## I. INTRODUCTION
Mr. X, a 78 y.o. retired Caucasian male accountant, breezed through the opening questions of his cognitive evaluation, confident that his sons were worrying needlessly about some lapses in his daily function that they had noticed. He smiled knowingly at the request of the neurologist to draw a clock face with all its markings and showing a specific time in its 12-hour span, having heard all about this test from friends and relatives. There was no reason to worry, couldn't he readily tell the time using his wristwatch, after all? It was only after he realized that there was something wrong with the lopsided ellipse with the missing numbers he kept drawing, that he realized there might be more to his children's worries. Scenes like this one are encountered with increasing frequency in aging populations. However, cognitive dysfunction can impact individuals of various age groups and isn't solely limited to the elderly population. Cognitive dysfunction can impact individuals of various age groups, not solely limited to the elderly population. This estimate is derived from the outcomes of specialized examinations and has been substantiated through prevalence studies, which have demonstrated cognitive impairments such as memory loss and executive dysfunction (Murman, 2015; Allott et al., 2016; Rosselli & Torres, 2019). Executive dysfunction involves deficits in cognitive processes such as attention, planning, decision-making, and problem-solving (Diamond, 2013), and is a common symptom seen in various neurological diseases (Amanzio et al., 2020). The frequency and severity of executive dysfunction can vary depending on the specific disease (Hanna-Pladdy, 2007). Incorporating cognitive assessments and screening tools into routine clinical practice is essential for the early detection and management of these conditions. By including cognitive assessments as part of the regular evaluation, healthcare professionals can identify cognitive impairments at an earlier stage, allowing for timely interventions and appropriate management strategies (Vanderploeg, 2014). For instance, neuropsychological assessment plays a crucial role in clinical practice, particularly for conditions like dementia (Prado et al., 2019). These assessments have the potential to enhance the precision of diagnosis and facilitate the continual observation of illness progress. However, it is often noticed in clinical practise, specifically in the field of autoimmune illnesses, that there is a tendency to give higher priority to somatic symptoms, sensorimotor difficulties, and laboratory test results, while unintentionally overlooking cognitive deficits. By acknowledging the significance of cognitive assessments and integrating them into regular clinical practice, healthcare professionals can augment the thorough evaluation of patients, which results in enhanced early detection, suitable treatment, and improved overall patient well-being, that involve mental health.
Diseases like Multiple Sclerosis (MS) and Systemic Lupus Erythematosus (SLE) can be associated with cognitive decline. A significant percentage of patients with these conditions around $40-60\%$ may experience impairments in cognitive functions such as memory, attention, and processing speed (Macias Islas & Ciampi, 2019; Mendelsohn et al., 2021). Regarding dementia, it is more commonly associated with older age and memory loss. The risk of developing dementia does increase with age, with most cases being diagnosed in individuals over the age of 65 (Brayne & Miller, 2017). However, it's important to note that some forms of dementia, such as early-onset Alzheimer's disease, can affect individuals as young as their 40s (Masellis et al., 2013). Besides this, there is often a challenge in distinguishing between mild cognitive impairment (MCI) and the spectrum of dementia (Knopman & Petersen, 2014). MCI refers to a stage where cognitive changes are present but do not meet the criteria for a diagnosis of dementia (Vega & Newhouse, 2014). There is no clear-cut between MCI and dementia, and it requires a comprehensive evaluation to make an accurate diagnosis (Lu et al., 2022). The discussed diseases have significant and complex molecular pathways and processes that differ and yet are similar to their clinical expression. On the other hand, neuroinflammation is the first negative observed effect in MS, then the results are myelin loss and axonal injury, whereas SLE leads to inflammation and affects the brain processes, both contributing to neurodegeneration (Mandel et al., 2004). Dementia, including Alzheimer's disease, is characterized by the presence of amyloid-beta plaques and tau protein tangles. However, the pathophysiology of SLE and MS is uncommonly associated with these molecules (Stancu et al., 2019). Additionally, small vessel disease and the reduced cerebral blood flow are significant contributors to MCI. Dementia with vascular involvement (Cai et al., 2015). These factors have as consequence the vascular changes, which are not a requisite feature of systemic lupus erythematosus (SLE) or multiple sclerosis (MS), however, in many cases, the symptoms are presented respectively, but it depends on many factors for the appearance of vascular changes on MS and SLE (Jácome Sánchez et al., 2018). Therefore, the pathophysiological along with cognitive examination can help to recognize such brain dysfunctions, targeting each type to the situation and the brain region. This type of examination can interpret issues related to cognitive processes and functions of the brain, such as attention, memory, and problem-solving, to identify any abnormalities or disruptions that may be indicative of neurological or autoimmune conditions.
## II. CLOX: AN EXECUTIVE CLOCK-DRAWING TASK- THE TOOL DESCRIPTION
The CLOX drawing assessment meets all the criteria of a remarkable neuropsychological tool, as it enables the researchers, to quickly and easily, control a wide range of cognitive functions, including comprehension, design, visual memory, spatial abilities, motor organization, and executive function, involving individuals' knowledge, abstract thinking, inhibition of attraction to sensory stimuli, concentration, and tolerance to frustration (Royall et al., 1998). The CLOX's structure is divided into two parts, The purpose of dividing the CLOX task into two sections is to differentiate between the executive control aspects of drawing a clock and the basic ability to draw a clock. Typically, the first portion of the CLOX task requires individuals to draw a clock face from memory while following specific time-setting instructions. This section evaluates the ability to draw a timepiece and visuospatial skills. The second section of the CLOX exercise focuses on the executive control aspects of drawing a clock. It entails supplying individuals with specific instructions and requirements, such as setting the time to a specific hour or adding particular elements to the clock face. This section evaluates the candidate's ability to follow directions, plan, organize, and carry out tasks in a goal-directed manner. By dividing the task into two parts, researchers and clinicians can distinguish between individuals with deficits in fundamental clock-drawing skills and those with deficits in executive functions related to clock drawing. This separation can provide valuable insights into the cognitive processes involved in clock drawing and assist in identifying specific areas of difficulty in individuals with cognitive impairments or neurological disorders. This helps to be better recognized the performance between the executive control of the clock drawing and the clock drawing itself. Moreover, the first part (CLOX1) assesses executive functioning, and the second part (CLOX2) the visuospatial and visuoconstructive capacities (Royall et al., 1998). The individual takes instructions to draw a clock that says 1:45 with the thought it is easy even a child could read this clock. This process is conducted on the empty page of the CLOX form, with no further instructions. The researcher repeats the instructions until they are totally understood, when the individual starts drawing no further details or aid can be given by the researcher. The researcher observes all the drawing procedures to score the 'CLOX1' (there is full detail for the scoring on the front page of the form). Then, follows the drawing procedures of 'CLOX2'. Initially, the researcher must perform the clock drawing while the individual observes the detailed process of drawing e.g. placing the 12, 6, 3, and 9 first. Then, the individual has to draw the CLOX2, and the researcher scores it accordingly (Royall et al., 1998) (please see Appendix 3)
# A. Dementia and Clox Tasks
CLOX task has been extensively examined and used as an evaluative tool for cognitive dysfunction, particularly in the context of dementia, including Alzheimer's disease (AD) (Kim et al., 2018; Rakusa et al., 2018). Royall et al., (1998; 1999) as the creators of CLOX: an executive clock drawing task, conducted many studies. One study examined the application of the CLOX task as a means of discriminating between persons afflicted with Alzheimer's disease and those affected by alternative types of dementia. The study's findings indicate that certain errors made during the CLOX task drawing exercise can effectively differentiate between Alzheimer's disease and non-Alzheimer's dementia, underscoring its diagnostic efficacy (Royall et al., 1999). Many studies have examined the longitudinal variations in the clox drawing tests performance of Alzheimer's disease patients (Lee et al., 2011). Deteriorating performance on the CLOX2 over time was associated with a progressive decline in cognitive function and the progression of Alzheimer's disease, according to the neuroimages and neuroanatomical findings (Shon et al., 2013). Similarly, it has been examined the performance of individuals with mild cognitive impairment (MCI), Alzheimer's disease, and healthy controls on the CLOX task, revealed an additional significant finding. They discovered that the CLOX2 component was particularly sensitive in distinguishing between MCI and Alzheimer's disease, indicating its diagnostic potential. These studies demonstrate the usefulness of the CLOX task for assessing executive functioning and cognitive impairment in patients with dementia spectrum disorders. They emphasize its diagnostic utility, longitudinal tracking abilities, and potential to distinguish between various forms of dementia (Huang et al., 2021). It is essential to note, that the specific results and implications of these studies may vary, and additional research is needed to continue the investigation of the CLOX task in dementia assessment. The multidimensional findings of brain mechanisms related to dementia and factors like biomarkers and brain segmentation are still under investigation.
### B. Mild Cognitive Impairment and Clox Tasks
The CLOX task has been identified as a useful tool for distinguishing between individuals with Mild Cognitive Impairment (MCI) and those experiencing normal cognitive aging. Research has indicated that individuals diagnosed with mild cognitive impairment (MCI) exhibit inferior performance on the CLOX task in comparison to individuals without cognitive impairment, which suggests the presence of initial executive dysfunction (Kim et al., 2018; Rakusa et al., 2018). The CLOX task has shown promising results as a predictor of progression from mild cognitive impairment (MCI) to dementia (Kim et al., 2018). An adequate number of researches conducted over an extended period of time have demonstrated the correlation between suboptimal results on the CLOX task and a heightened susceptibility to the onset of dementia, including AD, among individuals diagnosed with mild cognitive impairment and cognitive complaints (Forti et al., 2010; McGuinness et al., 2015). The CLOX task has been determined to possess sensitivity in identifying cognitive alterations over a period in persons diagnosed with MCI. The deterioration of performance on the CLOX task may serve as an indicator of additional cognitive decline and advancement toward the onset of dementia (Forti et al., 2010). The integration of this test with other cognitive assessments can yield a more helpful appraisal of cognitive dysfunction. Prof. Royall and his colleagues conducted research using the CLOX task to distinguish between individuals with MCI and healthy controls, as discussed previously. Although the findings were solid Royall et al. (2000) continued the research on this topic by employing neuropsychological batteries containing both cognitive and functional status measures in order to diagnose dementia more accurately relative to expert clinicians. Indicating that the use of Clox tasks in conjunction with tools such as the MMSE and IADL can enhance diagnostic performance and increase the likelihood of identifying MCI and dementia accurately (Royall et al., 2015). Similarly, Babins et.al., (2008) investigated the predictive value of the CLOX task for the progression of mild cognitive impairment to dementia. During follow-up evaluations, individuals with MCI who performed inadequately on the CLOX task at baseline were more likely to progress to dementia, including Alzheimer's disease.
## C. Multiple Sclerosis and Clox tasks
The relevance of the CLOX task in assessing executive functioning in individuals with Multiple Sclerosis has not been extensively examined, while many studies have used plenty of neuro psychometric tools (Rogers & Panegyres, 2007). For instance, Benedict et al. (2006) have used the Minimal Assessment of Cognitive Function in Multiple Sclerosis
(MACFIMS), which incorporates the CDT as one of its measures, this study investigated cognitive impairment in MS patients. According to their performance on the CDT, the authors discovered planning and organization deficits in MS patients. These findings have been suggested from similar studies, indicating that cognitive impairment in people with MS can be linked with poor performance on the CDT and general cognitive dysfunction (Achiron, 2003). Zwecker et al. (2018) used the Brief Repeatable Battery of Neuropsychological Tests (BRB-N), clock-drawing test (CDT), and P300 Event-Related Potentials (ERPs) to assess cognitive impairments in MS patients. The key study findings were that $61.5\%$ of 56 early-stage MS patients (less than 3 years) had sub-normal scores on at least two BRB-N or CDT cognition subtests; verbal fluency $(88.6\%)$, short-term memory $(70.5\%)$, visual-spatial learning $(59.1\%)$, CDT $(45.5\%)$, and sustained attention $(29.5\%)$ were impaired. However, a significant barrier to conducting CLOX tasks has been revealed by studies that have shown patients with MS often exhibit difficulties in performing the CDT due to motor disabilities. These difficulties may manifest as impaired planning and organization, reduced attention to detail, and difficulty conceptualizing and executing the task accurately. These deficits in CDT performance can be interpretative as cognitive impairment and can be used as an indicator of disease-related cognitive dysfunction in MS however, these occur because of spinal cord lesions related to MS (Lynch et al., 2005).
## D. Systemic Lupus Erythematosus and Clox Tasks
There is a lack of studies to determine whether Clox tasks are effective in identifying cognitive impairment caused by systemic lupus erythematosus (SLE). Assessing cognitive impairment in SLE can be intricate due to factors such as the diversity of cognitive deficits, the influence of coexisting medical conditions, and the similarity with other neuropsychiatric symptoms of SLE (Seet et al., 2021). Neuropsychological assessments are commonly utilized to evaluate cognitive impairment in SLE, involving a battery of tests that focus on various cognitive domains (Nowicka-Sauer et al., 2011). However, the utilization of CLOX in SLE appears to be restricted. The validity and utility of the CLOX task in this context lack substantial evidence in the research literature in the knowledge of the authors. While neuropsychological tests that evaluate executive functioning in SLE are the Trail Making Test A and B, Stroop Test, and Wisconsin Card Sorting Test (Hanly et al., 2010). Nonetheless, it is pertinent to highlight some noteworthy research studies. Hanly et al., (2006;2009;2010) have conducted many studies to examine cognitive impairment in individuals with SLE by utilizing a comprehensive neuropsychological battery to represent the initial exploration of cognitive-related SLE issues. Although the CLOX task was not explicitly referenced, researchers applied alternative cognitive evaluation measurements that focused on executive functions. According to Kozora et al. (2022) a significant proportion of individuals diagnosed with SLE demonstrate executive dysfunction, which has a discernible impact on their daily functioning. The researchers employed an innovative methodology to examine attentional and executive function in individuals with Systemic Lupus Erythematosus (SLE) using a digital platform. The findings of this study indicate that individuals diagnosed with SLE demonstrated significant improvements in their motor speed and executive functions. Hence, it has been highlighted that individuals diagnosed with SLE may present advantageous outcomes by engaging in cognitive interventions that are custom-designed to address the frontoparietal networks of the brain. It is crucial to note that the studies may involve a broader scope of assessments related to cognitive impairment and executive functioning in SLE, rather than solely concentrating on the CLOX task. However, additional research is required to address the limited amount of existing research on the utilization of the CLOX task in SLE. For this reason, a new research protocol would contribute to a more comprehensive comprehension of the cognitive difficulties experienced by individuals with SLE and facilitate the formulation of focused interventions and treatment strategies. Furthermore, it would be beneficial for future research to investigate alternative cognitive assessment tools that have the potential to capture various dimensions of cognitive functioning within the population affected by SLE. Additionally, given the numerous similarities with other autoimmune disorders like MS, it would be advantageous for researchers to employ specialized neuropsychological tools such as CLOX tasks. This approach would yield more accurate estimations of the results and facilitate meaningful comparisons and contrasts between these findings.
### E. Aim of this Study
The purpose of this study is to validate the neuropsychological tool -CLOX: An Executive Clock Drawing Task- to the Greek population and recognize its reliability in disease existence. Based on previous studies, the CLOX: an executive clock drawing task has been proven a useful neuropsychological tool for the assessment of executive function impairment. However, to the best of the researchers' knowledge, no previous protocol has aimed to validate the screening qualities of CLOX: an executive clock drawing and scoring system on the Greek population. Moreover, there has been no prior endeavor to validate the psychometric properties of this instrument on the Greek population diagnosed with Multiple Sclerosis, Systemic Erythematosus Lupus, Dementia spectrum, and MCI.
## III. METHODOLOGY
### 1. Participants
The study included a total number of participants 283 (76 males/207 females) who had visited the 2nd Neurology Department of Attikon University in the last 18 months, they were examined by experienced neurologists and psychologists who recorded data such as demographics and disease history. It is a convenient sample. The mean age of the subjects was 52,7 years, with a range 19-90; the mean number of years of education was 13 years with a range of 3-21 years. The subjects were divided based on their diagnosis and health control, therefore, patients with MCI $(n = 44$; 15.5% of the total sample), patients with Dementia types $(n = 58$; Dementia $= 39$, AD $= 19$, 20.1%), patients with MS $(n = 68$; 24.0% of the total sample), patients with SLE $(n = 71$, 25.1% of the total sample), and the healthy controls $(n = 43$; 15.2% of the total sample) (please, see Table 1). The recruitment of participants diagnosed with Dementia spectrum and Mild Cognitive Impairment (MCI) were enrolled based on their magnetic resonance imaging (MRI) findings and other relevant clinical assessments conducted prior to their inclusion in the study. The examinations encompass the analysis of specific molecules, namely phosphorylated tau (p-tau) and amyloid-beta (Aβ), which have been extensively researched in relation to mild cognitive impairment (MCI) and Alzheimer's disease (AD). Also, biomarkers, such as cerebrospinal fluid (CSF) levels of p-tau and Aβ, along with neuroimaging measures like frontotemporal type (FT-MCI), frontotemporal dementia (FTD), and normal pressure hydrocephalus (NPH), are being investigated as potential diagnostic and prognostic markers for mild cognitive impairment (MCI) and its progression to dementia. Inclusion criteria were diagnosis of the following diseases: dementia and Alzheimer's Disease based on DSM-V, with a previous clinical examination with cerebrospinal fluid (CSF) biomarker measurements such as beta-amyloid 42, tau, and phospho-tau as they are considered a hallmark for the diagnosis (Ashton et al., 2022), and structural brain imaging (CT or MRI); the individuals with MS were recruited as they had a definite diagnosis of MS according to the McDonald criteria 2017 (McDonald Criteria, 2022), their clinical condition was stable without relapses the last 6 months, they had MRI and they had expressed cognitive issues also they had previous examinations with biomarkers such as IL-6, CD14 e.g.; individuals with SLE with a diagnosis that meet the revised American College of Rheumatology (ACR) classification criteria, they had an MRI and had expressed cognitive issues; Healthy controls were random health visitors of the clinic as well as university students. Additional inclusion criteria; All participants were aged over 18 years, able to speak and read Greek fluently, and provide themselves or/and their caregivers with full detailed information and consent forms. Exclusion criteria were any type of comorbidities such as cardiovascular disease and psychiatric disorder; individuals with motor deficits in handwriting; other reasons such as medication or/and alcohol use that may affect participants' mental function and/or mental health; participants with a Covid-19 diagnosis were excluded.
#### 2. Ethics
The researchers requested permission from Professor Donald Royall, the creator of CLOX: Clock Drawing Executive Test. The researchers were granted permission, thereby facilitating the validation process to proceed. Following that, the investigators provided a thorough account of the research protocol, encompassing the methodologies for conducting the study and acquiring approval from the Attikon University Hospital Ethics Committee. The reference number assigned to the protocol in question is identified as 'BNEYR, EBD366/16-7-2021'. The researchers followed the guidelines specified in the General Data Protection Regulation (GDPR) throughout the entire study, thus guaranteeing the safeguarding of the participants' information.
#### 3. Procedure
All stages of the protocol were conducted at Attikon University Hospital. The participants recruited in the 2nd Neurological Department were assessed using interviews and a neuropsychological examination. The chosen location was specifically designed to facilitate the administration of the psychometric test in a controlled environment, devoid of any extraneous stimuli, such as sound. The process began by informing the participants about their involvement. After obtaining their consent, the interview proceeded by gathering demographic information and relevant documentation, including MRIs and biomarker tests, to validate their condition. The participants subsequently underwent a series of neuropsychological tests. The recruitment process spans a period of 18 months. The researchers implemented comprehensive measures to ensure the well-being of the participants, specifically focusing on mitigating the risks associated with the COVID-19 pandemic. or the diagnosis (Ashton et al., 2022), and structural brain imaging (CT or MRI); the individuals with MS were recruited as they had a definite diagnosis of MS according to the McDonald criteria 2017 (McDonald Criteria, 2022), their clinical condition was stable without relapses the last 6 months, they had MRI and they had expressed cognitive issues also they had previous examinations with biomarkers such as IL-6, CD14 e.g; individuals with SLE with a diagnosis that meet the revised American College of Rheumatology (ACR) classification criteria, they had an MRI and had expressed cognitive issues; Healthy controls were random health visitors of the clinic as well as university students. Additional inclusion criteria; All participants were aged over 18 years, able to speak and read Greek fluently, and provide themselves or/and their caregivers with full detailed information and consent forms. Exclusion criteria were any type of comorbidities such as cardiovascular disease and psychiatric disorder;
individuals with motor deficits in handwriting; other reasons such as medication or/and alcohol use that may affect participants' mental function and/or mental health; participants with a Covid-19 diagnosis were excluded.
Table 1: Sample Frequencies
<table><tr><td>Disease Type</td><td>Frequency</td><td>Percentage</td></tr><tr><td>Mild Cognitive Impairment</td><td>44</td><td>15.5%</td></tr><tr><td>Dementia</td><td>57</td><td>20.1%</td></tr><tr><td>Multiple Sclerosis</td><td>68</td><td>24.0%</td></tr><tr><td>Systemic Lupus Erythematosus</td><td>71</td><td>25.1%</td></tr><tr><td>Health Control</td><td>43</td><td>15.2%</td></tr></table>
## IV. RESULTS
### a) Internal consistency
The reliability of CLOX: an Executive Clock-Drawing Task, has been investigated as a scale of measurement in the Greek population. This examination focused on individuals with specific diseases, including MCI, dementia spectrum disorders, MS, SLE, and health control. A total of 283 participants successfully completed the scale. The evaluation of the internal consistency of the CLOX task was conducted based on the reported Cronbach's alpha values. The initial task, comprising 15 items, exhibited satisfactory internal consistency, as indicated by a Cronbach's alpha coefficient of.798. The second task, which also comprised 15 items, demonstrated satisfactory internal consistency, as evidenced by a Cronbach's alpha coefficient of.785. When examining the comprehensive scale comprising all 30 items, the internal consistency was found to be highly satisfactory, as indicated by a Cronbach's alpha coefficient of.874. The results of this study indicate that the CLOX: An Executive Clock-Drawing Task demonstrates a high level of reliability as a tool for evaluating executive functioning and cognitive impairment in the Greek population. This is particularly applicable to individuals with MCI, dementia, MS, and SLE. The high level of internal consistency, ranging from good to very good, suggests that the items within the CLOX task are effectively measuring a cohesive construct in a reliable manner. Therefore, the study proposes that the CLOX: An Executive Clock-Drawing Task is a dependable tool for evaluating executive functioning and cognitive impairment in the Greek population, particularly in individuals diagnosed with MCI, dementia, MS, and SLE, based on the obtained results. The high level of internal consistency observed in the task suggests that the items comprising the CLOX task consistently assess the targeted construct of executive functioning and cognitive impairment.
### b) Construct Validity
A Principal Component Analysis (PCA) was conducted on the CLOX task 1 items. The interpretations of Kaiser-Meyer-Olkin Measure of Sampling Adequacy:.787 indicates that the sample used in the analysis is considered adequate for conducting PCA. Generally, a value above.6 is considered acceptable. Bartlett's Test of Sphericity $(\chi 2105 = 1097.418(<.001)$ suggests that the correlation matrix is not an identity matrix, indicating that there is a sufficient correlation among the variables for PCA to be meaningful. Therefore, the significant p-value of less than 0.001 suggests that there is enough correlation among the variables in the CLOX task 1 items, indicating that PCA is meaningful and can be applied to extract underlying factors or dimensions from the data. For example, for CLOX1.1, the initial commonality is 1.000, indicating that the item explains $100\%$ of its own variance. The extraction communality is.428, suggesting that this item shares some common variance with the other items in the analysis (please see Appendix 1). Total Variance Explained shows the first component explains $27.360\%$ of the variance, the second component explains $12.006\%$, and so on. The cumulative percentage indicates how much total variance is explained by the successive components. For example, the first two components explain a cumulative percentage of $39.366\%$. Therefore, the PCA results suggest that there are several components (factors) that contribute to the variation in the CLOX task 1 items. The first few components explain a significant proportion of the variance, while the subsequent components contribute less.
In the same spirit, the Kaiser-Meyer-Olkin Measure of Sampling Adequacy for CLOX task 2 is.733, suggesting that the data used for factor analysis is reasonably adequate. Bartlett's Test of Sphericity examines the correlation matrix presenting $(\chi 2105 = 1436.865, p < 0.001)$. This result suggests that the correlation matrix is significantly different from an identity matrix, indicating that the variables are likely correlated and suitable for factor analysis. The extraction communalities range from 0.363 to 0.894, indicating the amount of variance explained by the factors after extraction. The first component explains $27.533\%$ of the variance, and cumulatively, the first three components explain $50.877\%$ of the variance. In the rotation sums of squared loadings, the first component explains $16.845\%$ of the variance, and cumulatively, the first three components explain $46.042\%$ of the variance (please see Appendix 2). Based on these results, it appears that there are meaningful components or factors in the data that explain a significant proportion of the variance.
### c) The Effect of Disease Types on CLOX Performance
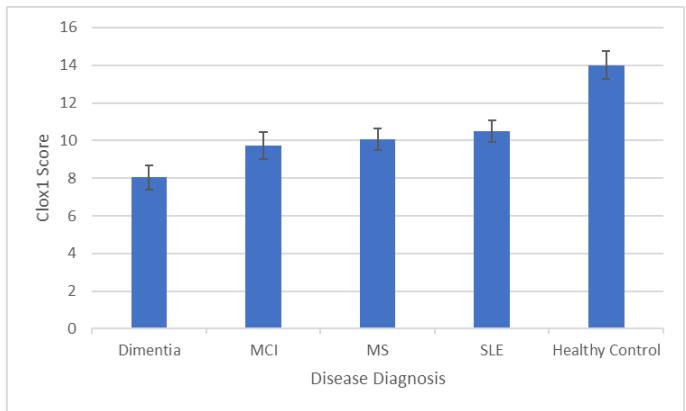
One-way ANOVAs were run with Disease Diagnosis as the IV and CLOX scores as the DV (Tukey's HSD was used for pairwise comparisons). There was a main effect of Disease Diagnosis on CLOX1 Scores $(F4,272 = 37.599$, $p <.001)$ where pairwise comparisons showed that all Disease Diagnoses were associated with poorer performance than Healthy Controls (all at $p <.001$ ), and those with a Dementia diagnosis performed worse than those with any other Disease Diagnosis (all at $p <.001$ see Figure 1).
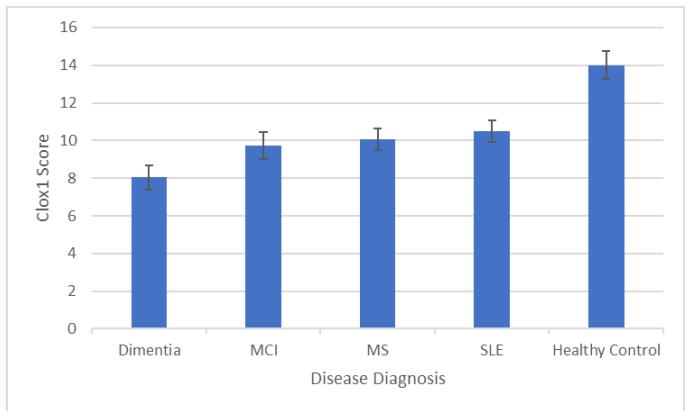 Figure 1: Mean CLOX1 Score by Disease Diagnosis (error bars show $95\%$ CI).
There was also a main effect of Disease Diagnosis on CLOX2 scores (F4, 278=34.1, p\<.001) where pairwise comparisons showed that all Disease Diagnoses were associated with poorer performance than Healthy Controls (all at $p <.001$ ) and those with a Dementia diagnosis performed worse than those with any other Disease Diagnosis (all at $p <.001$ see Figure 2).
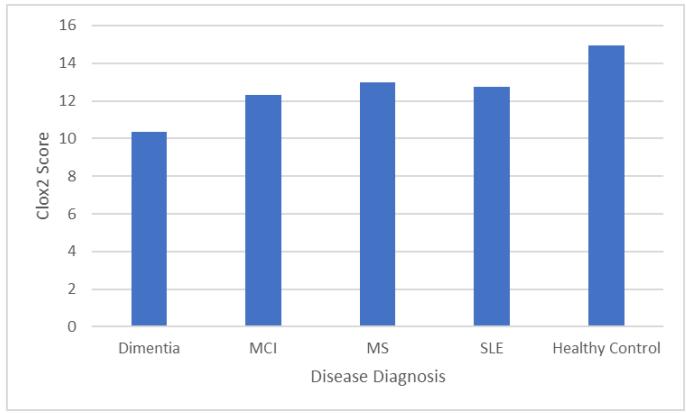 Figure 2: Mean CLOX2 Score by Disease Diagnosis (error bars show $95\%$ CI).
The results of this analysis suggest that individuals with MCI, SLE, MS, and dementia diagnoses exhibit lower performance on both the CLOX1 and CLOX2 tasks in comparison to a control group consisting of healthy individuals. Moreover, when it comes to disease diagnoses, individuals diagnosed with Dementia exhibit notably poorer performance on both tasks in comparison to individuals diagnosed with other diseases. The findings of this study indicate that the scores of CLOX1 and CLOX2 have the potential to be valuable indicators for differentiating between Healthy Controls and individuals diagnosed with SLE, MS, MCI, and specifically Dementia.
ROC curves and CLOX task1 and Task2 In accordance with the above findings, have run a ROC curve analysis as it is a valuable tool in psychometric validation, providing insights into the discriminatory power, optimal cut-off point, comparative analysis, and diagnostic accuracy of the CLOX as a tool. The Area Under the ROC Curve (AUC) is a commonly employed performance metric for classifiers and diagnostic tests. It measures the test's ability to distinguish between positive and negative cases. In our case, the AUC values for the variables represent test results. The test result variables being evaluated based on the ROC curve analysis are CLOX task 1 total score and CLOX task 2 total score. The actual condition being evaluated is Health Control. The Area Under the ROC Curve for CLOX task 1 was 0.938 and for CLOX task 2, it was 0.940. The AUC values indicate that both scores have a high ability to differentiate between genuine positive and negative states. The Classifier Evaluation Metrics provide additional information regarding the efficacy of the classifiers based on the Gini Index and the Kolmogorov-Smirnov (K-S) statistics.
Classifier Performance Metrics:
CLOXtask1 total score: Gini Index: 0.876, Max K-S Statistics: 0.759, Cutoff: 12.0000;
CLOXtask2 total score: Gini Index: 0.880, Max K-S Statistics: 0.834, Cutoff: 14.0000;
Both CLOX task1 total score and CLOX task 2 total score demonstrate strong performance in differentiating between positive and negative actual states, with CLOX task 2 having a slightly higher Area Under the ROC Curve. The Gini Index quantifies the disparity in the distribution of predicted probabilities. A higher Gini Index indicates a more effective classifier. In our case, the Gini Index for CLOX1.The total is 0.876, whereas it is 0.880 for CLOX2.Total. These values indicate that both classifiers have a high capacity for discrimination in predicting the actual positive state of Health Control. The K-S Statistics measure the greatest disparity between the cumulative distribution functions of the positive and negative categories. It indicates the classifier's capacity to distinguish between the two groups. CLOX task 1 total score and CLOX task 2 total score have Max K-S values of 0.75 and 0.83, respectively. The greater the Max K-S value, the more efficient the classifier. In both instances, the Max K-S values reported are associated with particular cut-off values. If multiple cut-off values exist, the largest is reported. These Max K-S values indicate that both the scores classifiers are able to distinguish between positive and negative actual states (please see the Figure 3 and 4).
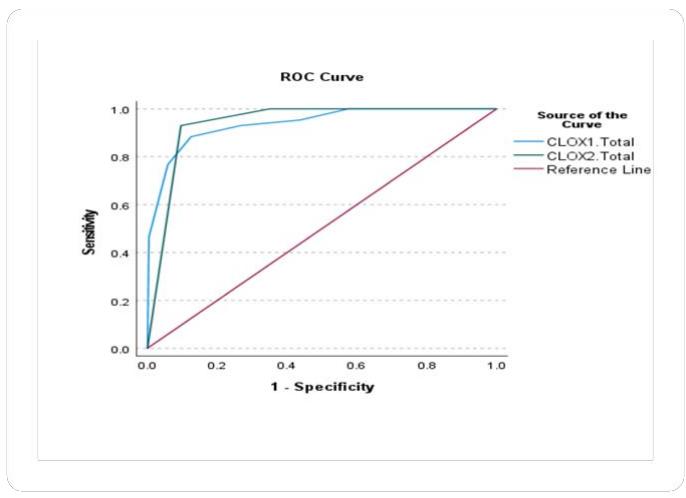 Figure 3: ROC curve
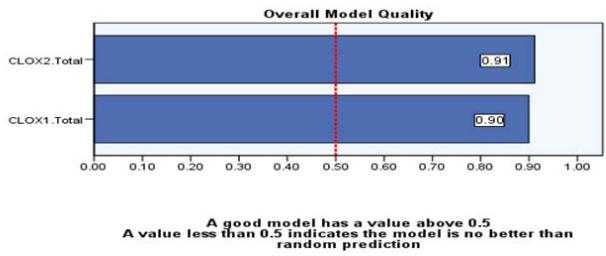 Figure 4: Model Quality
## V. CONCLUSION
The present study aimed to investigate the effectiveness of the CLOX: an Executive Clock-Drawing Task assessment in the Greek population. The availability of valid and reliable data is crucial in establishing representative performance for cultural elements as well as the disease of interest. Understanding the effectiveness of this assessment can have important implications for clinical and research settings. This investigation included a sample of the Greek population with varying types of diseases. The researchers aimed to make the sample as representative as possible of most age ranges. The study recruited participants from multiple clinical settings and ensured that individuals with MCI, dementia, MS, and SLE were included. The internal consistency and construct validity of the CLOX assessment provide strong evidence for its reliability and validity. This suggests that the CLOX assessment can be a valuable tool in clinical settings for diagnosing and monitoring individuals with MCI, dementia, MS, and SLE. However, it is important to note that our study had certain limitations. One limitation of our study is that we relied on specific neuropsychological tools, which may introduce potential bias, a more comprehensive approach to neuropsychological tests could have provided a more holistic understanding of cognitive functioning and psychosocial impact. Future studies could consider incorporating a wider range of assessments to capture the complexity of these disorders. Additionally, it used a relatively small sample size. These limitations should be considered when interpreting the results. Prior research, however, has explored the disease of MS, SLE, dementia, and MCI from a more holistic neuropsychological perspective, revealing the complex consequences of these disorders on cognitive functioning, emotional well-being, quality of life, and functional skills. Studies on MS, for example, have looked at comprehensive neuropsychological assessments that include cognitive testing, emotional assessment, and functional evaluation to capture the disease's cognitive and psychosocial impact (Benedict et al., 2017; Goverover et al., 2016). These studies frequently look at the connections between cognitive deficits, mood disorders, fatigue, and quality of life measurements in people with MS. Regarding SLE, studies have mainly focused on cognitive functioning, psychological welfare, and disease-related factors. Comprehensive neuropsychological batteries, as well as measures of depression, anxiety, disease activity, and quality of life, may be used in these investigations (Hanly et al., 2009; 2010). However, the goal is to comprehend the cognitive and psychosocial consequences of SLE as a primary factor in the disease onset, and also to understand link factors to the disease progress. In the case of dementia and MCI, holistic methods of neuropsychological examinations strive to capture cognitive functioning, functional abilities, mood abnormalities, and quality of life in these patients. Comprehensive assessment batteries, which include measures of cognition, functional abilities, psychiatric symptoms, and well-being, have been studied to provide a holistic understanding of an individual's cognitive decline and its impact on daily functioning (Hussenoeder et al., 2020). In the same way, there have been numerous research conducted to investigate the sex differentiations in cognitive abilities. It is crucial to acknowledge that although there are certain overarching patterns, there are significant individual variations within each sex category, and any detected disparities should not be taken as indicative of an individual's skills. Unfortunately, a notable variation in participant numbers between males and females is evident in this study. The past decade has seen a significant increase in studies on sex differences in executive function, particularly those using functional neuroimaging. Despite this growing body of knowledge, the effects of sex on executive function are still poorly understood due to methodological variability in executive function task selection, participant inclusion and exclusion criteria, and scanning procedures (Gaillard, Fehring & Rossell, 2021). A significant review suggested that the executive functions of monitoring, response inhibition, and cognitive shifting as executive function domains present differentiations, however, there is a need for further investigation. It is imperative to acknowledge this constraint to account for the impact of health, societal, and cultural influences on sex variations in cognitive test performance, especially in the discussed diseases. This study encompassed individuals ranging in age from 18 to 90 years old. The researchers tried to get a representative sample that covered each phase of human life. The aim of validating this specific psychometric tool is to enable its application in individuals diagnosed with neuroinflammatory and neurodegenerative diseases, or those suspected to have a decline in executive function. The purpose is to assess the individual's abilities in executive functioning by evaluating their approach to what was given to them. The participants are examined on variables that indicate the presence of adverse signs of planning, organization, initiation, and cognitive flexibility. Consequently, a well-executed clock drawing may suggest intact executive functioning, while errors or omissions may indicate executive dysfunction. Furthermore, the inclusion of participants from different age groups, including both younger and older individuals, enables researchers to discern patterns of cognitive performance across various stages of development. Also, including younger and older participants allows researchers to identify developmental trends in cognitive performance. For example, they can observe how executive functioning skills develop in young adults and whether they decline
or remain stable in older adults. Moreover, can be a helpful tool for the researchers to explore why some older individuals maintain strong executive functioning skills while others experience declines, potentially uncovering protective factors or interventions that promote cognitive health.
Conclusions To conclude CLOX: An Executive Clock-Drawing is a reliable and valid tool in the Greek population with the disorders described above suggesting that the CLOX assessment shows promise as a valuable tool for assessing executive function in individuals with specific disorders. Despite the positive findings in the Greek population with the described disorders, it is important to validate the reliability and validity of the CLOX assessment in other populations and with a broader range of disorders. This will help establish the generalizability of the findings and determine if the CLOX assessment can be used as a reliable tool across different clinical and research contexts. This ongoing research will enhance our understanding of the value and applicability of the CLOX assessment.
### Supplementary Materials: Not applicable
Author Contributions: Conceptualization Eleni Sideri; George P.Paraskevas; investigation, Eleni Sideri; Ioanna Tsantzali; resources, Georgios N. Papadimitropoulos, Claire Kelly, Dimitrios Kitsos, Stella Fanouraki, Angeliki Sterpi; writing—original draft preparation, Eleni Sideri; Ioanna Tsantzali; formal analysis, Eleni Sideri; George P.Paraskevas; George Tsivgoulis writing—review and editing, George Papadimitropoulos, Eleni Sideri, Claire Kelly; supervision, George P.Paraskevas, and Konstantinos Voumvourakis. All authors have read and agreed to the published version of the manuscript.
Funding: Not applicable
- Institutional Review Board Statement: BNEYR, EBD366/16-7-2021,
- Informed Consent Statement: Informed consent was obtained from all subjects involved in the study, also, there is written informed consent has been obtained from the participants to publish any paper related to this data.
- Data Availability Statement: Due to restrictions of the ethics committee patients' data cannot become publicly available, however, they may be provided by the correspondent author according to a reasonable request
- Acknowledgments: We sincerely thank Professor Donald Royall for his license to translate, validate, and use the CLOX: an executive drawing task and conduct this study. Conflicts of Interest: Not applicable.
#### APPENDICES
#### Appendix 1
Communalities
<table><tr><td></td><td>Initial</td><td>Extraction</td></tr><tr><td>CLOX1.1</td><td>1.000</td><td>.428</td></tr><tr><td>CLOX1.2</td><td>1.000</td><td>.483</td></tr><tr><td>CLOX1.3</td><td>1.000</td><td>.385</td></tr><tr><td>CLOX1.4</td><td>1.000</td><td>.556</td></tr><tr><td>CLOX1.5</td><td>1.000</td><td>.630</td></tr><tr><td>CLOX1.6</td><td>1.000</td><td>.507</td></tr><tr><td>CLOX1.7</td><td>1.000</td><td>.420</td></tr><tr><td>CLOX1.8</td><td>1.000</td><td>.672</td></tr><tr><td>CLOX1.9</td><td>1.000</td><td>.747</td></tr><tr><td>CLOX1.10</td><td>1.000</td><td>.642</td></tr><tr><td>CLOX1.11</td><td>1.000</td><td>.465</td></tr><tr><td>CLOX1.12</td><td>1.000</td><td>.594</td></tr><tr><td>CLOX1.13</td><td>1.000</td><td>.601</td></tr><tr><td>CLOX1.14</td><td>1.000</td><td>.691</td></tr><tr><td>CLOX1.15</td><td>1.000</td><td>.417</td></tr></table>
#### Appendix 2
<table><tr><td></td><td>Initial</td><td>Extraction</td></tr><tr><td>CLOX2.1</td><td>1.000</td><td>.839</td></tr><tr><td>CLOX.2.2</td><td>1.000</td><td>.894</td></tr><tr><td>CLOX2.3</td><td>1.000</td><td>.844</td></tr><tr><td>CLOX2.4</td><td>1.000</td><td>.608</td></tr><tr><td>CLOX2.5</td><td>1.000</td><td>.507</td></tr><tr><td>CLOX2.6</td><td>1.000</td><td>.363</td></tr><tr><td>CLOX2.7</td><td>1.000</td><td>.581</td></tr><tr><td>CLOX2.8</td><td>1.000</td><td>.615</td></tr><tr><td>CLOX2.9</td><td>1.000</td><td>.695</td></tr><tr><td>CLOX2.10</td><td>1.000</td><td>.763</td></tr><tr><td>CLOX2.11</td><td>1.000</td><td>.626</td></tr><tr><td>CLOX2.12</td><td>1.000</td><td>.641</td></tr><tr><td>CLOX2.13</td><td>1.000</td><td>.656</td></tr><tr><td>CLOX2.14</td><td>1.000</td><td>.502</td></tr><tr><td>CLOX2.15</td><td>1.000</td><td>.754</td></tr></table>
<table><tr><td></td><td>Initial</td><td>Extraction</td></tr><tr><td>CLOX2.1</td><td>1.000</td><td>.839</td></tr><tr><td>CLOX.2.2</td><td>1.000</td><td>.894</td></tr><tr><td>CLOX2.3</td><td>1.000</td><td>.844</td></tr><tr><td>CLOX2.4</td><td>1.000</td><td>.608</td></tr><tr><td>CLOX2.5</td><td>1.000</td><td>.507</td></tr><tr><td>CLOX2.6</td><td>1.000</td><td>.363</td></tr><tr><td>CLOX2.7</td><td>1.000</td><td>.581</td></tr><tr><td>CLOX2.8</td><td>1.000</td><td>.615</td></tr><tr><td>CLOX2.9</td><td>1.000</td><td>.695</td></tr><tr><td>CLOX2.10</td><td>1.000</td><td>.763</td></tr><tr><td>CLOX2.11</td><td>1.000</td><td>.626</td></tr><tr><td>CLOX2.12</td><td>1.000</td><td>.641</td></tr><tr><td>CLOX2.13</td><td>1.000</td><td>.656</td></tr><tr><td>CLOX2.14</td><td>1.000</td><td>.502</td></tr><tr><td>CLOX2.15</td><td>1.000</td><td>.754</td></tr></table>
©Royall, 1995
### CLOX
CLOX: EKTéλeσθ Φικμασία Σχεδιασης Πολογιό
Ovoataenovu: Hpeoynia:
BHMA 1: Topiote m oiao npiv kevn enpavveia eot o nnpakato kikoc v eaiv opatoc. Znjnte ano towiy oumueteyov/ooa va oyidaev ev pooi oio niopoe. Aotne tc e jnnpopoeic; Eoyeiote ev pooi nov va deyiv neip a 45. Bateouc dckekac taou apheuoc otnv enpavvraou pooyou etot oe nei aoiau maopei va ra daiaea. Eaavalafete n onnyie; meyoi va yivow katavontc. Molc o n oumueteyov/ooa apxoe va oyidae, dev entepenatai papertpo bontheia. Baoholoyntote au to pooi (CLOX 1).
BHMA 2: Etnpvee oe uyny nlaupa ncsolao, cydoane eva poal stov npakato kko evo on Omueteyow/ovaa cagnapakoloebe. Tonoetne pnoata ra 12,63kai 9. ouanpoote rouv unolouov apheuoc. Oeikteva deoyuvw'145'. Kavte rouc deikte oav bcln. O deiknt nov deiyev tny opa va eivai no jmuoc Znntote Tovn oumueteyow/ovaa va atnypeae to pool ot mky katw deia yovia. Bauoloyntote avo to pooi (CLOX 2).
<table><tr><td colspan="4">Aξωλόγηη</td></tr><tr><td>Kατενθυνιρία Στοχεία</td><td>Βαβμοι</td><td>CLOX 1</td><td>CLOX 2</td></tr><tr><td>To σγίμα μοιαίς με ρολίς;</td><td>1</td><td></td><td></td></tr><tr><td>Yπάργει o εἰδεταρίας καὶος;</td><td>1</td><td></td><td></td></tr><tr><td>Διαμετρος ≥ 2,5 cm;</td><td>1</td><td></td><td></td></tr><tr><td>Ολοι αἰσρήμαι μεσα στον καὶος;</td><td>1</td><td></td><td></td></tr><tr><td>Av ὄργουν λαδή Αστις Αποτάσες, σημεία διορβασης; η δαυγαρής;</td><td>1</td><td></td><td></td></tr><tr><td>12,6,3,9,μακηκα πράτα;</td><td>1</td><td></td><td></td></tr><tr><td>Yπάργουν σωστές; αποτάσες; εἰνα συμετρίας; εκατερώνν τοῦ ἀςουν 12-6;</td><td>1</td><td></td><td></td></tr><tr><td>Av ὄργουν λαδή Αστις Αποτάσες,∪αργουν σημεία διορβασης; η δαυγαρής;</td><td>1</td><td></td><td></td></tr><tr><td>Móvo αραβκοι αρεβμοία;</td><td>1</td><td></td><td></td></tr><tr><td>Móvo αραβκοι 1-12 μεταύν τον αραβμίαν; (αγνοστε η τη σημειραρία)</td><td>1</td><td></td><td></td></tr><tr><td>H δαβογην τον αραβμίαν 1-12 σαστή (χαρίς; παραλίνες; η παρεθίκας);</td><td>1</td><td></td><td></td></tr><tr><td>Móvo 2 δείκετε; (αγνοστε η τη τανύνμημι/σημαίδα διορβασης)</td><td>1</td><td></td><td></td></tr><tr><td>Oι δείκετε; γαραρεστην συν βέλη;</td><td>1</td><td></td><td></td></tr><tr><td>O αραδόδεκτην; εἰνα Ανμεσα στο 1 κατ to 2;</td><td>1</td><td></td><td></td></tr><tr><td>O λεπτοδεκτήν; μεγαλίνετος; ανο τον αραδόδετη;</td><td>1</td><td></td><td></td></tr><tr><td>Κανείνα σατο τα παρακίατο. 1) δείκητην; σαν δείψην 4 η έ 5</td><td>1</td><td></td><td></td></tr><tr><td>2) Γραρηκεν "1:45"</td><td></td><td></td><td></td></tr><tr><td>3) Μασαδόπος αλλη σημειραρία (π.χ. "9:00");</td><td></td><td></td><td></td></tr><tr><td>4) Παβλ Εδεγουν προς; τα μεδος;</td><td></td><td></td><td></td></tr><tr><td>5) Παρεμβόλες αὐτο"δείκες" καν "δυν"</td><td></td><td></td><td></td></tr><tr><td>6) Yπάργουν λεδείς, γαρμματα η ἀσγετες, εκόνες;</td><td></td><td></td><td></td></tr><tr><td>7) Στοχεία σαν τον καταβεν τούκλο;</td><td>ΣΥΝΟΙ</td><td></td><td></td></tr></table>

Generating HTML Viewer...