## I. INTRODUCTION
The pulmonary zygomycosis is an unusual fungi infection which occurs mostly in immunodepressive patients $^{11}$. In the CGD, the lung infection from fungi etiology is the one with higher prevalence, with the Aspergillus fungus as the more prominent reason $^{2}$.
Our report describes a patient with CGD plus pulmonary zygomycosis.
## II. CASE REPORT
Male patient, five years, coming from Alagoin has-BA, admitted at the hospital Martagão Gesteira (HMG) in Salvador-BA, diagnosed with pneumonia for five months and not responding to large spectrum antibiotic therapy. The patient developed fever and dyspnea five months back; he had received previous medical treatment in his hometown with the following antibiotics in sequence: Ceftriaxone and Oxacillin, Azithromycin, Tazocin, Sulfamethoxazole (plus Trimethoprim, Vancomycin, Meropenem and Fluconazole). The recurrent fever and tachypnea persisted, only the respiratory discomfort showed improvement.
The genitor denied knowledge on the disease although reported previous recurrent pneumonia since the patient was two-years old and abscesses episodes (1 scalp abscesses when newborn; 1 perianal and 1 cervical) that in turn led to many hospitalizations and previous use of antibiotics. Two maternal cousins were reported deceased in the family.
Regarding physical examination, low stature (Z-2 Height) and very thin (BMI Z-2), presenting tachypnea (40 BPM), SaO2 at $92\%$ in room air, widely distributed murmur with crackles, mild intercostal retractions.
Examinations results held at reference hospital:
Computerized tomography (CT) of the chest (FIGURE 1)
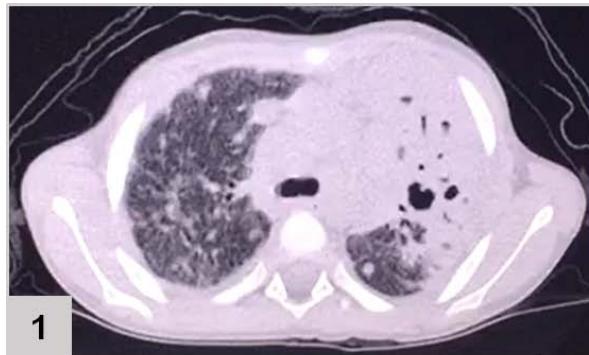
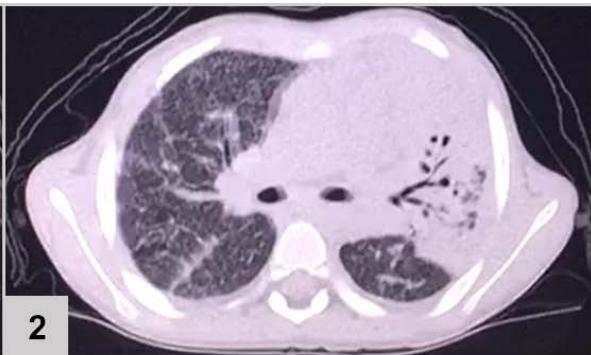
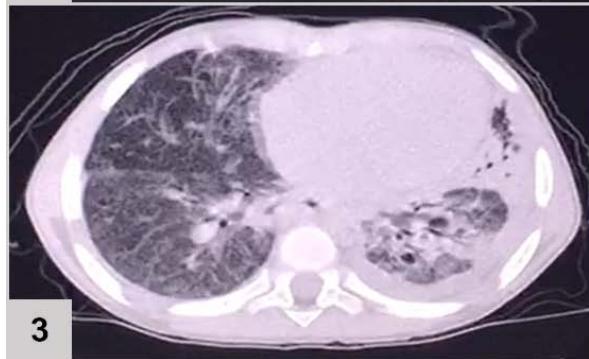
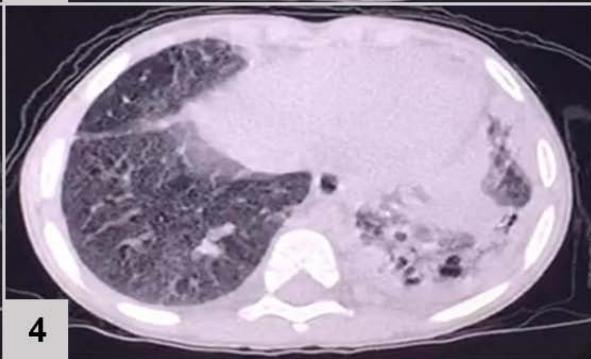 Figure 1: CT of the chest results: extensive consolidation, with air bronchograms and cystic images in between, in the left lung (3 and 4); ground-glass opacities, diffusion in the lower left lobe (1 and 2); nodular opacity, no sign of calcification in between, on both lungs (1, 2 and 3); reticular and linear opacity and parenchymal bands in the upper lobes (3 and 4); small pleural effusion on the left (1 and 2); adjacent subsegmental atelectasis (2 and 4)
Hemogram: hemoglobin: 10g/l; White blood cell count: 9400/mm3; lymphocytes: 1692/mm3; neutrophils: 7144/mm3; eosinophil: 188/mm3; platelets: 425.000/mm3. Negative blood culture. PPD no reaction. Normal range for Alpha-1 antitrypsin. Negative HIV. Normal sweat chloride values.
Primary immune evaluation (dose of complement, serum immunoglobulin and immune-phenotyping) all set in normal values for the patient's age.
Lungbiopsy (FIGURE 2)
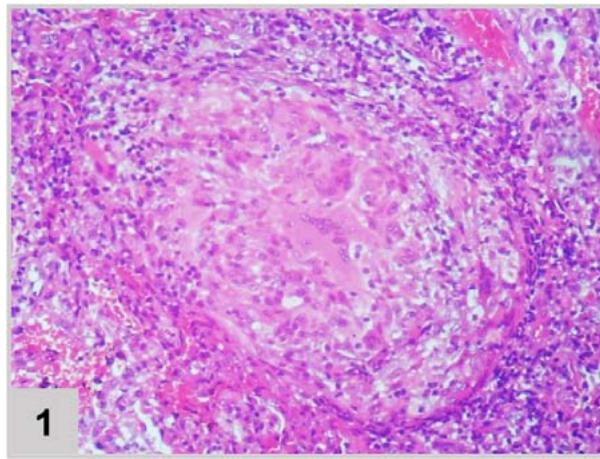
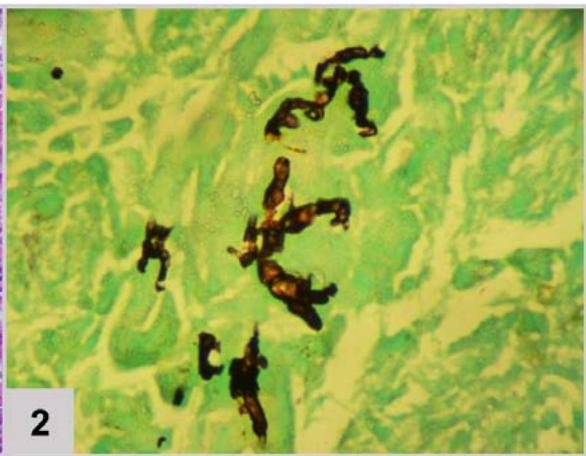 Figura 2: Lung biopsy (200x magnification). (1) Pulmonary tissue in hematoxylin and eosin showing granulomatous reaction with epithelioid histiocytes and multinucleated giant cells. (2) Pulmonary tissue in Grocott's stainrevealing fragments of broad, wavy and short hyphae, matching Zygomycosis by mucorales, longitudinal and cross-sectioned With the fungal lung infection diagnosis (zygomycosis), without previously response to Fluconazole, Amphotericin B was administered. The fever disappeared and there was no need for supplemental oxygen, the patient was discharged from the hospital with mild tachypnea.
With the medical record of pneumonia and recurrent abscesses, as well as fungal pneumonia, Bactrim and Itraconazole were prescribed as a prophylactic measure and dihydrodorhodamine test (DHR) was ordered to evaluate the oxidase burst of phagocytes.
DHR test has revealed no sign of oxidation after stimulation with phorbol myristate acetate, PMA (from
59,2 to 48,1) has shown in comparison with the threshold an increase (from 34,7 to 91,6) which in turn confirmed the Chronic Granulomatous Disease (CGD) diagnosis. The antibiotic and antifungal prophylaxis has demonstrated good evolution and weight-stature gain (reverting FTT), the patient is listed for transplant.
## III. DISCUSSION
The CGD is an innate immunity disorder that is inherited, mostly characterized as an X-linked recessive mode, marked by the presence of two early childhood deaths of first-degree male cousins in the patient's family history (FIGURE 3).
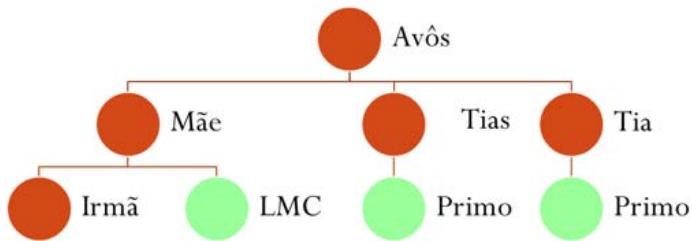 Figure 3: Patient Family history showing two childhood deaths of first-degree male cousins
This transmission pattern has been present in approximately $67\%$ of cases, more prominent and susceptible in male sex, linked to mutations in the CYBB gene, located on locus Xp21.1, which cause a functional deficit of gp91-phox, an enzyme complex component NADPH oxidase of phagocytes. This protein complex is responsible for producing oxygen metabolism intermediates in phagocytes, including oxidants with antifungal and antibacterial action.
Phagocytes, neutrophils, monocytes, macrophages and dendritic cells are cells capable of binding to pathogens and providing phagocytosis of these, through trapping within a phagosome. Several mechanisms, such as the release of lysozymes, acid hydrolases and toxic oxidants, are used by these cells of the immune system to destroy the pathogen. To carry out the production and release of oxidative compounds, phagocytic cells are able to assemble NAPH-oxidase complexes in the phagolysosome membrane, which are responsible for catalyzing the production of these compounds[^13]. The protein complex provides the generation of $\mathsf{O}^{2-}$, which is unstable and has a weak bactericidal action, through the transfer of electrons from intracellular NADPH molecules to molecular oxygen. The generated $\mathsf{O}^{2-}$ will later undergo dismutation reactions, catalyzed by the enzyme superoxide dismutase (SOD), which will convert it into hydrogen peroxide $(\mathsf{H}_2\mathsf{O}_2)$. Finally, the enzyme myeloperoxidase (MPO) present in primary granules (mainly from neutrophils) is released into the phagosome and converts $\mathsf{H}_2\mathsf{O}_2$ into oxidants with great bactericidal power, reactive oxygen species (ROS), such as hypochlorous acid and radicals free (such as OH<sup>-</sup> and $\mathrm{O}^{-})^{14}$. After activation and assembly of the complex, the phagocytic cell enters a transient stage of oxidative explosion, where there is a significant increase in oxygen consumption $^{13}$.
Thus, CGD patients possess dysfunctional neutrophils and incapable oxidative burst, which in turn disable catalase-positive and intracellular microorganisms' elimination $^{11}$. There is also granuloma formation, mostly sterile, a characteristic that may be directly connected to the hyperinflammatory state common in CGD patients $^{10}$. Granuloma is a means of containing the proliferation of the microorganism that is difficult to eliminate and isolating it from the rest of the organism $^{10}$.
Morphologically, granulomas in patients with CGD, in general, have the following histopathological organization:
- Neutrophils located predominantly in the central portion, with extremely reduced activity and some even in the apoptotic phase.
- Lymphocytes located both in the central and peripheral portions, being highly induced to apoptosis.
- Macrophages located essentially in the peripheral portion, usually with high expression of TNF-alpha cytokines, which recruit and induce the action of inflammatory cells, IL-10, which inhibits the function of neutrophils and induces cell healing and the IDO enzyme (Indoleamine 2, 3-dioxygenase), which converts L-tryptophan, an important bacterial substrate, into L-kynurenine, a protective inflammatory agent that induces apoptosis in lymphocytes.
- Epithelioid cells located in the central portion of the granuloma are modified, squamous and almost dysfunctional macrophages, due to their intense activity in the attempt to remove the infectious agent that is difficult to eliminate. Multinucleated giant cells, formed from the fusion of epithelioid cells, can also be observed.
Those are patients with a phenotype of life-threatening early life infections[^1], where diagnosis usually occurs between 2 and 3 years, and more aggressive. Our patient had shown abscesses and pneumonia since his first months of life, weight-stature gain compromise. As the individual with CGD has dysfunctional phagocytes, catalase-positive microorganisms become difficult to eliminate by the immune system, so that the patient diagnosed with CGD has a tendency of developing several infections through life; pneumonia is the most infectious and prominent, followed by lymphadenitis, suppuration, infectious dermatitis and abscesses formation in the skin, lungs, brain and liver, as well as osteomyelitis, meningitis and sepsis. There has also been manifestation related to the obstruction of gastrointestinal and urinary tract due to granuloma formation[^2].
On the one hand, the most common gram-positive bacteria are Staphylococcus aureus and Nocardia spp. On the other hand, gram-negatives which stand out are Serratia marcescens, Burkholderia cepacia, Klebsiella pneumoniae $^{1,2}$.
The most frequent fungus in CGD is the Aspergillus spp., constantly associated with pneumonias (generally the most prevalent agent), osteomyelitis, suppurative lymphadenitis, and also lung and brain abscesses[^2].
Our patient had persistent pulmonary infection by fungi of the Mucorales order (Zygomycosis), a rare fungal type whose pathogenicity consists of the invasion of blood vessels of infected tissues, resulting in infarction and tissue necrosis $^{12}$.
The patient's chest CT scan for the evaluation of pulmonary zygomycosis showed suggestive findings of fungal balls in both lungs, visualized as non-calcified nodular opacities and pleural effusion on the left, which is an infrequent radiological sign[^6], but of great importance for diagnosis, because the mutual presence of multiple fungal balls with pleural effusion are independent predictors of pulmonary zygomycosis[^5].
The identification of the etiologic agent was possible through microscopy (FIGURE 2) that showed granulomatous tissue, and, through Grocott staining, fragments of hyphae compatible with Mucorales fungi. Histopathological examination represents the most reliable method for diagnosing pulmonary zygomycosis. Direct histomorphological visualization allows differentiation between Zygomycetes (short, tortuous broad hyphae of variable caliber with random branching at right angles) and Aspergillus fungi, and is extremely important because, clinically, lung infections caused by these fungi are indistinguishable[^7].
In this case report, the patient's clinical improvement was achieved after taking Amphotericin B, the drug of choice for zygomycosis therapy. This drug is available from the Ministry of Health in two formulations: Amphotericin B deoxycholate (lower cost and higher toxicity) and Liposomal Amphotericin B (higher cost and lower toxicity), both with high efficacy. The starting dose tends to be $5\mathrm{mg / Kg / day}$, usually increasing to $10\mathrm{mg / Kg / day}$, much higher than the usual dose $(1.5\mathrm{mg / Kg / day})$, and therapy should be continued until there is complete resolution of clinical and radiological signs of infection. To avoid the toxic effects of Amphotericin B, especially on the kidneys, bone marrow, and cardiovascular system, the use of the later less toxic liposomal form is indicated.
Posaconazole is an important therapeutic alternative, since it also has activity against zygomycosis. Itraconazole has low effectiveness, while other azole antifungals, such as fluconazole and voriconazole, as well as flucitocin and echinocandin, are ineffective against mucorales fungi[^4].
The diagnosis of CGD should be suspected in patients with a family history of first-degree relatives with early death, clinical history of severe infections (pneumonias, abscesses, osteomyelitis, lymphadenitis) and recurrent, early onset infections by catalase-positive germs, and it can be confirmed by biochemical tests which evaluate the oxidative function of phagocytes. In this report, measurement of superoxide anion production using the dihydrorhodamine (DHR) method demonstrated that after stimulation with PMA there was no oxidative burst (increased production of superoxide) in the phagocytes $^{8}$.
Prophylactic treatment is indicated for CGD patients to prevent fungal and Staphylococcus infections in addition to removing sources of pathogens. The recommended prophylaxis for these patients is a daily oral regimen of Sulfamethoxazole + Trimethoprim and Intraconazole, but since the latter drug has low activity against mucorales fungi, a substitution with Posaconazole may be considered. The patient should continue to receive routine vaccinations, except those containing live bacteria[^1].
If a patient has pneumonia that is unresponsive to broad spectrum antibiotics, an investigation for fungal pneumonia should be extended to indicate specific treatment, and immunologic investigation in the care of a specialist for innate immune error should be initiated. Early diagnosis allows treatment of the infection as well as prophylactic management to decrease morbidity and mortality and discuss bone marrow transplantation, improving chances of survival and quality of life.
## ACKNOWLEDGEMENTS
This case report was supported by the Liga Académica de Imunologia da Bahia (Academic League of Immunology in Bahia), linked to Universidade Salvador (Salvador University).
### Conflict of Interest
The authors declare that this report was produced without any commercial or funding relationship that could build a potential conflict of interest.
#### Financial Support
The authors declare that they did not obtain any payment, investment, or any financial support for the construction of this report.
Generating HTML Viewer...
References
16 Cites in Article
Jw; Leiding,S Holland Chronic Granulomatous Disease [Internet.
J Winkelstein,M Marino,R Johnston,J Boyle,J Curnutte,J Gallin (2000). Chronic Granulomatous Disease: Report on a National Registry of 368 Patients.
F Filippin,L Souza (2006). Eficiência terapêutica das formulações lipídicas de anfotericina B.
T Rogers (2008). Treatment of zygomycosis: current and new options.
Methee Chayakulkeeree,M Ghannoum,J Perfect (2006). Zygomycosis: the re-emerging fungal infection.
Joandes Fonteque,Mere Saito,Priscila Rizelo,William Teixeira,Aguemi Kohayagawa (2015). Comparação entre duas metodologias para a realização do teste de redução do tetrazólio nitroazul (NBT) em caprinos.
Dimitrios Kontoyiannis,Russell Lewis (2010). Agents of Mucormycosis and Entomophthoramycosis.
Douglas Mandell,Bennett (2015). s Principles and Practice of Infectious Diseases.
T Kuijpers,R Lutter (2011). Inflammation and repeated infections in CGD: two sides of a coin.
N Rider,M Jameson,C Creech (2018). Chronic Granulomatous Disease: Epidemiology, Pathophysiology, and Genetic Basis of Disease.
G Cox (2007). Gastric Zygomycosis (Mucormycosis).
C Kauffman,K Hall (2021). Up To Date.
(2017). SAJS Volume 120 Issue 9/10: van Breda et al. Supplementary Material.
Christof Meischl,Dirk Roos (1998). The molecular basis of chronic granulomatous disease.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Claudia Plech Garcia Barbosa. 2026. \u201cPulmonary Zygomycosis in a Patient with X-CGD\u201d. Global Journal of Medical Research - F: Diseases GJMR-F Volume 24 (GJMR Volume 24 Issue F2).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Subject: Global Journal of Medical Research - F: Diseases
Authors:
Claudia Plech Garcia Barbosa, Matheus Cana Brasil Xavier da Silva, Rafael Burlacchini de Carvalho Magalhaes, Victor Farias Coelho, Thainá Catao Lopes, Joao Carlos Coelho Filho (PhD/Dr. count: 0)
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.