of the pair:
$$
U _ {2 1} = \frac {U _ {2 2}}{2} = \frac {e ^ {2}}{2 \pi \varepsilon_ {0} r _ {2}} = 8 9, 7 7 4 2 e V \tag {21}
$$
At a higher speed in the orbit of a pair of electrons from formula (19), the kinetic energy of a pair of electrons in this orbit naturally also increases, but again it is exactly half of the potential energy of a pair of electrons:
$$
E _ {c} = 2 m \frac {v _ {2} ^ {2}}{2} = 2 m \frac {\frac {e ^ {2}}{2 \pi \varepsilon_ {0} r _ {2} m}}{2} = \frac {e ^ {2}}{2 \pi r _ {2} \varepsilon_ {0}} = U _ {2 1} = I _ {2 2} \tag {22}
$$
So the energy diagram for a pair of electrons, and for each electron of this pair, is completely similar to the diagram shown in Fig. 2. And to pull out a pair of electrons from this level, you need eV, which is close to the Total Ionization Energy attributed to helium $-79.005151042(40)$ eV.
And in order to pull out one electron from this orbit, half of this ionization Energy of the pair is required - 44.8871 eV. But this energy value is almost twice the tabular value of the First Ionization Potential of helium 24.47 eV.
It also follows from the above reasoning that the level of the remaining one electron of the helium atom decreases after primary ionization, and during ionization it needs to additionally overcome the Coulomb force of the nucleus charged by the first ionization. But for the time being, we will not be distracted by these arguments, which may and will help explain the discrepancies in the energy levels of the helium atom, which is more complex than hydrogen. And immediately we will use the quantum addition to the classical calculations of the orbital of a pair of electrons on the basis of its energy diagram obtained.
To do this, first again, from the equality of forces acting on a pair of electrons in orbit, in the classical consideration, we can obtain, similarly to formula (8), in addition to (19), the dependence of the frequency of rotation of this pair around the nucleus on the radius of the pair orbit
$$
2 m \omega^ {2} r = \frac {e ^ {2}}{\pi \varepsilon_ {0} r ^ {2}} \Rightarrow \omega_ {2} ^ {2} = \frac {e ^ {2}}{2 \pi \varepsilon_ {0} r _ {2} ^ {3} m} \Rightarrow \omega_ {2} = \frac {e}{\sqrt {2 \pi \varepsilon_ {0} m}} \frac {1}{r _ {2} ^ {3 / 2}} \tag {23}
$$
We use the Planck equation and the Planck constant to relate to the frequency of light, the quantum of which is equal to the energy of the ionization Potential and which, according to formula (22), is equal to the kinetic energy of a pair of electrons in orbit
$$
I _ {2 2} = \hbar \omega_ {P h 2} = E _ {C 2} = 2 m \frac {\nu_ {2} ^ {2}}{2} = m \omega_ {2} ^ {2} r _ {2} ^ {2} \tag {24}
$$
This equation complements the classical ones and, thus, gives an additional connection between the rotation frequency and the radius of the orbit. And, thus, if we ASSUME that the frequency of a light quantum correlates with the frequency of light, it makes it possible to obtain a single value of both the frequency and the radius of the classical orbit.
In particular, if these frequencies are equal, then we obtain, in addition to equation 8, another equation relating the frequency to the radius.
$$
\omega_ {2} = \frac {\hbar}{m r _ {2} ^ {2}} \tag {25}
$$
And equating the frequencies, we again get one equation with one unknown radius:
$$
\frac {\hbar}{m r _ {2} ^ {2}} = \frac {e}{\sqrt {2 \pi \varepsilon_ {0} m}} \frac {1}{r _ {2} ^ {3 / 2}} \tag {26}
$$
The solution of the obtained equation gives us the only value of the radius of the RESONANT orbit of a pair of electrons in a helium atom, which is 8 times less than the radius of an electron orbit in hydrogen
$$
\frac{\hbar}{e} \sqrt{\frac{2\pi\varepsilon_0}{m}} = r_2^{1/2} \Rightarrow r_2 = \frac{2\pi\varepsilon_0 \hbar^2}{m e^2} = \frac{1}{8} r_1 = 26.52\mathrm{pm} (27)
$$
This one, obtained from the World Constants, with equal frequencies of light and rotation of a pair of electrons per orbit, e, the radius of the circular orbit of rotation of a pair of electrons $26.52\mathrm{pm}$ is close to the radius of helium $32\mathrm{pm}$. Consequently, the parametric coupling is weaker, and, as already noted, the dipole of electron waves does not appear on the orbit. Therefore, the excitation of the entire pair of electrons is unlikely. But we will not be special consideration of the Second Ionization Potentials in this work. We simply note that the use of the quasi-nuclear model for a completely filled shell of a pair of electrons makes it possible, even with an increase in the atomic number, to estimate from the first principles both the radius of the additional electron shell and the energy level corresponding to it, as approximately equal to their values in the hydrogen atom. This numerically differs from the experimental values by tens of percent (Fig. 4, blue dotted lines), which is natural in the first approximation by the quasi-nuclear model, which does not take into account the repulsion of the upper electron by the filled inner shell. But even these discrepancies, which can be seen how to eliminate, are not catastrophic, by orders of magnitude, which gives the Schrödinger equation [3, 11].
The quasi-nuclear model with the calculation algorithm described above gives a significant jump in the size of the atom and its First Ionization Potential even when one more electron is added to the completely filled named c-shell. And here there is a relatively small change in the atomic number and radius of the orbit of the added outer electron, and its First Ionization Potential (Fig. 4, red dotted lines). But for a more rigorous calculation than the quasi-nuclear model, according to the described algorithm, of course, it is necessary to take into account the symmetry of the tetrahedron of electron shells when the c-shell is completely filled for the second and third periods, and their even more complex symmetry for the following periods, which manifests itself during the transition from - shells to the so-called p-shell (green dotted curves in Fig. 4), where the equivalent orbitals form, as previously shown, a tetrahedron.
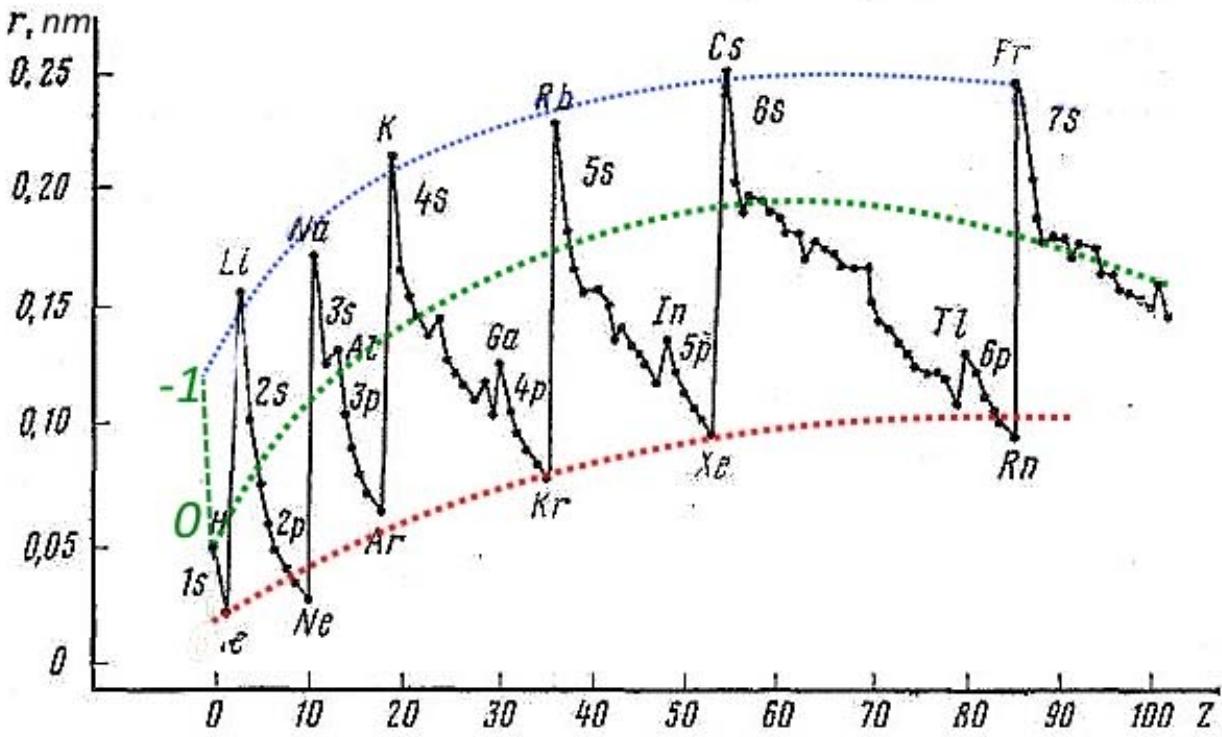
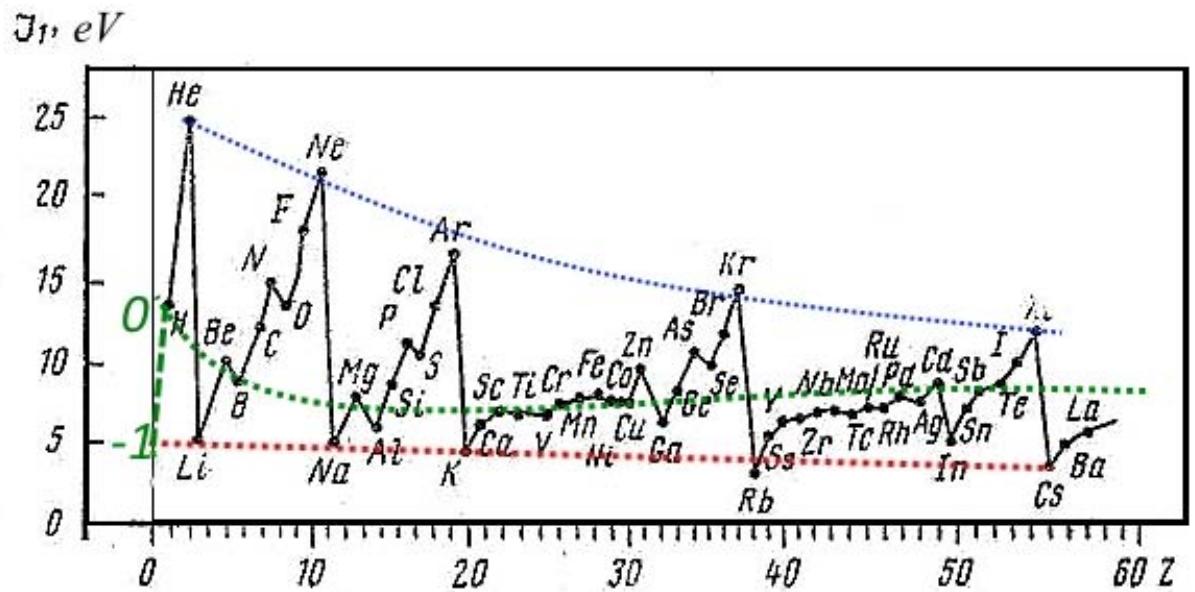 Fig. 4: Dependence of the radius of the outer electron shell and its First Ionization Potential on the atomic number (Zero denotes the "zero" element used in the construction of the periodic table by Mendeleev, and minus one is an additional conditional element with minus one electron) And so, as it should be, correctly chosen Basic Models give/describe the correct order of observed phenomena, in contrast to erroneous, but Canonized Models, which give catastrophic discrepancies with experiment and are suitable only for decoration and deception of the layman.
And the algorithm described in this paper makes it possible to obtain the "Second" Principle from the First Principles - how all electron orbitals and their corresponding levels of allowed energies are formed in an atom with any atomic number:
The deepest orbital and its energy level, the deepest, are formed in the Coulomb field of the nucleus of the field from the maximum number of equivalent orbitals satisfying point spatial symmetry (this number of electrons of the inner shell is less than or equal to the total number of electrons of the atom).
The remainder of the total number of electrons, in turn, is divided by the maximum number of electrons for reduced symmetry, which form the next higher allowed energy level.
So, in a recurrent way, using the quasi-nuclear model, it is possible to calculate from first principles - from bottom to top, all the electronic levels of different atoms, from an atom with a completely filled electron shell to atoms by filling a new shell with electrons with a quasi-nucleus from the nucleus and the last fully filled shell. Only for a quantitatively rigorous calculation it is necessary to take into account the degree of screening of the nucleus by deeper filled shells, which, of course, is higher for the lowest shell and is enhanced by subsequent overlying shells and is minimal for the upper shell from one orbit with two electrons.
Generating HTML Viewer...
References
11 Cites in Article
Stanislav Ordin (2024). Chaos—Imaginary Ostensibility—Orthogonality.
Ian Mills,Tomislav Cvitas,Nikola Klayshomman,Kozo Kallay,Kuchitsu (2012). Quanities, Units and Symbols in Physical Chemistry.
S Ordin (2020). Frontier Chemistry Aspects.
Marat Vladimirovich,Guryev (2017). Wave Function Is not Amplitude of Probability Density.
Stanislav Ordin (2019). Quantization Essence.
Stanislav Ordin (2021). Non-Elementary Elementary Harmonic Oscillator.
Stanislav Ordin,Book (2017). Refinement of basic physical models.
Stanislav Ordin (2024). Analysis of Newton's Elementary Particle.
Stanislav Ordin (2022). Dualism of newton's elementary particle.
Stanislav Ordin (2022). Imagination and real quantization.
Stanislav Ordin (2018). Quasinuclear Foundation for the Expansion of Quantum Mechanics.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Dr. Stanislav V. Ordin. 2026. \u201cNon-Schrödinger Orbitals\u201d. Global Journal of Science Frontier Research - A: Physics & Space Science GJSFR-A Volume 24 (GJSFR Volume 24 Issue A2).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.