Safety and Diagnostic Accuracy of Biopsy of Targeted Splenic Lesions Under Ultrasound Guidance Using the Multiple-Pass Technique Without Co-Axial in Kinshasa Hospitals
The most frequent splenic pathologies can pose a diagnostic challenge to clinicians, radiologists and pathologists. These pathologies are innumerable, and may be of malignant or benign tumoral origin. Six techniques are currently available to obtain splenic tissue samples for pathological evaluation. Less invasive percutaneous techniques, performed by an interventional radiologist under ultrasound or CT guidance, include fine-needle aspiration biopsy and core tissue biopsy. They are associated with fewer complications and greater precision.
## I. INTRODUCTION
The spleen is a voluminous vascular lymphatic organ located in the upper part of the abdominal cavity on the left side, between the two extremities of the abdomen, the stomach and the diaphragm, composed of white and red pulp. The white pulp is made up of lymphoid nodules and lymphoid tissue, while the red pulp is made up of venous sinusoids between which lie the splenic cords. The stroma of the red and white pulp is composed of fibers and reticular cells [1]. The spleen is the body's largest lymphoid organ. Unlike the lymph nodes, the spleen is not drained by the lymphatic system, but rather is connected to the systemic circulation. The most common splenic pathologies can pose a diagnostic challenge to clinicians, radiologists and pathologists alike. These pathologies are innumerable, possibly of malignant or benign tumoral origin such as; lymphomas and metastases; of specific or non-specific infectious origin with tuberculosis in the lead, of fungal origin without forgetting infiltrative processes such as sarcoidosis
[1-4]. As imaging has not proven accurate for diagnosis, splenic tissue samples may be required to accurately determine its malignant and or infectious tumoral nature [1,2]. Splenic tissue samples can be obtained either by splenectomy or percutaneous biopsy [3-4].
Splenectomy is accompanied by morbidity (8.6%-37%) and mortality of (0%-2.9%). This is mainly due to infection generated [6-7]. Percutaneous biopsy, with reported complication rates of 0.5% for organs such as the liver and kidneys, is a potentially safer alternative to surgical biopsy [8-11]. Historically, biopsy percutaneous image-guided biopsy was approached with apprehension by radiologists, due to accessibility and bleeding risk [12]. This reluctance may be linked to a high rate of major complications (13%) for percutaneous spleen biopsy performed with a large 14 Gauge [13] needle. Several recent publications have reported much lower complication rates with small-gauge needles (18 Gauge or smaller) [4-6,14]. The diagnostic accuracy of splenic biopsy varies according to publications, ranging from 84% to 90% [4,11,14]. In the Democratic Republic of Congo; no study to our knowledge has reported on this technique; hence the main objective of the present study, which consisted in demonstrating the safety and diagnostic accuracy of targeted splenic biopsy puncture using the multiple-pass technique without co-axial under ultrasonographic guidance; in hospital settings in Kinshasa; university clinics in Kinshasa in particular.
## II. MATERIALS AND METHODS
### a) Type and Period of Study
This was a descriptive, multicenter, observational study of the various radioguided percutaneous splenic puncture-biopsy procedures performed using the multiple-pass technique without placement of a co-axial tru-cuts over a period of six and a half years, i.e. from January 2018 to June 2024:
### b) Study Setting
We carried out this study in eight hospital institutions in the city and province of Kinshasa, including one tertiary-level institution, the University Clinics of Kinshasa, and the following primary and secondary-level institutions: Center medical Diamant de Kinshasa, Clinique présidenteille de l'unité Africaine, Centre Hospitalier de Kingasani, Centre d'imagerie medicale pilote Kokolo, Centre de diagnostic spécialisé, d'expertise et d'Imagerie Interventionnelle de la RDC, Pistis medical center de Limete, Centre spécialisé de Kinshasa and Vision medicale pour tous. All these data were collated at the Kinshasa university clinics, located on the Kimwenza road, in the Mont Amba district, in the commune of Lemba.
### c) Study Population
A total of eighteen patients who had undergone biopsy of splenic lesions were included in the present study. These patients ranged in age from 5 to 64 years, with a median age of 35.5 years. Of the 18 patients, twelve were male and six female.
### d) Inclusion Criteria
Any male or female patient referred for biopsy puncture of a splenic lesion and/or echoguided drainage of a radio-guided splenic abscess with a medical imaging result (abdominal ultrasound, abdominal CT scan and/or abdominal MRI); Any patient with a haemostatic assessment deemed fair (Bleeding time: Clotting time); Any patient with a hemoglobin and/or hematocrit level within acceptable limits (Hemoglobin $\geq 7$ g/dl and Hematocrit $\geq 21\%$ ); Any patient who has freely given written consent for interventional radiology (IR) at splenic level.
### e) Criteria for Non-Inclusion
The following were not included in the present study: Any patient referred for a splenic IR procedure without a prior medical imaging result; Any patient with a haemostatic balance below the set thresholds (TS:TC:); Any patient with a hemoglobin and/or hematocrit level below the set thresholds ( $Hb \leq 7\,g/dl$ and $Hct \leq 21\%$ ) refractory after correction, any patient who has not freely given written consent for the indicated splenic interventional radiology procedure.
### f) Parameters of Interest and Operational Definitions Parameters of Interest:
1. Socio-demographic parameters included age, sex and place of origin.
2. Clinical Parameters: Included clinical reason for referral to IR unit, incident after IR procedure (minor, major, etc).
3. Biological Parameters: Included hemoglobin level, hematocrit level, red blood cell count, bleeding time, coagulation time, pathological findings.
4. Radiological Parameters: Included the means of imaging used to perform the procedure, the indication for biopsy or drainage of intra-splenic collections, the equipment used (automatic gun, semi-automatic gun, suction gun, drainage trocar, etc.). the technique used (co-axial or indirect technique, axial or direct technique), the type of anaesthesia used (local versus general anaesthesia), the anaesthetic product used, the number of cores taken, the number of passes made, whether or not the biopsy path was embolized, whether or not haemostats were used, whether or not clots were used, the type of premedication, the anatomopathological result and, lastly, the technique used to take the cores
(multiple-pass technique without coaxial (T-MPSC).
5. Materials and Technique of Multiple Passages without Coaxial (T-MPSC).
Precautions: Oral and written consent was obtained. A haemostasis test was performed 48 hours prior to the procedure. A platelet count greater than or equal to $150,000 / \mathrm{mm}^3$ and a prothrombin time (PT) greater than or equal to $55\%$ were required for the procedure. We used 16 to 14 gauge (G) automatic and semi-automatic biopsy needles $10 - 20 \mathrm{~cm}$ in length.
Equipment: After radiological consultation, we proceeded to review the examinations and reassess the lesion using B-mode ultrasound and triplex Doppler with convex and linear multi-frequency probes. Several ultrasound devices from the following brands were used: Phillips U-22, Sonoscope Light, Sonoscope S-50 and Mindray.
6. Materials and Technique of Multiple Passages without Coaxiality (T-MPSC).
Precautions: Oral and written consent was obtained. A haemostasis test was performed 48 hours before the procedure. A platelet count greater than or equal to $150,000 / \mathrm{mm}^3$ and a prothrombin time (PT) greater than or equal to $55\%$ were required for the procedure. We used automatic and semi-automatic biopsy needles of gauge (G) 16 to 14, with a length of 10 to $20~\mathrm{cm}$.
Equipment: After the radiological consultation, we reviewed the examinations and reassessed the lesion using B-mode ultrasound and triplex Doppler with convex and linear multi-frequency probes. Several ultrasound machines of the following brands were used: Phillips U-22, Sonoscope Light, Sonoscope S-50 and Mindray. The patient was placed in a left lateral decubitus position for 40 minutes to an hour after the procedure, before being allowed to return home or to the hospital ward. On discharge, painkillers were systematically prescribed, but antibiotic prophylaxis was recommended on a case-by-case basis in consultation with the referring physician.
## III. RESULTS
Analysis of patients' socio-demographic characteristics revealed that: the age groups most affected in the present series were those $\leq 25$ years with $33.3\%$ and those between 48-58 and 59-69 years with respectively: 22.2 and $22.2\%$. Table I. Gender analysis, shows a predominance of males in the present series with $66.7\%$ against $33.3\%$ of females. Table I. Clinical characteristics of patients: splenomegaly was the most frequent clinical parameter, accounting for $44.44\%$ of cases.
Table II. The sensation of a mass in the left hypochondrium was found in $33.33\%$ of patients in the series. Table II. Polytransfusion and other symptoms such as weight loss, anorexia, untimed fever, abdominal bloating, including left hypochondrium pain were respectively found in $11.1\%$. Table II. In relation to the anatomopathological characteristics of patients, lymphomatous origin came out on top at $33.3\%$. Table III; $11.1\%$ for CD20-negative diffuse large-cell lymphoma, $11.2\%$ for non-Hodgkin's lymphoma and $11.1\%$ for NOS hepatosplenic T-cell lymphoma. Splenic tuberculosis was found in $22.2\%$ of patients, while Gauchier's disease was also found in $22.2\%$.
Table III. In terms of guidance methods, ultrasound was the most widely used radiological guidance method, accounting for $88.9\%$. Table III. Computed tomography was used in $11.1\%$ of patients in the series. Table III. With regard to sampling equipment, the 14-gauge automatic gun was the most widely used, accounting for $44.4\%$ of patients, versus the semi-automatic 14-gauge gun, which was used by $33.3\%$.
Table IV. In relation to the guidance technique, the co-axial or indirect technique was used in almost all cases (100.0%). Table IV. With regard to core sampling, we used our own technique, known as the multiple-pass technique without co-axial, abbreviated T-MPSC. Table IV. Local anesthesia was used in almost all cases (100%). Table IV. No complications, minor or major, were encountered in this series. Table IV. Eight patients in the series (44.44%) had disturbed haemostasis levels below 10 g/dl. Hemoglobin levels were 7 g/dl in two patients and 10 g/dl in six. Psychological preparation was carried out in almost all patients (100.0%); analgesics, atropine premedication and antibiotic prophylaxis were instituted in almost all patients in the series (100.0%).
IV. With regard to the number of cores taken for pathological diagnosis, the majority of patients in the series (88.9%) had 4-5 cores. In 11.1% of patients, the number of cores taken was more or less than 4 samples.
## IV. DISCUSSION
The spleen is not commonly affected by diseases; however, those that do affect it are numerous, including malignancies (lymphoma, metastatic), infections (tuberculosis, fungi) and infiltrative processes such as sarcoidosis [2,3]. Analysis of sociodemographic characteristics revealed a predominance of age groups $\leq 25$ years with $33.3\%$ and those between 48-58 and 59-69 years with respectively: 22.2 and $22.2\%$. As for gender, we found a male predominance of $66.7\%$ out of a total of eighteen patients corrected in the present study. All our patients underwent radio-guided biopsy sampling. A review of the literature reveals that there are currently six techniques for obtaining splenic tissue samples for pathological evaluation. The surgical techniques complement open biopsy, open splenectomy, laparoscopic splenectomy and laparoscopic biopsy. Splenectomy is associated with relatively high morbidity $(8.6\% -37\%)$ and mortality $(0\% -2.9\%)$, mainly due to infection [15-18], but the current surgical trend is to preserve the spleen whenever possible. Asplenia is known to predispose to infection. A particularly important and specific infectious complication of splenectomy is post-pleenectomy infection. It is caused by encapsulated organisms such as Streptococcus pneumoniae, Neisseria meningitidis and Hemophilus influenzae type B. This condition occurs at an annual frequency rate of $0.5\%$ in splenectomy patients, and is associated with a mortality rate of $50\%$. Thrombosis, particularly portal vein thrombosis, is a well-known complication of splenectomy. Improvements in the precision and frequency of imaging examinations have led to increased detection of portal vein thrombosis. As for the sampling equipment used, the 14-gauge automatic gun was the most widely used at $44.4\%$, compared with the semi-automatic 14-gauge gun, used at $33.3\%$. The use of the 14 G gun could be explained by the fact that it was more available in our stock during the study period; however, no major complications were found in the present series. The results of our study corroborate the results of the study by Wani, et al [19] who like us had no major complications, but there were $11.11\%$ minor complications. However, Lindgren et al [14] reported on a series of 32 spleen biopsies performed with a 14G needle: four out of 32 patients $(12.5\%)$ presented with major bleeding during the procedure, requiring transfusion [14]. More recent series have instead shown lower complication rates [7,8,20-26 J, but focused on fine-needle aspiration biopsy (FNAB) having included a relatively small number. The meta-analysis by McInnes et al [20] examined pooled complication rates for FNAB and core needles biopsy (CNB) and found an overall complication rate of $4.2\%$, with a major complication rate of $2.5\%$. with a major complication rate of $2.2\%$; these figures rise to $5.8\%$ and $3.2\%$, respectively, for CNB alone. Oslon et al [27], of their 92 biopsies, had seven $(7.2\%)$ minor complications and only one $(1.0\%)$ major complication. Data from our study reported a major complication rate $(0\%)$, similar to that reported $0\%$ in the study published by Patel et al [28], in which major and minor complication rates were $0\%$ and $1.9\%$ respectively $(1/52)$. However, studies including McInnes et al [30] "Percutaneous Image-guided Biopsy of the Spleen: Systematic Review and Meta-Analysis of the Complication Rate and Diagnostic Accuracy" showed a major complication rate of $1.3\%$ $(95\%$ CI: $0.6\%, 2.5\%)$ for biopsies performed with an 18-gauge needle. In their study, Olson et al [29] found $7.2\%$ minor complications and $1.0\%$ major complications. of minor complications and a major complication rate of $1.0\%$. The overall complication rate was $8.2\%$ ( $n = 8$ ). In these studies, the average major complication was $1.2\%$, and one study showed $7.2\%$ minor complications. In our study, minor complications were not highlighted, although they amounted to simple pain in the left hypochondrium, soothed by post-procedural analgesics. In relation to radiological guidance, the co-axial or indirect technique was used in all our patients ( $100.0\%$ ). The splenic tissue samples were collected using our own technique, known as the co-axial-free multiple-pass technique (abbreviated T-MPSC). The present study demonstrated a diagnostic accuracy of around $100\%$. This is superior to that reported in the study by Wani, et al [19] $94.12\%$ with a $95\%$ confidence interval [CI], $71.5\%$, $71.3\%-99.85\%$ and a diagnostic yield of $94.4\%$. Sensitivity was $93.7\%$ ( $CI,69.77\%-99.84\%$ ) and, as there were no false-positive biopsies, specificity was $100\%$ ( $95\%$ CI, $2.5\%-100\%$ ). In their study, Gomez-Rubio et al [29] demonstrated that the diagnostic accuracy of splenic biopsy was $92\%$, and that for splenic lymphomas, the accuracy of NBC was $100\%$. Olson et al [27] showed that diagnostic accuracy was $94.5\%$.
## V. CONCLUSION
Radiation-guided percutaneous biopsy of splenic lesions with automatic or semi-automatic 14-16 G tru-cuts, using the coaxial-free multiple-pass technique (T-MPSC), yields large-calibre tissue material, facilitating accurate diagnosis during pathological analysis. This technique also avoids embolization of the co-axial pathway and per- or post-biosurgical haemorrhagic complications. In conclusion, percutaneous radio-guided splenic biopsy is an effective alternative to splenectomy in patients with single or multiple splenic lesions. Percutaneous tru-cutaneous splenic biopsy is a safe procedure, with no evidence of an increased risk of major complications compared with surgical splenic biopsy.
## VI. LIMITATIONS
This is a retrospective study with a small sample size. Study Merits:
This is the first study to be carried out in the DRC, and in the hospital environment of Kinshasa in particular, which introduced a new biospic technique for the sampling of splenic tissue, known as T-MPSC. This study also showed that splenic biopsies could be performed with 14G tru-cut without major complications.
Protection of Human and Animal Rights:
The authors declare that this study did not involve experimentation on patients, subjects or animals.
### Confidentiality of Data
The authors declare that this study does not contain any personal data that could identify the patient or subject.
#### Study Funding
This study has not received specific funding from any public or private institution.
#### Declaration of Conflict of Interest
The authors declare that they have no conflict of interest in relation to this article.
Table I: Socio-Demographic Characteristics of Patients
<table><tr><td>Age (years)</td><td>Count</td><td colspan="2">Percentage</td></tr><tr><td>≤25</td><td>6</td><td colspan="2">33,3</td></tr><tr><td>26-36</td><td>2</td><td colspan="2">11,1</td></tr><tr><td>48-58</td><td>4</td><td colspan="2">22,2</td></tr><tr><td>59-69</td><td>4</td><td colspan="2">22,2</td></tr><tr><td>≥70</td><td>2</td><td colspan="2">11,1</td></tr><tr><td>Total</td><td>18</td><td colspan="2">100,0</td></tr><tr><td>Sex</td><td>Count</td><td colspan="2">Percentage</td></tr><tr><td>F</td><td>6</td><td colspan="2">33,3</td></tr><tr><td>M</td><td>12</td><td colspan="2">66,7</td></tr><tr><td>Total</td><td>18</td><td colspan="2">100,0</td></tr><tr><td rowspan="2">Age</td><td colspan="2">Sexe</td><td rowspan="2">Total</td></tr><tr><td>F</td><td>M</td></tr><tr><td>≤25</td><td>4</td><td>2</td><td>6</td></tr><tr><td>26-36</td><td>0</td><td>2</td><td>2</td></tr><tr><td>48-58</td><td>0</td><td>4</td><td>4</td></tr><tr><td>59-69</td><td>2</td><td>2</td><td>4</td></tr><tr><td>≥70</td><td>0</td><td>2</td><td>2</td></tr><tr><td>Total</td><td>6</td><td>12</td><td>18</td></tr></table>
Tableau II: Clinical Information of Patients
<table><tr><td>Clinical information</td><td>Count</td><td>Percentage</td></tr><tr><td>• Other: physical asthenia, weight loss, anorexia, fever without a specific time, abdominal bloating, pain in the left hypochondrium</td><td>2</td><td>11,11%</td></tr><tr><td>• Massive splenomegaly</td><td>8</td><td>44,44%</td></tr><tr><td>• Sensation of a splenic mass</td><td>6</td><td>33,33%</td></tr><tr><td>• History of transfusion</td><td>2</td><td>11,11%</td></tr><tr><td>Total</td><td>18</td><td>100,00</td></tr></table>
Table III: Histopathological Results of Patients and Methods of Guidance used
<table><tr><td>Histopathological Results</td><td>Effectifs</td><td>Pourcentages</td></tr><tr><td>• Diffuse large B-cell lymphoma CD 20 negative</td><td>2</td><td>11,1</td></tr><tr><td>• Non – hodgkin lymphoma</td><td>2</td><td>11,1</td></tr><tr><td>• Hepatosplenic T-cell lymphoma NOS</td><td>2</td><td>11,1</td></tr><tr><td>• Gaucher's disease</td><td>4</td><td>22,2</td></tr><tr><td>• Not available</td><td>4</td><td>22,2</td></tr><tr><td>• Tuberculose splénique</td><td>4</td><td>22,2</td></tr><tr><td>Total</td><td>18</td><td>100,0</td></tr></table>
<table><tr><td>Imaging</td><td>Count</td><td>Pourcentage</td></tr><tr><td>• Ultrasound</td><td>16</td><td>88,9</td></tr><tr><td>• CT scan</td><td>2</td><td>11,1</td></tr><tr><td>• Arteriography</td><td>0</td><td>0</td></tr><tr><td>• MRI</td><td>0</td><td>0</td></tr><tr><td>• Fluoroscopy</td><td>0</td><td>0</td></tr><tr><td>• Cone beam CT</td><td>0</td><td>0</td></tr><tr><td>• Ultrasound + CT scan</td><td>0</td><td>0</td></tr><tr><td>Total</td><td>18</td><td>100,0</td></tr></table>
<table><tr><td colspan="2">Imaging techniques used</td><td colspan="2">Total</td></tr><tr><td>Histopathological Results</td><td>CT SCAN</td><td>Echographie</td><td>Total</td></tr><tr><td>• Diffuse large B-cell lymphoma CD 20 negative</td><td>0</td><td>2</td><td>2</td></tr><tr><td>• Non-hodgkin lymphoma</td><td>0</td><td>2</td><td>2</td></tr><tr><td>• Hepatosplenic T-cell lymphoma NOS</td><td>0</td><td>2</td><td>2</td></tr><tr><td>• Gaucher's disease</td><td>0</td><td>1</td><td>1</td></tr><tr><td>• Not available</td><td>0</td><td>7</td><td>7</td></tr><tr><td>• Splenic tuberculosis</td><td>2</td><td>2</td><td>4</td></tr><tr><td>Total</td><td>2</td><td>16</td><td>18</td></tr></table>
Tableau IV: Techniques used, Types of Guns used, Type of Anesthesia, Hemostasis Balance and Complications after Surgery
<table><tr><td>Types of needles</td><td>Automatic needle</td><td>Aiguille semi-automatique</td><td>Aiguille aspiratif</td></tr><tr><td>G14</td><td>8</td><td>6</td><td>0</td></tr><tr><td>G16</td><td>0</td><td>4</td><td>0</td></tr><tr><td>G17</td><td>0</td><td>0</td><td>0</td></tr><tr><td>G18</td><td>0</td><td>0</td><td>0</td></tr><tr><td>Total</td><td>8(44,4%)</td><td>10(55,6%)</td><td>0</td></tr><tr><td>Technique used</td><td>Count</td><td>Pourcentages</td><td>Total</td></tr><tr><td>Co-axial technique (indirect)</td><td>18</td><td>100,0</td><td>100,0</td></tr><tr><td>Axial technique (direct)</td><td>0</td><td>0</td><td>0</td></tr><tr><td>Embolization of the tract</td><td>0</td><td>0</td><td>0</td></tr><tr><td>Multiple passage sampling technique without co-axial (T-MPSC)</td><td>18</td><td>100,0</td><td>100,0</td></tr><tr><td>Type of anesthesia</td><td></td><td>Oui</td><td>Non</td></tr><tr><td>Local</td><td>With preservative</td><td>0</td><td>18</td></tr><tr><td></td><td>Without preservative</td><td>18</td><td>0</td></tr><tr><td>General</td><td></td><td>0</td><td>146</td></tr><tr><td>Produit utilize</td><td>Count</td><td>Pourcentage</td><td>Total</td></tr><tr><td>Lidocaine (5ml)</td><td>18</td><td>100,0</td><td>100,0</td></tr><tr><td>Lidocaine+Bicarbonate</td><td>0</td><td>0</td><td>0</td></tr><tr><td>Complications</td><td>Count</td><td>Pourcentages</td><td>Total</td></tr><tr><td>Minor incidents</td><td>0</td><td>0</td><td>0</td></tr><tr><td>Major incidents</td><td>0</td><td>0</td><td>0</td></tr><tr><td>Assessment</td><td>Result</td><td>Effectifs</td><td>Pourcentages</td></tr><tr><td></td><td><10</td><td>8</td><td></td></tr><tr><td></td><td>≥10</td><td>10</td><td></td></tr><tr><td>Hematocrite level</td><td><30</td><td>8</td><td></td></tr><tr><td></td><td>≥30</td><td>10</td><td></td></tr><tr><td>Coagulation time</td><td>Normal</td><td>18</td><td>100,0</td></tr><tr><td>Bleeding time</td><td>Normal</td><td>18</td><td>100,0</td></tr><tr><td>Blood group</td><td></td><td></td><td></td></tr><tr><td>Preparationbefore the procedure</td><td>Count</td><td>Pourcentages</td><td>Total</td></tr></table>
<table><tr><td>Psychologicalpreparation</td><td>18</td><td>100,0</td><td>100,0</td></tr><tr><td>Premedication</td><td>18</td><td>100,0</td><td>100,0</td></tr><tr><td>Hemostaticassessment</td><td>18</td><td>100,0</td><td>100,0</td></tr><tr><td>Proton pump inhibitors</td><td>18</td><td>100,0</td><td>100,0</td></tr><tr><td>Minor analgesics</td><td>18</td><td>100,0</td><td>100,0</td></tr><tr><td>Antibioticprophylaxis</td><td>18</td><td>100,0</td><td>100,0</td></tr></table>
#### Iconography
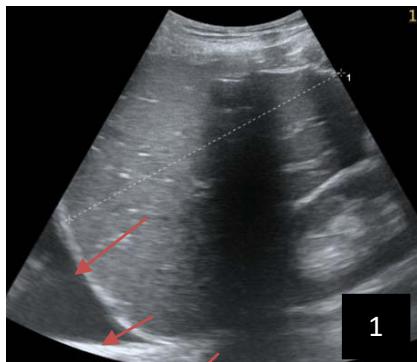
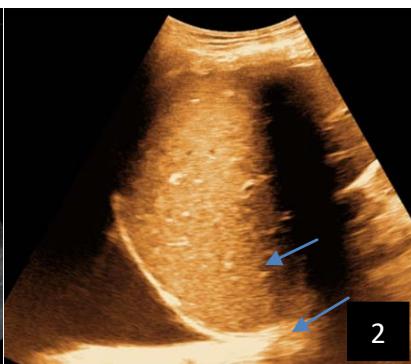
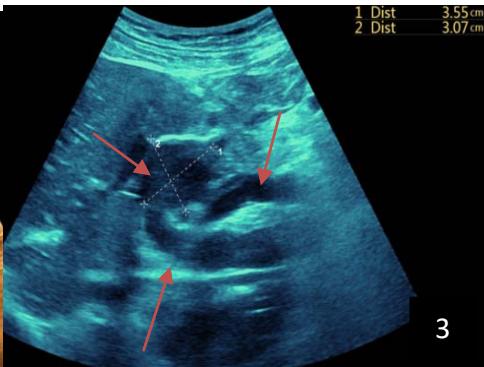
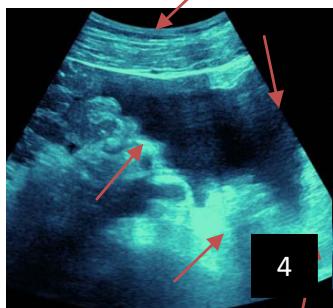
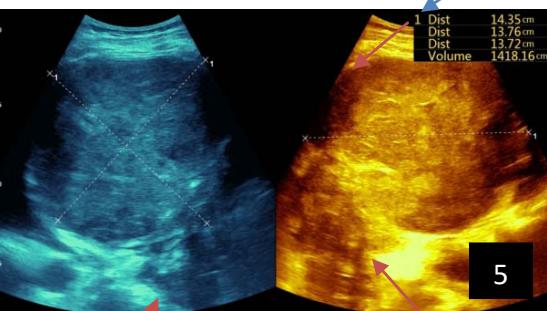
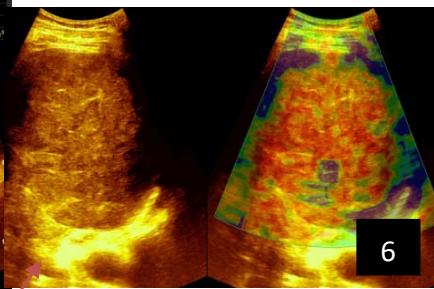
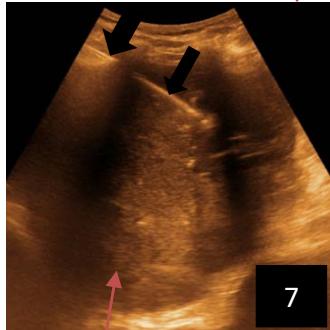
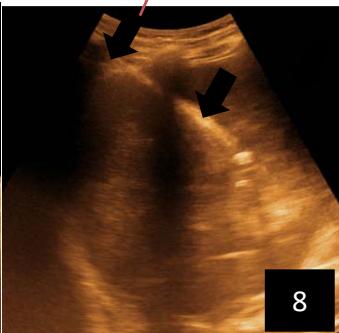
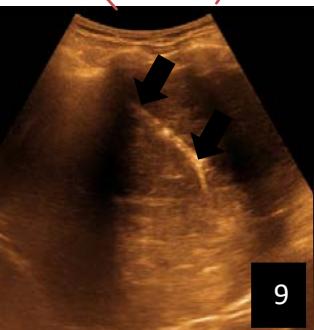
Figure 1: Number of Corescollected
9
10
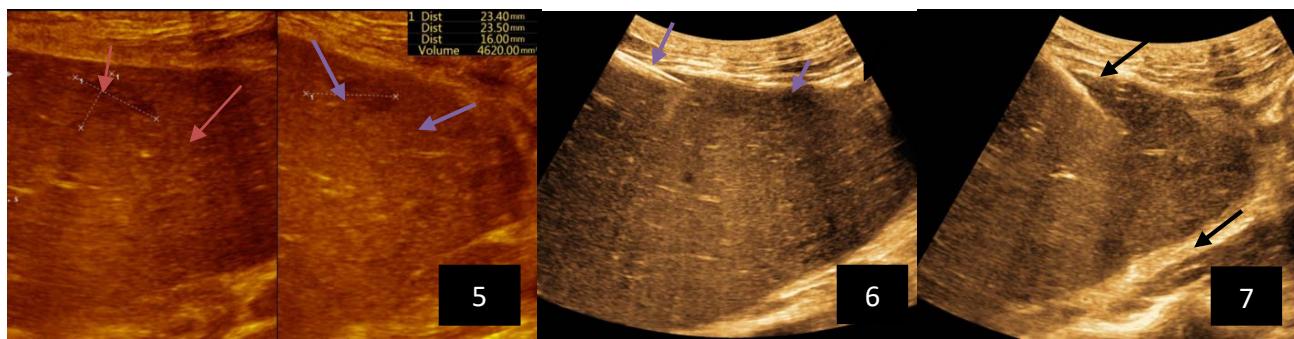
Iconography 1: 54-year-old patient with an intra-splenicsolid mass, anatomopathologically compatible with lymphoma: images 1 and 2: right pleurisy of moderateabundance, image3: celiacadenopathy, image 4: free ascites of moderateabundance, images 5 and 6: intra-splenicsolid mass of around $1418.16\mathrm{ml}$ in B mode and elastographyshowing areas of tumourrigidity, images 7-10: oblique linear images, showingbiopsy gun tracks.
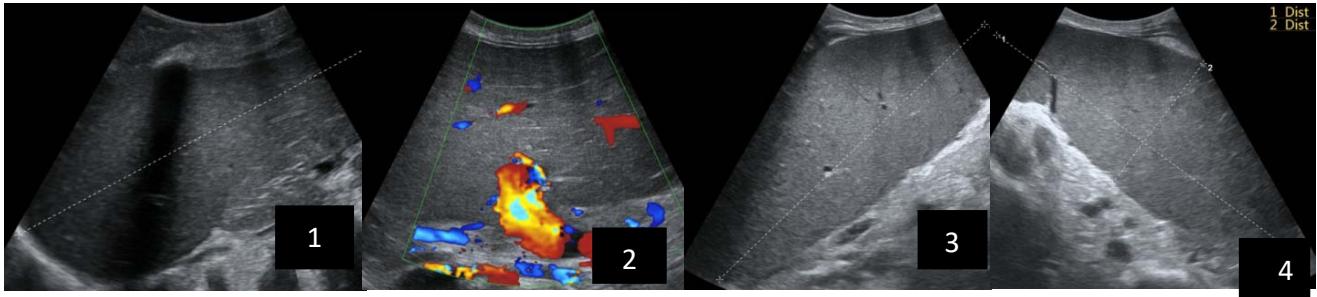
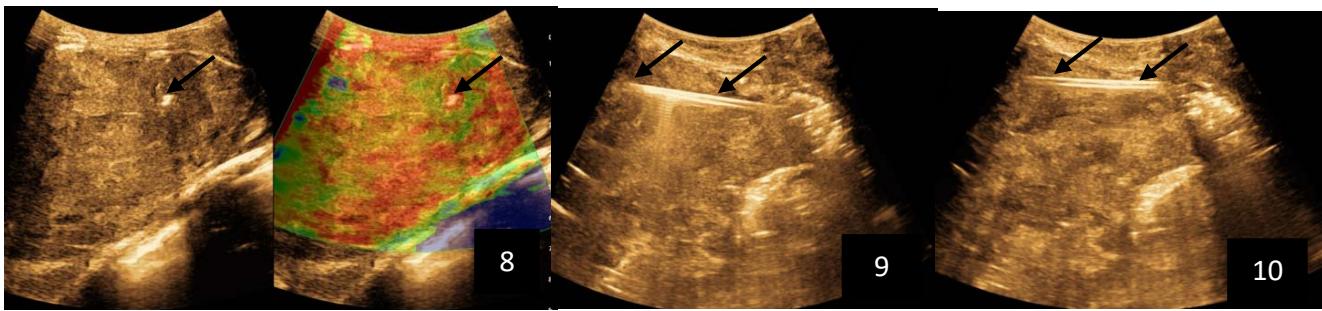
Iconography B: 1 to 4: ultrasound images in mode B showing a large size, 5: ultrasound image in mode B showingsolidhyperechogenic nodules under the capsulorhexis, 6 to 10: ultrasound images in mode B showinglinearhyperechogenic images without overlay in relation to the trajectories of the aiguilles of biopsy.
Generating HTML Viewer...
References
28 Cites in Article
Frank Deland (1970). Normal Spleen Size.
Kunwar Bhatia,Anju Sahdev,Rodney Reznek (2007). Lymphoma of the Spleen.
Eva Compérat,Armelle Bardier-Dupas,Philippe Camparo,Frédérique Capron,Frédéric Charlotte (2007). Splenic Metastases: Clinicopathologic Presentation, Differential Diagnosis, and Pathogenesis.
Giuseppe Civardi,Daniele Vallisa,Raffaella Bertè,Antonio Giorgio,Carlo Filice,Marcello Caremani,Eugenio Caturelli,Maurizio Pompili,Ilario De Sio,Elisabetta Buscarini,Luigi Cavanna (2001). Ultrasound‐guided fine needle biopsy of the spleen: High clinical efficacy and low risk in a multicenter Italian study.
Alda Tam,Savitri Krishnamurthy,Edmund Pillsbury,Joe Ensor,Sanjay Gupta,Ravi Murthy,Kamran Ahrar,Michael Wallace,Marshall Hicks,David Madoff (2008). Percutaneous Image-guided Splenic Biopsy in the Oncology Patient: An Audit of 156 Consecutive Cases.
Ali Cadili,Chris De Gara (2008). Complications of Splenectomy.
Machado No,C Grant,S Alkindi (2009). Splenectomy for haematological disorders: a single center study in 150 patients from Oman.
Gustav Lehne,Einar Hannisdal,Ruth Langholm,Ole Nome (1994). A 10-year experience with splenectomy in patients with malignant non-Hodgkin's lymphoma at the Norwegian Radium Hospital.
Thomas Atwell,Ryan Smith,Gina Hesley,Matthew Callstrom,Cathy Schleck,W Harmsen,J Charboneau,Timothy Welch (2010). Incidence of Bleeding After 15,181 Percutaneous Biopsies and the Role of Aspirin.
A Bravo,S Sheth,S Chopra (2001). Liver biopsy.
F Piccinino,E Sagnelli,G Pasquale,G Giusti (1986). Complications following percutaneous liver biopsy: a multicentre retrospective study on 68, 276 biopsies.
O Hergesell,H Felten,K Andrassy,K Kuhn,E Ritz (1998). Safety of ultrasound-guided percutaneous renal biopsy-retrospective analysis of 1090 consecutive cases.
M Keogan,K Freed,E Paulson,R Nelson,L Dodd (1999). Imaging-guided percutaneous biopsy of focal splenic lesions: update on safety and effectiveness..
Per Lindgren,Hans Hagberg,Barbro Eriksson,Bengt Glimelius,Anders Magnusson,Christer Sundström (1985). Excision biopsy of the spleen by ultrasonic guidance.
M Gómez-Rubio,A López-Cano,P Rendón (2009). Safety and diagnostic accuracy of percutaneous ultrasound-guided biopsy of the spleen: a multicenter study.
Ali Cadili,Chris De Gara (2008). Complications of Splenectomy.
N Machado,C Grant,S Alkindi,S Daar,N Al-Kindy,Al Lamki,Z (2009). Splenectomy for haematological disorders: A single center study in 150 patients from Oman.
G Lehne,E Hannisdal,R Langholm,O Nome (1994). A 10year experience with splenectomy in patients with malignant non-Hodgkin's lymphoma at the Nor wegian Radium Hospital.
M Mcinnes,A Kielar,D Macdonald (2011). Imageguided percutaneous splenic biopsy: systematic review and meta-analysis of complication rate and diagnostic accuracy.
(2009). Modified from Common Terminology Criteria for Adverse Events (CTCAE) (version 3.0).
Alda Tam,Savitri Krishnamurthy,Edmund Pillsbury,Joe Ensor,Sanjay Gupta,Ravi Murthy,Kamran Ahrar,Michael Wallace,Marshall Hicks,David Madoff (2008). Percutaneous Image-guided Splenic Biopsy in the Oncology Patient: An Audit of 156 Consecutive Cases.
Sivan Lieberman,Eugene Libson,Bella Maly,Pinchas Lebensart,Dina Ben-Yehuda,Allan Bloom (2003). Imaging-Guided Percutaneous Splenic Biopsy Using a 20- or 22-Gauge Cutting-Edge Core Biopsy Needle for the Diagnosis of Malignant Lymphoma.
A Tam,S Krishnamurthy,E Pillsbury,J Ensor,S Gupta,R Murthy (2008). Image-guided percutaneous splenic biopsy in the oncologic patient: An audit of 156 consecutive cases.
T Siniluoto,M Päivänsalo,T Tikkakoski,M Apaja-Sarkkinen (1992). Ultrasound-Guided Aspiration Cytology of the Spleen.
Brian Lucey,Giles Boland,Michael Maher,Peter Hahn,Debra Gervais,Peter Mueller (2002). Percutaneous Nonvascular Splenic Intervention: A 10-Year Review.
M Olson,T Atwell,W Harmsen,R King,Y Lin,D Wall (2015). Safety and accuracy of image-guided percutaneous splenic biopsy.
Nirav Patel,Gemma Dawe,Ken Tung (2015). Ultrasound-guided percutaneous splenic biopsy using an 18-G core biopsy needle: our experience with 52 cases.
Tae-Seok Seo,Joo Oh,Yup Yoon,Joo Lim,Seong Park,Sung-Goo Chang,Yang Jeon (2000). Acetic acid as a sclerosing agent for renal cysts: Comparison with ethanol in follow-up results.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
Tshibasu Tshienda. 2026. \u201cSafety and Diagnostic Accuracy of Biopsy of Targeted Splenic Lesions Under Ultrasound Guidance Using the Multiple-Pass Technique Without Co-Axial in Kinshasa Hospitals\u201d. Global Journal of Medical Research - K: Interdisciplinary GJMR-K Volume 25 (GJMR Volume 25 Issue K2): .
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
The most frequent splenic pathologies can pose a diagnostic challenge to clinicians, radiologists and pathologists. These pathologies are innumerable, and may be of malignant or benign tumoral origin. Six techniques are currently available to obtain splenic tissue samples for pathological evaluation. Less invasive percutaneous techniques, performed by an interventional radiologist under ultrasound or CT guidance, include fine-needle aspiration biopsy and core tissue biopsy. They are associated with fewer complications and greater precision.
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.
Lorem ipsum dolor sit amet, consectetur adipiscing elit. Ut elit tellus, luctus nec ullamcorper mattis, pulvinar dapibus leo.
Safety and Diagnostic Accuracy of Biopsy of Targeted Splenic Lesions Under Ultrasound Guidance Using the Multiple-Pass Technique Without Co-Axial in Kinshasa Hospitals