## I. INTRODUCTION
The causes of the onset of pain in diabetic patients are diverse and include mainly the metabolic deficit of the neuronal cell and reduced vascular supply, currently, these two factors are identified as the main reasons for early hospitalization and responsible for $50\%$ of amputations of lower members globally $^{1-3}$.
The repercussions of diabetes mellitus (DM) in face of vascular impairment is centered on the damage at the level of peripheral nerve cells, thus the microvascular deficit and the constant increase in free radicals generate impairment in the non-myelinated fibers of the sympathetic system and in the cholinergic-type vasodilator nerve endings, reducing the release of acetylcholine (ACh) and limiting the production of nitric oxide (ON), by means of the enzyme nitric oxide synthase (eONs) $^{4}$.
Nervous impairment resulting from diabetes mellitus not only affects the sensorimotor nerve fibers but, at an advanced stage, can have repercussions for the peripheral autonomic fibers $^{5}$, thus the diabetic distal polyneuropathy is the most frequent of the neuropathies, affecting mainly the lower extremities $^{6-8}$.
The sensory axons are the most affected because they have a larger mitochondrial population, leaving them in a condition more vulnerable to oxidative stress $^{9,10}$. Diabetic polyneuropathy is identified as the main factor for the appearance of clinical signs and symptoms in these patients, since $20\%$ of patients may experience neuropathic pain $^{11}$.
The choice of a non-pharmacological therapeutic line, which involves the relief of neuropathic pain in the clinical environment, still deserves further investigation. Clinical and experimental evidence suggests that changes in cell function, resulting in oxidative stress, act as a major factor in the development and progression of pain, diabetic neuropathies $^{12}$.
The great effects resulting from photobiomodulation (laser or LED) are attributed to the heme cofactors existing in erythrocytes and mitochondria. Thus, half of the pho-tons generated by the irradiation are attenuated by the enzyme cytochrome C oxidase of the respiratory chain, although hemoglobin, myoglobin, and melanin may also present a high attenuation coefficient for photons, mainly in the red light spectrum $^{13,14}$.
Today, in clinical practice, two resources are used for photobiomodulation (Laser and LED), although the laser emits collimated light, different of LED light, both have the same therapeutic effect $^{15}$, since this difference ends when the light reaches the biological tissues, due to its dispersion.
Scientific evidence shows that some biological tissues have a high coefficient of therapeutic light attenuation (photobiomodulation), such as the skin (higher melanin concentration) $^{16}$, the erythrocyte $^{17,18}$, nervous tissue $^{19-21}$, and muscle tissue $^{22,23}$. Thus, when the light is absorbed by photoreceptors, photonic energy is trans-formed into chemical energy, making photobiomodulation a non-pharmacological alter-native that can generate an increase in energy metabolism $^{24,25}$, vasodilation, in-creased blood flow, angiogenesis, increased inflammatory response, accelerated healing process $^{26,27}$, increased oxygen affinity hemoglobin $^{18}$, and reduction of neuro-pathic symptoms through cytokine stimulation and release mechanisms $^{28}$.
Given the above, the purpose of this study was to show that photobiomodulation with light emitting diode (LED) can improve the signs and symptoms of pain generated by the metabolic disorder of lower limbs in diabetic patients with polyneuropathy (Painful polyneuropathy), from the effects of metabolic increase and favoring biophysical photobiomodulation responses.
## II. MATERIALS AND METHODS
### a) Ethical aspects
This study is a randomized double-blinded placebo-controlled clinical trial, it was approved by the Ethics Committee on Experimentation with Human Beings of the Clinical Hospital - FMRP/USP (process no. 3.805.967) and registered as a clinical trial on ClinicalTrials.gov. (NCT03369834). Patients who agreed to participate in the study were informed about the objectives and procedures and signed a free and informed consent form. The study was developed from April 2018 to March 2020.
### b) Sample and Randomization
The sample calculation was performed using the Ene® software (version 3.0, Barcelona, Spain). The sample size was calculated based on the study by Lorne et al. (2004) [29], which evaluated pain in diabetic polyneuropathy using laser as a therapeutic tool. We considered the average values of the Visual Analog Pain Scale (VAS), based on the four evaluation periods. The calculation was based on the detection of comparison between the groups, with a mean of the reference group of 9 and the experimental group 9. Considering the statistical power of $90\%$ and alpha 0.05, the number of 10 patients per group was estimated. Considering a sample loss of $10\%$, 12 patients were recruited per group.
Seventy patients with type 2 diabetes mellitus of both sexes with more than 5 years of diagnosis at the age of 45 to 70 years old were recruited at the Hospital das Clínicas de HC/USP. 58 of whom had neuropathic pain ranging from 4 to 6 points (DN4), classified as moderate to severe on the scale of diabetic distal polyneuropathy (DSDDP) in the lower limbs. The exclusion criteria involved neurological lesions that that impeded the proposed exams, chronic renal failure, and patients who have had part of the saphenous vein removed for myocardial revascularization. Some of the volunteers took medication to control blood pressure, among them ACE antidiuretics and beta-blockers. All were instructed not to take analgesics or anti-inflammatory drugs during the week of the study.
Randomization was performed using a table of random numbers in the Excel software, which was placed in sealed opaque paper envelopes, opened only in the presence of the patient. Patients were randomly divided into the following groups: Control (C, n = 11), Sham (S, n = 12), Red LED (R, n = 11), Infrared LED (IR, n = 13), Red+Infrared LED (R+IR, n = 11).
Researchers 2 and 3 and the patients themselves were blinded regarding the distribution of groups and the interventions applied. The blinding of the patient in relation to the groups, with the exception of the control, was accomplished with the use of a blindfold. As shown in the flowchart in Figure 1.
Randomization and Allocation
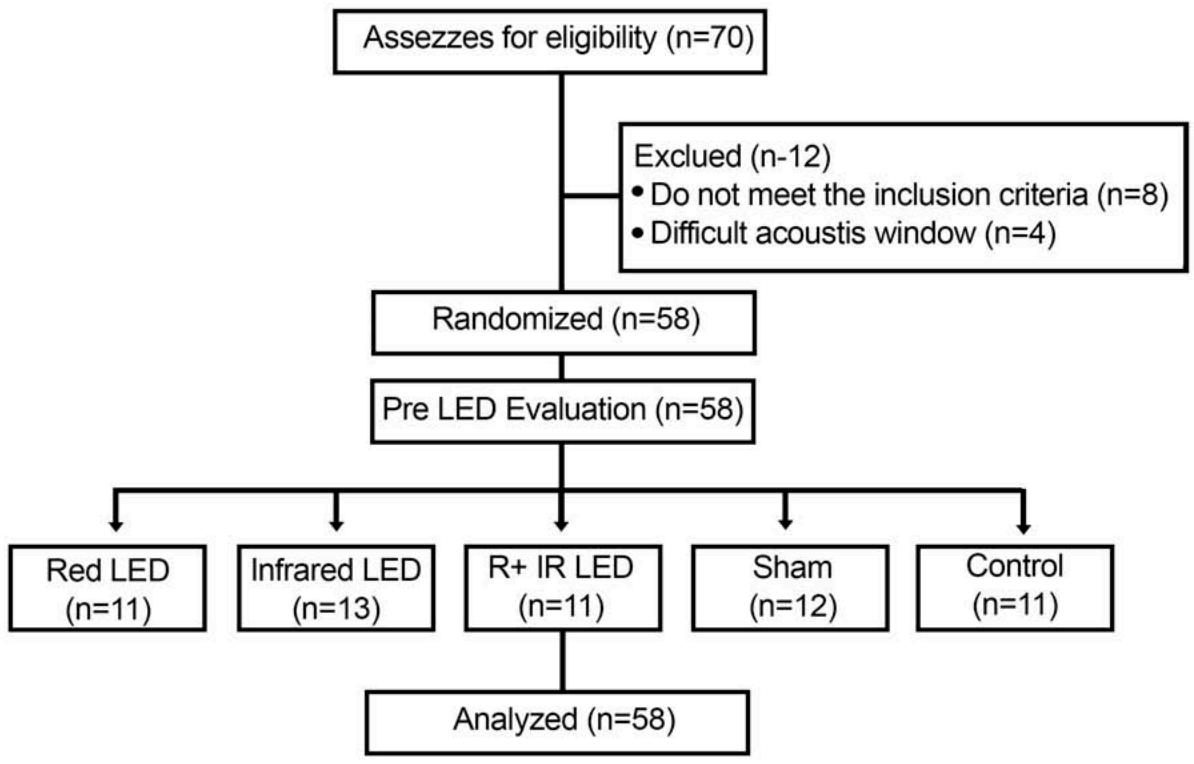 Fig. 1: Flowchart of the study design
### c) Procedure
The study procedures were performed in 5 days. On the first day, the researchers collected the anthropometric and clinical data, applied the diagnostic scale for distal diabetic polyneuropathy (DSDDP) and the neuropathic pain questionnaire (DN4), and evaluated the blood flow in the posterior and dorsal tibial arteries through Doppler ultrasound. From the second to the fourth day, the patients received irradiation (treatment group) and the simulation of application (Sham group) of photobiomodulation with light emitting diode (LED) for one minute and 37 seconds, on average, in each lower limb, totaling 3 minutes per patient, respecting the 24-hour interval between irradiations. On the fifth day, reassessments were carried out using the same assessment tools of the first day (Figure 2).
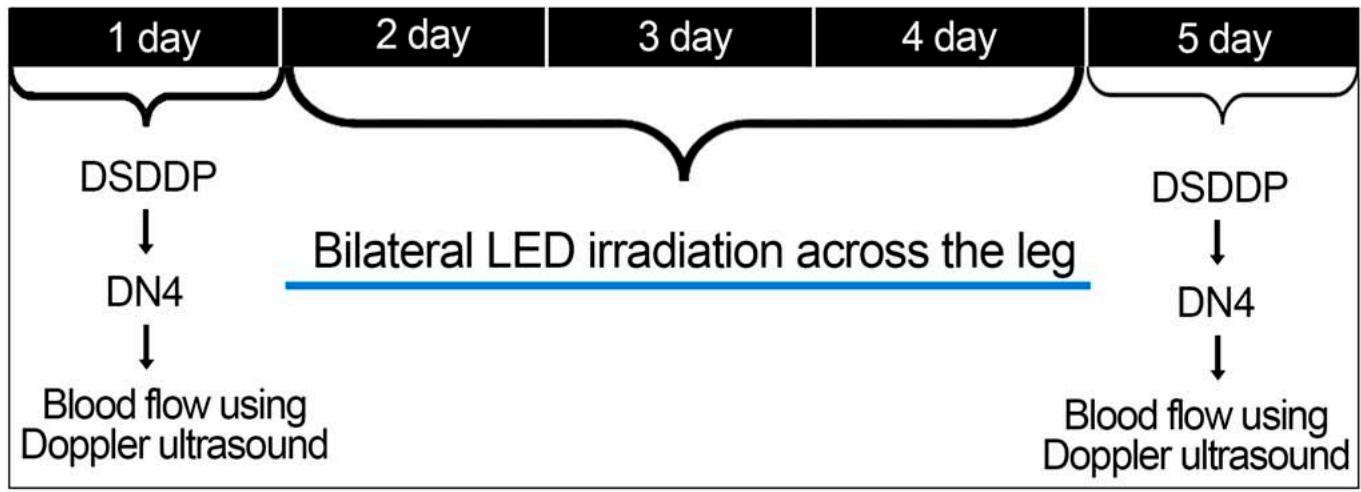 Figure 2: Description of the evaluation, intervention and reassessment during the proposed 5 days.
### d) Assessment of neuropathy
## i. Diagnostic scale for diabetic distal polyneuropathy
To quantify the degree of polyneuropathy, the Diagnostic Scale for Diabetic Distal Polyneuropathy (DSDDP) was used, which evaluates neuropathic symptoms with six questions and neuropathic impairment with the Aquileu reflex tests and the hallux vibratory, painful and thermal sensitivity $^{30}$. Thus, patients should have a score equal to or greater than 5 for symptoms (ESN), associated with a score equal to or greater than 3 for signs (ECN). To characterize neuropathic pain, the patient should have a score equal to or greater than 4 on the Neuropathic Pain Diagnosis questionnaire (DN4).
## ii. Aquileu reflex test
The Aquileu reflex test was performed using Buck's neurological hammer to as-sess motor changes in alpha-thick myelinated A-fibers $^{30}$. With the patient seated, the foot hanging and in a neutral position, percussion was performed with the reflex hammer on the Achilles tendon of both lower limbs. As a positive answer, the volunteer is expected to do reflex plantar flexion, as a consequence of percussion, and the absent or diminished reflex response means an altered result. Therefore, a second percussion was performed to confirm the altered result $^{31,32}$.
## iii. Vibratory sensitivity test
For the vibration sensitivity test, a 128 Hertz frequency tuning fork was used to test changes in betathick myelinated A-fibers $^{30}$. With the patient in the supine position, the vibrating tuning fork was applied with the end of the nail, perpendicular and with constant pressure, on the dorsal portion of the distal phalanx of the hallux and medial malleolus or tibial tuberosity, as an alternative, if the volunteer could not feel it in the first region tested. Two applications were made, alternating with a false application, in which the tuning fork was not vibrating. Because of this, the result should be negative, that is, the absence of protective sensitivity, with two incorrect answers from the three applications. The test was performed on both lower limbs $^{31,32}$. According to Perkins et al. $^{33}$, the vibration test has a sensitivity of $53\%$ and specificity of $99\%$ for peripheral neuropathy.
## iv. Pain and thermal sensitivity test
Buck's neurological hammer needle was used for the painful sensitivity test, to evaluate sensory changes in C-thin unmyelinated fibers[30]. With the volunteer in the supine position, the tip of the reflex hammer was applied to the back of the hallux, bilaterally, with sufficient pressure to deform the skin. An altered result was the inability to feel the pressure exerted on the hallux<sup>31,32</sup>. In the assessment of thermal sensitivity, the test was carried out with a test tube containing ice water to assess the sensitive alterations of delta-thin myelinated A-fibers[30]. With the volunteer in the supine position, a test tube containing ice water was applied to the dorsal region of the feet of both lower limbs, in which the result was considered altered when the patient did not report the cold sensation at the test site $^{33}$.
## v. DN4 Questionnaire for diagnosis of neuropathic pain
For the qualification of neuropathic pain, the DouleurNeuropathique 4 (DN4) questionnaire was used, it has seven questions and three sensory exams, which are able to discriminate neuropathic pain from nociceptive pain. It was translated to Portuguese and validated, with $100\%$ sensitivity and $93.2\%$ specificity [34].
For scoring, DN4 assigns 1 when it is positive and 0 when it is negative (total score range varies from 0 to 10). The cut-off value for the diagnosis of neuropathic pain is a total score equal to or greater than 4
### Assessment of arterial blood flow
A portable continuous-wave Doppler device with spectral analysis (Nicolet Vascular Versalab SE, San Carlos, CA, USA) was used, coupled to a notebook. To capture and process the signals, Care Fusion 7.0 software (Nicolet Vascular Versalab SE, San Carlos, CA, USA) was used, which allows the quantification of blood flow, including the peak systolic velocity and resistivity index, as well as its qualification in an interval of time.
Blood flow was assessed after 10 minutes of rest in the supine position. Collections were performed in the posterior and dorsal tibial arteries, with frequencies of 4 and $8\mathrm{MHz}$, respectively, in both lower limbs.
Photobiomodulation
Irradiation was performed individually in 58 patients with moderate to severe diabetic distal polyneuropathy. Thus, 116 legs were irradiated during the second, third, and fourth days of therapy. The light was irradiated by a $33 \times 42 \mathrm{~cm}^2$ LED mat, using SMD5050 diodes fed with 12-volt voltage and fixed in an ethylenevinyl-acetate (EVA) plate with equidistant distribution (1 cm) between the LEDs [35]. The energy applied to each leg was 180 J, on the anterior and posterior regions, bilaterally. The blanket was malleable to adapt to the contour of the leg, so the application of photobiomodulation was directly on the skin involving the entire leg of the patients (Figure 3).
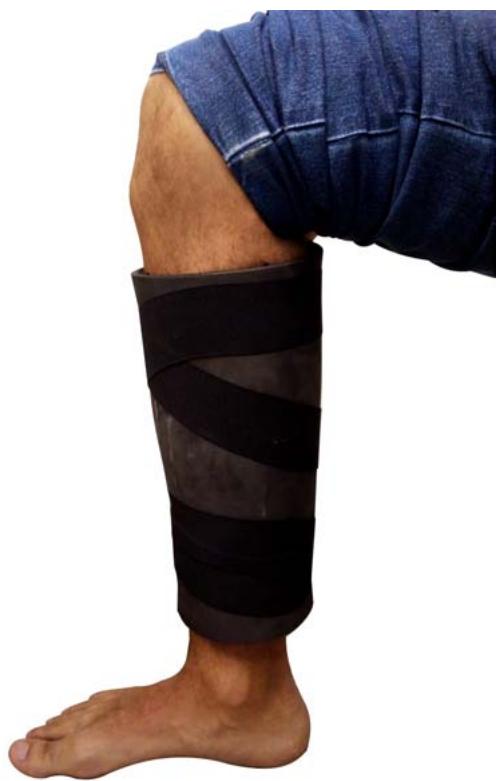 Figure 3: Application of the LED mat directly on the skin involving the whole leg bilaterally.
All LEDs were previously checked at the Photobiophysics Laboratory of the Faculty of Philosophy, Sciences, and Letters of Ribeirão Preto, University of São Paulo, where the wavelengths, power, and power density were evaluated. 285 LEDs were used in both blankets (red, infrared), and 320 LEDs for the mixed blanket, 160 LEDs in the red spectrum (620 nm), and 160 in the infrared spectrum (940 nm) (Table 1).
Table 1: Physical parameters of light-emitting diodes (LED) and photobiomodulation therapy protocol.
<table><tr><td>Variables</td><td>Red</td><td>Infrared</td><td>Red+Infrared</td></tr><tr><td>Wave-length</td><td>620±10nm</td><td>940±10nm</td><td></td></tr><tr><td>Number of diodes</td><td>285</td><td>285</td><td>320</td></tr><tr><td>Diode diameter</td><td>0.125 cm2</td><td>0.178 cm2</td><td></td></tr><tr><td>Power Density</td><td>52.86 mW/cm2</td><td>33.7 mW/cm2</td><td></td></tr><tr><td>Diode power</td><td>0.0066 W</td><td>0.006 W</td><td></td></tr><tr><td>Total blanket power</td><td>1.88 W</td><td>1.71 W</td><td>2.01 W</td></tr><tr><td>Application time</td><td>96 s</td><td>106 s</td><td>90 s</td></tr><tr><td>Total energy per leg</td><td>180 J</td><td>180J</td><td>180 J</td></tr></table>
### e) Statistical analysis
Statistical analysis was performed using SPSS for Windows (version 20; SPSS Inc., Chicago, IL). The mean differences (initial minus final value) of the groups and the $95\%$ confidence intervals (CI) were calculated using mixed linear models, with the baseline as a covariate. The Bonferroni correction test was used to compare the groups, with a p value of 0.05.
To determine the size of the clinical effect of the proposed therapies, Cohen's d was used, with interpretation of the values based on the classification established by Cohen (1988): less than 0.2, small effect; around 0.5, moderate effect; and above 0.8, great effect.
## III. RESULTS
Of the 70 recruited patients, 58 were included and randomized. To characterize the homogeneity of the sample, anthropometric assessments and some clinical routes are available in table 2.
Table 2: Anthropometric and clinical data of patients with type 2 diabetes mellitus, distributed in the experimental groups.
<table><tr><td></td><td></td><td>C
n=11</td><td>S
n=12</td><td>R
n=11</td><td>IR
n=13</td><td>R+IR
n=11</td></tr><tr><td>Age</td><td></td><td>63.66 (4.18)</td><td>61 (5.37)</td><td>61.38 (4.44)</td><td>60.08 (7.31)</td><td>61.3 (6.99)</td></tr><tr><td></td><td>Male</td><td>10</td><td>5</td><td>9</td><td>5</td><td>4</td></tr><tr><td>Sex</td><td>Female</td><td>3</td><td>7</td><td>4</td><td>8</td><td>7</td></tr><tr><td>Weight</td><td></td><td>80.15 (13.78)</td><td>80.5 (18.46)</td><td>89.36 (21.78)</td><td>86.38 (13.42)</td><td>97.54
(19.65)</td></tr><tr><td>BMI</td><td></td><td>30 (4.50)</td><td>30.25 (7.72)</td><td>32.49 (5.72)</td><td>32.39 (4.78)</td><td>36.73 (4.97)</td></tr><tr><td>Time of diagnosis (years)</td><td></td><td>17.23 (6.67)</td><td>14.91 (9.17)</td><td>13.92 (6.34)</td><td>14.08 (6.76)</td><td>16.27 (8.79)</td></tr><tr><td>Arterial hypertension</td><td></td><td>9</td><td>10</td><td>8</td><td>10</td><td>9</td></tr><tr><td>DSDDP</td><td>DM2 Symptoms</td><td>6.46±1.89</td><td>6.76±2.16</td><td>7.8±1.72</td><td>6.25±2.22</td><td>7.00±1.63</td></tr><tr><td></td><td>Signals</td><td>4.61±1.12</td><td>4.07±0.75</td><td>4.54±1.21</td><td>4.00±0.73</td><td>4.23±0.83</td></tr><tr><td>DN4 (pain initial)</td><td></td><td>6.3 (1.94)</td><td>5.6 (1.42)</td><td>6 (2.30)</td><td>6.9 (3.66)</td><td>6.7(1.42)</td></tr></table>
As for the assessment of neuropathic pain by the DN4 questionnaire, after irradiation with LED, there was a significant difference in the comparison between the groups that received the treatment (LED) with the control and sham groups. Specifically, when comparing LED red groups with control and sham, the difference $(p < 0.001)$ between the means (lower limit and upper limit) was -4.18 (-5.95 -2.40) and -4.08 (-5.81 -2.35), respectively; when comparing infrared LED with control and sham there were also differences $(p < 0.001)$ with values of -4.21 (-5.91 -2.50) and -4.11 (-5.77 -2.45) respectively; as well as LED red+infrared compared to control and sham with differences $(p < 0.001)$ of -4.58 (-6.37 -2.80) and -4.49 (-6.23 - 2.74), respectively. In the comparisons between the different groups irradiated with LED, there was no significant difference, as well as between control and sham. The size of the clinical effect of photobiomodulation therapy in the different groups, using Cohen's d, had a great effect on relieving neuropathic pain for all groups irradiated with LED (Table 3).
Table 3: Values of differences between means, $95\%$ confidence interval, upper and lower limits, and assessment of effect size of different groups, considering signs and symptoms of neuropathic pain from the neuropathic Douleur 4 questionnaires (DN4).
<table><tr><td rowspan="2">Comparison between groups</td><td rowspan="2">Difference of means</td><td colspan="2">95% confidence intervals</td><td rowspan="2">Effect size Cohen's d</td></tr><tr><td>Inferior limit</td><td>Upper limit</td></tr><tr><td>R x IR</td><td>0.03</td><td>-1.66</td><td>1.73</td><td>-0.02</td></tr><tr><td>R x R+IR</td><td>0.40</td><td>-1.36</td><td>2.18</td><td>-0.08</td></tr><tr><td>R x C</td><td>-4.18*</td><td>-5.95</td><td>-2.40</td><td>-2.08</td></tr><tr><td>R x S</td><td>-4.08*</td><td>-5.81</td><td>-2.35</td><td>-1.92</td></tr><tr><td>IR x R+IR</td><td>0.37</td><td>-1.33</td><td>2.08</td><td>0.05</td></tr><tr><td>IR x C</td><td>-4.21*</td><td>-5.91</td><td>-2.50</td><td>-2.15</td></tr><tr><td>IR x S</td><td>-4.11*</td><td>-5.77</td><td>-2.45</td><td>-1.61</td></tr><tr><td>R+IR x C</td><td>-4.58*</td><td>-6.37</td><td>-2.80</td><td>-2.18</td></tr><tr><td>R+IR x S</td><td>-4.49*</td><td>-6.23</td><td>-2.74</td><td>-1.07</td></tr><tr><td>C x S</td><td>-0.09</td><td>-1.82</td><td>1.63</td><td>-0.01</td></tr></table>
As for the qualitative assessment of the signs and symptoms of DN4, there was a reduction from $75\%$ to $100\%$ for responses to pain qualification after the application of LED and from $50\%$ to $85\%$ in responses to physical examination of pain. Considering the groups, the table shows that the reduction in pain, with regard to their qualification, occurred in the three groups irradiated in an equitable way, unlike the groups sham and control that did not present any changes. For the answers to the exams, it is observed that only in red+infrared there was pain reduction (Table 4).
Table 4: Number of patients who present the characteristics/symptoms of pain before and after application of LED, in the Douleur neuropathic 4 questionnaire (DN4).
<table><tr><td>DN4</td><td colspan="2">R</td><td colspan="2">IR</td><td colspan="2">R+IR</td><td colspan="2">S</td><td colspan="2">C</td></tr><tr><td>Features/Symptoms</td><td>Pre</td><td>Post</td><td>Pre</td><td>Post</td><td>Pre</td><td>Post</td><td>Pre</td><td>Post</td><td>Pre</td><td>Post</td></tr><tr><td>Burning</td><td>8</td><td>0</td><td>8</td><td>1</td><td>8</td><td>2</td><td>6</td><td>7</td><td>7</td><td>7</td></tr><tr><td>Painful cold sensation</td><td>7</td><td>0</td><td>5</td><td>0</td><td>6</td><td>1</td><td>5</td><td>5</td><td>4</td><td>4</td></tr><tr><td>Electric shock</td><td>8</td><td>1</td><td>8</td><td>1</td><td>7</td><td>0</td><td>5</td><td>5</td><td>7</td><td>7</td></tr><tr><td>Tingling</td><td>7</td><td>1</td><td>11</td><td>1</td><td>8</td><td>1</td><td>8</td><td>8</td><td>10</td><td>10</td></tr><tr><td>Pinned and needled</td><td>8</td><td>0</td><td>11</td><td>1</td><td>7</td><td>1</td><td>6</td><td>6</td><td>6</td><td>6</td></tr><tr><td>Fall asleep</td><td>7</td><td>1</td><td>10</td><td>1</td><td>6</td><td>0</td><td>7</td><td>7</td><td>8</td><td>7</td></tr><tr><td>Itching</td><td>5</td><td>1</td><td>8</td><td>2</td><td>4</td><td>0</td><td>3</td><td>4</td><td>3</td><td>3</td></tr><tr><td>Exams</td><td></td><td></td><td></td><td></td><td></td><td></td><td></td><td></td><td></td><td></td></tr><tr><td>Hypoesthesia to the touch</td><td>2</td><td>3</td><td>1</td><td>1</td><td>7</td><td>1</td><td>5</td><td>5</td><td>6</td><td>6</td></tr><tr><td>Needle prick hypoesthesia</td><td>2</td><td>3</td><td>1</td><td>1</td><td>8</td><td>3</td><td>5</td><td>5</td><td>6</td><td>6</td></tr><tr><td>Brushing</td><td>6</td><td>3</td><td>6</td><td>6</td><td>6</td><td>3</td><td>5</td><td>5</td><td>6</td><td>6</td></tr></table>
Considering that neuropathic pain could be associated with circulatory restriction, we opted for the Doppler flow assessment of the lower limb arteries, distally. The results did not show significant changes $(p > 0.050)$ for the PVS, IR, IP, and HR parameters for any of the groups, despite presenting a clinical effect size between mild to moderate for the groups irradiated with LED (Table 5).

Table 5: Values of the difference in means and the confidence interval of blood flow variables and do Effect Size of the Doppler ultrasound variables for the tibial and dorsal arteries of the lower limbs.
<table><tr><td></td><td>Variáveis</td><td>R x IR</td><td>R x R+IR</td><td>R x S</td><td>R x C</td><td>IR x R+IR</td><td>IR x S</td><td>IR x C</td><td>R+IR x S</td><td>R+IR x C</td><td>S x C</td></tr><tr><td rowspan="4">Dorsal R</td><td>PVS</td><td>4,21(-10.07 18.50)</td><td>2.56(-12.40 17.53)</td><td>3.36(-10.94 17.66)</td><td>-1.38(-16.67 13.91)</td><td>-1.65(-15.65 12.34)</td><td>-0.85(-14.12 12.41)</td><td>-5.59(-20.02 8.82)</td><td>0.80(-13.15 14.76)</td><td>-3.94(-18.82 10.94)</td><td>-4.74(-19.00 9.51)</td></tr><tr><td>ES</td><td>0.34</td><td>0.00</td><td>0.08</td><td>-0.48</td><td>-0.32</td><td>-0.80</td><td>-0.28</td><td>0.07</td><td>-0.46</td><td>0.57</td></tr><tr><td>IR</td><td>0.09(-0.03 0.22)</td><td>0.03(-0.10 0.17)</td><td>-0.02(-0.15 0.11)</td><td>-0.00(-0.14 0.13)</td><td>-0.05(-0.18 0.07)</td><td>-0.11(-0.24 0.00)</td><td>-0.09(-0.23 0.03)</td><td>-0.06(-0.19 0.07)</td><td>-0.04(-0.18 0.09)</td><td>0.01(-0.11 0.15)</td></tr><tr><td>ES</td><td>0.74</td><td>0.42</td><td>-0.22</td><td>0.00</td><td>-0.40</td><td>-0.98</td><td>-0.73</td><td>-0.67</td><td>-0.40</td><td>-0.22</td></tr><tr><td rowspan="4">Tibial R</td><td>PVS</td><td>1.71(-16.15 19.59)</td><td>-1.82(-20.81 17.17)</td><td>-2.66(20.55 15.22)</td><td>-12.89(-31.34 5.54)</td><td>-3.54(-22.32 15.24)</td><td>-4.38(-22.06 13.30)</td><td>-14.61(-32.82 3.59)</td><td>-0.84(-19.64 17.95)</td><td>-11.07(-30.39 8.24)</td><td>-10.23(-28.46 7.99)</td></tr><tr><td>ES</td><td>0.11</td><td>-0.07</td><td>-0.16</td><td>-0.78</td><td>-0.22</td><td>-0.27</td><td>-0.89</td><td>-0.09</td><td>-0.79</td><td>0.73</td></tr><tr><td>IR</td><td>0.05(-0.06 0.18)</td><td>0.00(-0.13 0.13)</td><td>-0.03(-0.16 0.08)</td><td>-0.02(-0.15 0.10)</td><td>-0.05(-0.19 0.07)</td><td>-0.09(-0.22 0.02)</td><td>-0.08(-0.21 0.04)</td><td>-0.04(-0.17 0.09)</td><td>-0.02(-0.16 0.10)</td><td>0.01(-0.11 0.14)</td></tr><tr><td>ES</td><td>0.59</td><td>0.00</td><td>-0.46</td><td>-0.29</td><td>-0.61</td><td>-1.05</td><td>-0.82</td><td>-0.50</td><td>-0.31</td><td>-0.11</td></tr><tr><td rowspan="4">Dorsal L</td><td>PVS</td><td>-0.40(-17.88 17.08)</td><td>-8.11(-26.71 10.49)</td><td>-15.77(-33.26 1.71)</td><td>-9.59(-27.87 8.68)</td><td>-7.70(-25.94 10.52)</td><td>-15.37(-32.48 1.73)</td><td>-9.18(-27.10 8.72)</td><td>-7.66(-25.91 10.59)</td><td>-1.47(-20.47 17.51)</td><td>6.18(-11.73 24.10)</td></tr><tr><td>ES</td><td>-0.03</td><td>-0.54</td><td>-1.02</td><td>-0.54</td><td>-0.63</td><td>-1.17</td><td>-0.63</td><td>-1.17</td><td>-0.09</td><td>-0.39</td></tr><tr><td>IR</td><td>-0.01(-0.15 0.12)</td><td>-0.02(-0.16 0.12)</td><td>-0.08(-0.21 0.05)</td><td>-0.04(-0.19 0.09)</td><td>-0.00(-0.14 0.13)</td><td>-0.06(-0.19 0.06)</td><td>-0.03(-0.17 0.10)</td><td>-0.05(-0.19 0.08)</td><td>-0.02(-0.17 0.12)</td><td>0.03(-0.10 0.17)</td></tr><tr><td>ES</td><td>-0.08</td><td>-0.15</td><td>-0.62</td><td>-0.34</td><td>-0.09</td><td>-0.70</td><td>-0.34</td><td>-0.53</td><td>-0.20</td><td>-0.43</td></tr><tr><td rowspan="4">Tibial L</td><td>PVS</td><td>4.39(-15.34 24.12)</td><td>4.79(-17.58 27.18)</td><td>-1.68(-22.12 18.76)</td><td>-10.81(-31.83 10.19)</td><td>0.40(-21.25 22.06)</td><td>-6.07(-25.71 13.56)</td><td>-15.20(-35.45 5.03)</td><td>-6.47(-28.76 15.80)</td><td>-15.61(-38.44 7.21)</td><td>-9.13(-30.08 11.81)</td></tr><tr><td>ES</td><td>0.29</td><td>0.38</td><td>-0.18</td><td>-0.63</td><td>0.04</td><td>-0.83</td><td>-0.42</td><td>-0.48</td><td>-0.89</td><td>0.42</td></tr><tr><td>IR</td><td>0.04(-0.11 0.20)</td><td>0.04(-0.13 0.22)</td><td>-0.02(-0.19 0.13)</td><td>-0.04(-0.20 0.12)</td><td>0.00(-0.17 0.17)</td><td>-0.08(-0.23 0.07)</td><td>-0.08(-0.25 0.07)</td><td>-0.08(-0.26 0.09)</td><td>-0.08(-0.27 0.09)</td><td>0.00(-0.16 0.17)</td></tr><tr><td>ES</td><td>0.30</td><td>0.34</td><td>-0.35</td><td>-0.45</td><td>0.00</td><td>-0.69</td><td>-0.82</td><td>-0.65</td><td>-0.76</td><td>0.09</td></tr></table>
## IV. DISCUSSION
The hypothesis that photobiomodulation with LED could be effective in reducing the signs and symptoms of neuropathic pain of the lower limbs was confirmed, being an effective therapy in short-term intervention, with a great clinical effect for all groups irradiated with LED.
According to Kallenborn-Gerhardt et al. (2013) $^{36}$, the main causes of pain in peripheral neuropathy still need further studies. In diabetic patients, the emergence of neuropathy is believed to be due to multiple factors, including the increased production of reactive oxygen species at the mitochondrial level, reduced antioxidant capacities such as superoxide dismutase catalase and glutathione in enzymatic and non-enzymatic cells $^{37}$, and vascular impairment generated by chronic hyperglycemia $^{38,39}$. These factors are identified as the main causes of reverse neuroinflammation, which promotes an imbalance in the production of ATP and the induction of apoptosis in nerve cells. Thus, the results of this study open a window so that photobiomodulation can be used as a non-pharmacological alternative, contributing to the reduction of pain generated by painful neuropathy.
In the light of the evidence, the results of Cg et al. (2015) $^{40}$ corroborated the results found in this study, based on the hypothesis that the biophysical responses generated by photobiomodulation may control neurological pain in diabetic patients with polyneuropathy. Thus, it is highlighted that the three LED irradiations in the leg were sufficient to reduce pain in the lower limbs. Gobbi et al. (2020) $^{41}$ report that PBMT applied for a short period does not bring important gains for the muscular performance and functionality of diabetic individuals.
As it is a subjective experience, pain has some limitations regarding its measurement. The use of a standardized and validated questionnaire that involves other aspects than just its intensity is important since it contemplates the subjective experience, involving the affective responses associated with pain. Thus, the application of DN4 sought to expand this analysis.
The quantification of pain through DN4 showed a reduction between 4.7 and 5.5 points, on a 10 point scale, in the LED groups, so it is believed that this response is due to the effects of metabolic increment and favoring biophysical responses generated by photobiomodulation, as proposed by Janzadeh et al. (2016) $^{42}$ in an animal model, where they emphasized that photobiomodulation can generate an increase in the levels of antioxidants, such as superoxide dismutase catalase and glutathione, improve mitochondrial function, safeguard the survival of neural cells, and improve symptoms of pain.
It speculates that the affective responses associated with pain reduction in the red+infrared LED group are caused by the proportional increase in the metabolic effect generated by the combined irradiation of the two light spectra. Since the two lengths tend to improve electrochemical activity and increase ATP resynthesis $^{14}$. In addition, irradiation with LED in the infrared spectrum promotes an increase in the concentration of oxyhemoglobin and total oxymyhemoglobin, which increases the availability of oxygen $^{18}$ that may favor aerobic metabolism.
Although the main factor for the emergence of neuropathic pain is assigned to metabolic stresses (ROS) $^{43}$, it is also emphasized that the vascular impairment, generated by reduced flow and hypoperfusion due to hyperglycemia, can lead to the failure of several tissues and contribute to the onset of neuropathic pain early $^{44}$.
Although there were no significant changes in blood flow, the clinical effect was also in favor of the irradiated groups.
Considering that oxidative stress plays a central role in the development of microvascular complications of diabetes $^{45}$, as well as a secondary variable, the blood flow of the posterior and dorsal tibial arteries was measured in order to support the context of blood circulation in the appearance of neuropathic pain. Although there was no significant improvement in the quantitative assessments of Doppler ultrasound, it is believed that photobiomodulation tends to improve blood flow and reduce arterial resistance, since there was an improvement from mild to moderate in the effect size in the irradiated groups, for the peak systolic velocity variables and the resistivity index in the three therapeutic intervention groups. The mechanisms proposed to explain the vascular changes induced by glucose and lipids in diabetes include the accelerated formation of advanced glycation end products (AGEs), activation of protein C kinase, inflammatory signaling, and oxidative stress $^{46}$. Accordingly, the deficits in the bioavailability of nitric oxide and the large concentrations of ROS are the main responsible for vasculopathy in diabetic patients, $^{47,48}$ responses that can be reversed with PBM.
According to our searches, in order to confront the findings of the influence of LED light on blood flow, there was an absence of studies that analyzed vascular resistance (IR) and improved blood flow (PVS) in patients with pain secondary neuropathic diabetes mellitus, what leads to a limitation in the discussion of the results presented.
In view of the results obtained, we believe that the irradiation time of three days is one of the factors that can be extended, allowing a longer response time of the tissues in face of photobiomodulation, allowing an effect that can generate changes that can be reflected at the level of macrocirculation. In addition, using different means of evaluation, such as microcirculation, tissue perfusion of the lower limbs, biomarkers of neuronal dysfunction, and systemic inflammation, could complement the analysis of neuropathic pain and vascular c condition, secondary to diabetes mellitus.
Thus, despite the positive limitations faced due to lack of equipment to better assess the painful polyneuropathy caused by type 2 diabetes mellitus, this study opens a new window for photobiomodulation by light-emitting diode (LED) in the red, infrared, or associated spectrum is now filled as a nonpharmacological therapeutic line for reducing the signs and symptoms of neuropathic pain in patients with type 2 diabetes with polyneuropathy of the lower limbs.
### ACKNOWLEDGEMENTS
This work was carried out with the support of the Coordination for the Improvement of Higher Education Personnel - Brazil (CAPES)
Conflict of Interest
The authors declare no conflicts of interest.
Funding
This study was financed in parts by the Coordination for the Improvement of Higher Education Personnel - Brazil (CAPES) - Finance Code 001
Ethics approval
Comité de Éticaem Experimentação com Seres Humanos do Hospital das Clínicas - FMRP/USP (no. 3.805.967)
Generating HTML Viewer...
References
48 Cites in Article
Jan Ulbrecht,Peter Cavanagh,Gregory Caputo (2004). Foot Problems in Diabetes: An Overview.
Andrew Boulton,Loretta Vileikyte,Gunnel Ragnarson-Tennvall,Jan Apelqvist (2005). The global burden of diabetic foot disease.
S Schreml,M Berneburg (2017). The global burden of diabetic wounds.
A Bello,S Biliaminu,K Wahab,E Sanya (2019). Distal symmetrical polyneuropathy and cardiovascular autonomic neuropathy among diabetic patients in Ilorin: Prevalence and predictors.
A Chan,E Wilder-Smith (2016). Small fiber neuropathy: Getting bigger! Muscle & nerve.
Kelly Gwathmey,Kathleen Pearson (2019). Diagnosis and management of sensory polyneuropathy.
Brian Callaghan,Hsinlin Cheng,Catherine Stables,Andrea Smith,Eva Feldman (2012). Diabetic neuropathy: clinical manifestations and current treatments.
J Leahy (2005). Pathogenesis of type 2 diabetes mellitus.
M Lotfy,J Adeghate,H Kalasz,J Singh,E Adeghate (2017). Chronic Complications of Diabetes Mellitus: A Mini Review.
R Marfella,F Nappo,L De Angelis,G Paolisso,M Tagliamonte,D Giugliano (2000). Hemodynamic effects of acute hyperglycemia in type 2 diabetic patients..
S Avetisov,N Chernenkova,Z Surnina (2017). Clinical features and diagnosis of diabetic polyneuropathy.
Mark Babizhayev,Igor Strokov,Valery Nosikov,Ekaterina Savel’yeva,Vladimir Sitnikov,Yegor E. Yegorov,Vadim Lankin (2015). The Role of Oxidative Stress in Diabetic Neuropathy: Generation of Free Radical Species in the Glycation Reaction and Gene Polymorphisms Encoding Antioxidant Enzymes to Genetic Susceptibility to Diabetic Neuropathy in Population of Type I Diabetic Patients.
D Pastore,M Greco,S Passarella (2000). Specific heliumneon laser sensitivity of the purified cytochrome c oxidase.
Michael Hamblin (2018). Mechanisms and Mitochondrial Redox Signaling in Photobiomodulation.
Vladimir Heiskanen,Michael Hamblin (2018). Photobiomodulation: lasers vs. light emitting diodes?.
P Brondon,I Stadler,R Lanzafame (2007). Melanin density affects photobiomodulation outcomes in cell culture.
Nadeem Wajih,Swati Basu,Kamil Ucer,Fernando Rigal,Aryatara Shakya,Elaheh Rahbar,Vidula Vachharajani,Martin Guthold,Mark Gladwin,Lane Smith,Daniel Kim-Shapiro (2019). Erythrocytic bioactivation of nitrite and its potentiation by far-red light.
S Linares,T Beltrame,C Ferraresi,Gam Galdino,A Catai (2020). Photobiomodulation effect on local hemoglobin concentration assessed by nearinfrared spectroscopy in humans.
Marcelie Rosso,Daniela Buchaim,Natália Kawano,Gabriela Furlanette,Karina Pomini,Rogério Buchaim (2018). Photobiomodulation Therapy (PBMT) in Peripheral Nerve Regeneration: A Systematic Review.
Chiung-Chyi Shen,Yi-Chin Yang,Tsung-Bin Huang,Shiuh-Chuan Chan,Bai-Shuan Liu (2013). Low-Level Laser-Accelerated Peripheral Nerve Regeneration within a Reinforced Nerve Conduit across a Large Gap of the Transected Sciatic Nerve in Rats.
M Hennessy,M Hamblin (2017). Photobiomodulation and the brain: a new paradigm.
Dos Santos,S Serra,A Stancker,T (2017). Effects of Photobiomodulation Therapy on Oxidative Stress in Muscle Injury Animal Models: A Systematic Review.
Cleber Ferraresi,Danilo Bertucci,Josiane Schiavinato,Rodrigo Reiff,Amélia Araújo,Rodrigo Panepucci,Euclides Matheucci,Anderson Cunha,Vivian Arakelian,Michael Hamblin,Nivaldo Parizotto,Vanderlei Bagnato (2016). Effects of Light-Emitting Diode Therapy on Muscle Hypertrophy, Gene Expression, Performance, Damage, and Delayed-Onset Muscle Soreness.
E Miranda,L De Oliveira,F Antonialli,A Vanin,T De Carvalhopde,Leal-Junior Ec (2015). Phototherapy with combination of super-pulsed laser and lightemitting diodes is beneficial in improvement of muscular performance (strength and muscular endurance), dyspnea, and fatigue sensation in patients with chronic obstructive pulmonary disease.
Fernando Nampo,Vinícius Cavalheri,Francyelle Dos Santos Soares,Solange De Paula Ramos,Enilton Camargo (2016). Low-level phototherapy to improve exercise capacity and muscle performance: a systematic review and meta-analysis.
K Desmet,D Paz,J Corry (2006). Clinical and experimental applications of NIR-LED photobiomodulation.
J Eells,M Wong-Riley,J Verhoeve (2004). Mitochondrial signal transduction in accelerated wound and retinal healing by near-infrared light therapy.
M Wong-Riley,H Liang,J Eells (2005). Photobiomodulation directly benefits primary neurons functionally inactivated by toxins: role of cytochrome c oxidase.
Lorne Zinman,Mylan Ngo,Eduardo Ng,Khin Nwe,Sven Gogov,Vera Bril (2004). Low-Intensity Laser Therapy for Painful Symptoms of Diabetic Sensorimotor Polyneuropathy.
R Moreira,A Castro,M Papelbaum (2005). Tradução para o português e avaliação da confiabilidade de umaescala para diagnóstico da polineuropatia distal diabética.
A Milech,A Angelucci,A Golbert,A Matheus,Ajf Carrilho,A Ramalho (2016). Diretrizes da sociedadebrasileira de diabetes (2015-2016.
A Boulton,D Armstrong,S Albert (2008). Comprehensive foot examination and risk assessment.
B Perkins,D Olaleye,B Zinman,V Bril (2001). Simple screening tests for peripheral neuropathy in the diabetes clinic.
R Van Seventer,C Vos,W Meerding,I Mear,M Le Gal,D Bouhassira,F Huygen (2010). Linguistic validation of the DN4 for use in international studies.
Jesus De,R Guirro,G De Carvalho,A Gobbi,F De Oliveira Assuncao,Souza De,N Borges,L Bachmann (2020). Measurement of Physical Parameters and Development of a Light Emitting Diodes Device for Therapeutic Use.
U Dongre,V Meshram,S Pitale Oxidative Stress and 3243 A/G Mitochondrial Dna Mutation In Maternally Inherited Type 2 Diabetes Mellitus.
R Sandireddy,V Yerra,A Areti,P Komirishetty,A Kumar (2014). Neuroinflammation and oxidative stress in diabetic neuropathy: futuristic strategies based on these targets.
Gérard Said (2007). Diabetic neuropathy—a review.
S Cg,A Maiya,H Hande,S Vidyasagar,K Rao,K Rajagopal (2015). Efficacy of low level laser therapy on painful diabetic peripheral neuropathy.
A Gobbi,G De Carvalho,A Sapalo,Jesus De,R Guirro (2021). Acute application of photobiomodulation does not bring important gains for the muscular performance and functionality of diabetic individuals.
Atousa Janzadeh,Farinaz Nasirinezhad,Masoume Masoumipoor,Seyed Jameie,Parisa Hayat (2016). Photobiomodulation therapy reduces apoptotic factors and increases glutathione levels in a neuropathic pain model.
Caroline Volpe,Pedro Villar-Delfino,Paula Dos Anjos,José Nogueira-Machado (2018). Cellular death, reactive oxygen species (ROS) and diabetic complications.
Si Kim,Kang Park,Jinse Park,Sam Ha,Sung Kim,Byung Lee,Kyong Shin (2019). Vascular factors and neuropathy in lower limb of diabetic patients.
Caroline Domingueti,Luci Dusse,Maria Carvalho,Lirlândia De Sousa,Karina Gomes,Ana Fernandes (2016). Diabetes mellitus: The linkage between oxidative stress, inflammation, hypercoagulability and vascular complications.
M Potenza,S Gagliardi,C Nacci,M Carratu,M Montagnani (2009). Endothelial dysfunction in diabetes: from mechanisms to therapeutic targets.
Ferdinando Giacco,Michael Brownlee (2010). Oxidative Stress and Diabetic Complications.
U Hink,H Li,H Mollnau (2001). Mechanisms underlying endothelial dysfunction in diabetes mellitus.
No ethics committee approval was required for this article type.
Data Availability
Not applicable for this article.
How to Cite This Article
André Timóteo Sapalo. 2026. \u201cAcute Effect of Photobiomodulation with LED in Individuals with Diabetic Neuropathy\u201d. Global Journal of Medical Research - F: Diseases GJMR-F Volume 24 (GJMR Volume 24 Issue F1).
Explore published articles in an immersive Augmented Reality environment. Our platform converts research papers into interactive 3D books, allowing readers to view and interact with content using AR and VR compatible devices.
Your published article is automatically converted into a realistic 3D book. Flip through pages and read research papers in a more engaging and interactive format.
Subject: Global Journal of Medical Research - F: Diseases
Authors:
André Timóteo Sapalo, Aline Gobbi, Gabriela de Carvalho, Elaine Caldeira de Oliveira Guirro, Rinaldo Roberto de Jesus Guirro (PhD/Dr. count: 0)
Our website is actively being updated, and changes may occur frequently. Please clear your browser cache if needed. For feedback or error reporting, please email [email protected]
Thank you for connecting with us. We will respond to you shortly.